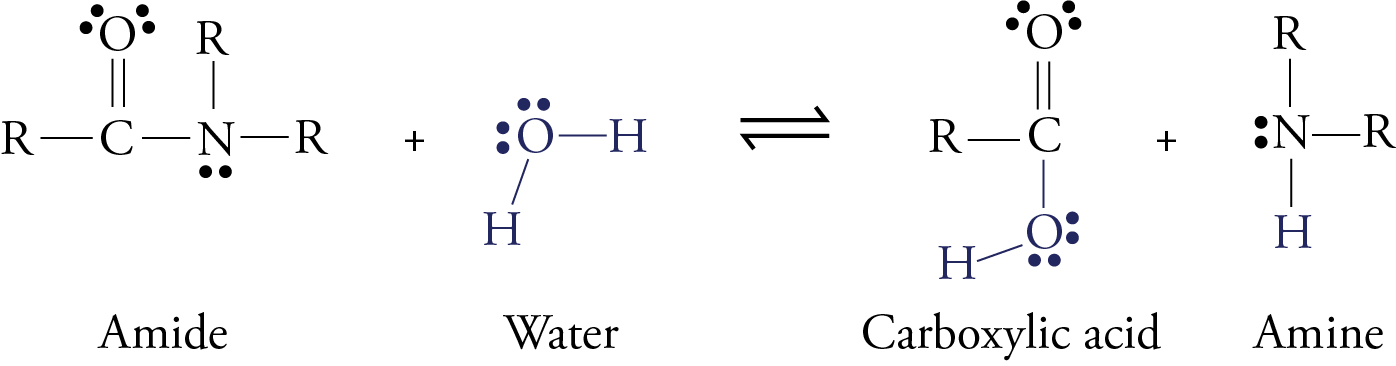net ionic equation for hydrolysis
|
REACTIONS OF SALTS WITH WATER
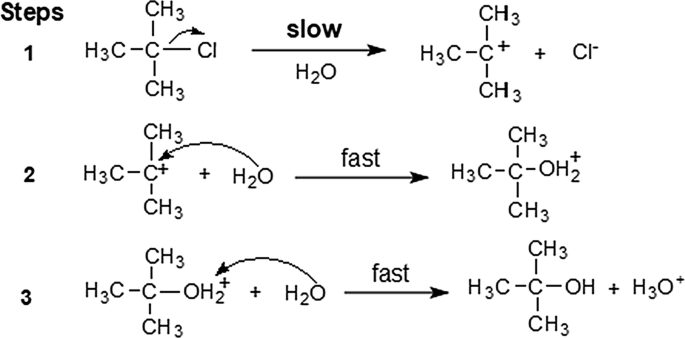
Hydrolysis as applied to water solutions of inorganic compounds can be defined as the reaction of water with one or both ions of a salt to form a weak acid and a OH- or a weak base and H+ ion or both For example - In a solution of NaCN: - |
|
1) What is the net ionic equation for the hydrolysis of NH 4ClO4? (1
2) What is the equilibrium constant expression for the predominant reaction between the hydrogen oxalate ion HC2O4 - and water? (1 mark) |
Hydrolysis: the reaction of a substance with water.
Both cations and anions can undergo hydrolysis, but not all ions react in this fashion.
When an ionic salt dissolves in water, both the free cation and the free anion are formed.
What is the net ionic equation for the hydrolysis of nh4cl?
Answer and Explanation:
The equation of hydrolysis of ammonium chloride in water is given as; N H 4 C l ( s ) + H 2 O → N H 4 ( a q ) + + C l ( a q ) − , It gets dissociated completely.
Also, it is known that hydronium ion gives hydrogen ion and water on dissociation.

