molar concentration formula
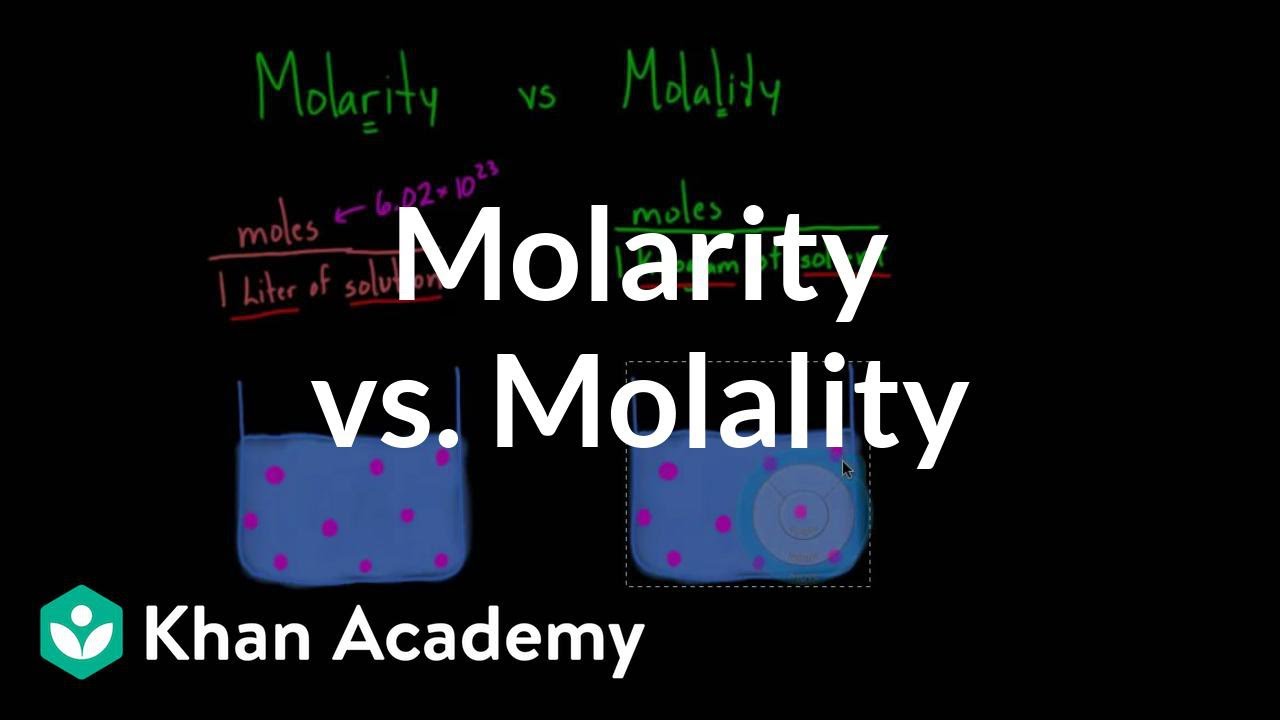
What is molar concentration?
Molar concentration can be used to convert between the mass or moles of solute and the volume of the solution. In real life, we often encounter substances that are mixtures of different elements and compounds. One example of a mixture is the human body. Did you know that the human body is approximately 57 % water by mass?
How do you find the molarity of a solution?
Find the molarity and volume of your solution. Make sure that the units for the volume are the same as for the volume part of the molarity (e.g., mL and mol/mL). Multiply the volume by the molarity. This is the number of moles present. Is molarity the same as concentration? Molarity is not the same as concentration, although they are very similar.
How do you abbreviate molarity?
Molarity has units of mol liter , which can be abbreviated as molar or M (pronounced "molar" ). The molar concentration of the solute is sometimes abbreviated by putting square brackets around the chemical formula of the solute. For example, the concentration of chloride ions in a solution can be written as [ Cl −] .
How do you calculate molar concentration of sulfuric acid?
We can now plug in the moles of sulfuric acid and total volume of solution in the molarity equation to calculate the molar concentration of sulfuric acid: [ H 2 SO 4] = mol solute L of solution = 0.02401 mol 0.050 L = 0.48 M Concept check: What is the molar concentration of H + ions in a 4.8 M H 2 SO 4 solution?

Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations

Molarity Made Easy: How to Calculate Molarity and Make Solutions

Molarity Practice Problems
|
NovaTeinBio Converting Protein Mass Concentration to Molar
%20or%20vice%20versa.pdf |
|
Thermo Fisher Scientific
Application of a molar extinction coefficient in the calculation yields an expression of concentration in terms of molarity:. |
|
Chapter 1 Fundamentals of Mass Transfer
In this equation c is the total molar concentration |
|
Chapter 4: Calculations Used in Analytical Chemistry
particles as represented by a chemical formula. Molar analytical concentration is the total number of moles of a solute. |
|
Water Potential (?)
Construct a graph by plotting % increase or decrease in mass and sucrose molarity. Determine the molar concentration of the potato core. |
|
Calculating Nucleic Acid or Protein Concentration Using the GloMax
07-Aug-2009 Where A = absorbance ? = molar extinction coefficient |
|
UNITS OF CONCENTRATION.pdf
To convert concentrations in mg/L (or ppm in dilute solution) to molarity equations to solve for the number of moles of analyte in given sample volume. |
|
B75 Lab manual_ss_pt1
To determine how to mix up a smaller volume of solution follow the equation below. Solutions of Differing Molarity Concentrations. Molarity Concentration |
|
Beers Law: Determining the Concentration of a Solution
where A is the absorbance (unitless) ? is the molar absorptivity coefficient (M-1cm-1) |
|
Laboratory Math II: Solutions and Dilutions
What units you will be reporting the concentration in. ? If molarity or normality the molecular or formula weight of the substance (solute). |
|
Chapter 4: Calculations Used in Analytical Chemistry - İYTE
particles as represented by a chemical formula ➢ It is the amount of Molar analytical concentration is the total number of moles of a solute, regardless of its |
|
Calculations involving concentrations, stoichiometry Metric System
(relative) formula weight (FW) Ionic salts: The molar mass of a substance in grams has the Example: Calculate molar concentration of Na2HPO4 solution c |
|
Molar Concentration of Solutions
Molar Concentration of Solutions 1 What is the molarity of a solution made by dissolving 3 00 moles of NaCl in enough water to make 6 00 liters of solution? 2 |
|
SOLUTIONS, CONCENTRATIONS, STOICHIOMETRY - Cal State LA
1 atom O(16 0 amu/atom) = 16 0 amu Formula Weight = 18 0 amu/formula Molar Mass = 18 0 grams/mol III Units of Concentration Concentration is expressed |
|
UNITS OF CONCENTRATION
To convert concentrations in mg/L (or ppm in dilute solution) to molarity, divide equations to solve for the number of moles of analyte in given sample volume |
|
Molarity Concentration Activity
Molarity (M) measures concentration as moles of solute per liters of solution The formula equation for Molarity can be found on reference Table T The unit for |
|
Moles and molarity
concentration in yet more ways: 0 5 mol/l = 0 5 mmol/ml = 500 µmol/ml = 500,000 pmol/µl Now, let's say you wanted to determine the amount of cholesterol in |
|
Lab Math Solutions, Dilutions, Concentrations and Molarity - APHL
25 avr 2016 · ➢Serial dilutions ➢Making fixed volumes of specific concentrations from liquid (= parts per hundred) ➢Molar solutions (unit=M=moles/L) |
|
Converting protein mass concentration to molar concentration, or
From molar concentration to mass concentration: The simple formula is: ( µg/mL ) = ( µM ) * ( MW in KD) , ( |

















/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)