How to Write the Net Ionic Equation for NH4Cl + NaOH = NaCl + H2O + NH3

Acid Base Neutralization Reactions & Net Ionic Equations

How to Write Complete Ionic Equations and Net Ionic Equations
|
Hydrolysis: Examples:
Hydrolysis: - Reaction between a salt (ion or ions in a salt) and water to produce an acidic or basic solution. - Net ionic equations for hydrolysis:. |
|
1) What is the net ionic equation for the hydrolysis of NH 4ClO4? (1
CHEMISTRY 12 – HYDROLYSIS & TITRATIONS EXTRA PRACTICE oxalate ion HC2O4 ... b) Write the equation for the hydrolysis reaction that occurs. (1 mark). |
|
Exp 17 REACTIONS OF SALTS WITH WATER F 08
2. THE HYDROLYSIS REACTIONS. Write net-ionic equations for the reaction of each salt with water below to illustrate the observed pH given on page 5. |
|
Untitled
salts both anion and cation hydrolyze and the resulting pH depends on which to write a net ionic equation for each hydrolysis. |
|
Sec 4.13 – Hydrolysis (notes)
Reaction between a salt (ion or ions in a salt) and water to produce an acidic or basic solution. - Net ionic equations for hydrolysis:. |
|
Untitled
Write dissociation equations for each of the following salts Write the net ionic equation for the predominant hydrolysis reaction when each of the. |
|
911 Metallurgist
Hydrolysis: - Reaction between a salt (ion or ions in a salt) and water to produce an acidic or basic solution. - Net ionic equations for hydrolysis:. |
|
EXPERIMENT 11
b) Write net ionic equations representing the hydrolysis of salts + ion reacts with water (called hydrolysis) according to the equation:. |
|
During Class Invention Salts I
Salts are ionic compounds which are formed in a neutralization reaction between an acid and a base. (aq) net ionic equation b. Write the equation which ... |
|
Hydrolysis - SSS Chemistry - D Colgur
The formula equations for some of these are: SO2(g) + H2O(l) → H2SO3 (aq) (sulphurous acid) 2NO2(g) + H2O(l) → HNO3 (aq) + HNO2 (aq) (nitric and nitrous acids) Once these acids are formed, they can ionize (strong ones 100 , weak ones < 100 ) to form H3O+ ions |
|
Reactions of Salt with Water
Hydrolysis as applied to water solutions of inorganic compounds, can be defined as the Write the net ionic equation for the hydrolysis of NaC2H3O2 2 |
|
1) What is the net ionic equation for the hydrolysis of NH 4ClO4? (1
2) What is the equilibrium constant expression for the predominant reaction between the hydrogen oxalate ion, HC2O4 -, and water? (1 mark) 3) Which of the |
|
Hydrolysis - Arcuric Acid
spectators are eliminated in net ionic equations for hydrolysis Process – if given salt (dissociate → eliminate → evaluate) 1 Write dissociation equation 2 |
|
Hydrolysis: - 911 Metallurgist
spectators are eliminated in net ionic equations (NIE's) for hydrolysis Process – if given salt (dissociate → eliminate → evaluate) 1 Write dissociation equation |
|
Key
a) Salt K2CO: Dissociation Equation Chemistry 12-Worksheet 4-5--Hydrolysis Write the net ionic equation for the predominant hydrolysis reaction when |
|
Chemistry 12 HYDROLYSIS Name(s)
Cation Hydrolysis Equation 2 Write a balanced Net-Ionic Hydrolysis Equation for each Anion which hydrolyzed in Part 1 of the lab (Don't repeat any) Salt |
|
413 Salts and Hydrolysis - VSB BLOGS
ii) The ion will react with water to form a basic solution if the ion is on the base ( right) side of the Table of Relative Strengths p 334 eg Na2SO3 (s) → Na+ |
|
Write the hydrolysis reaction(s) for each of the following substances
solution is weakly basic Note: Stoichiometric coefficients only balance the equation they are in They do not carry through to subsequent reactions |
|
Experiment 31
Net ionic equation if there is hydrolysis CuCl2 Cupric chloride Net ionic equation if there is hydrolysis K2CO3 Potassium carbonate Net ionic equation if there |


![Kinetics of hydrolysis of diltiazem - [PDF Document] Kinetics of hydrolysis of diltiazem - [PDF Document]](https://i1.rgstatic.net/publication/327532572_Kinetic_study_of_hydrolysis_of_ethyl_acetate_using_caustic_soda/links/5b93bcd84585153a53083d5e/largepreview.png)





















![Kinetics of hydrolysis of diltiazem - [PDF Document] Kinetics of hydrolysis of diltiazem - [PDF Document]](https://0.academia-photos.com/attachment_thumbnails/30726055/mini_magick20190426-19222-wexme7.png?1556320662)