nomenclature of amides pdf
|
Nomenclature of Organic Chemistry IUPAC Recommendations and
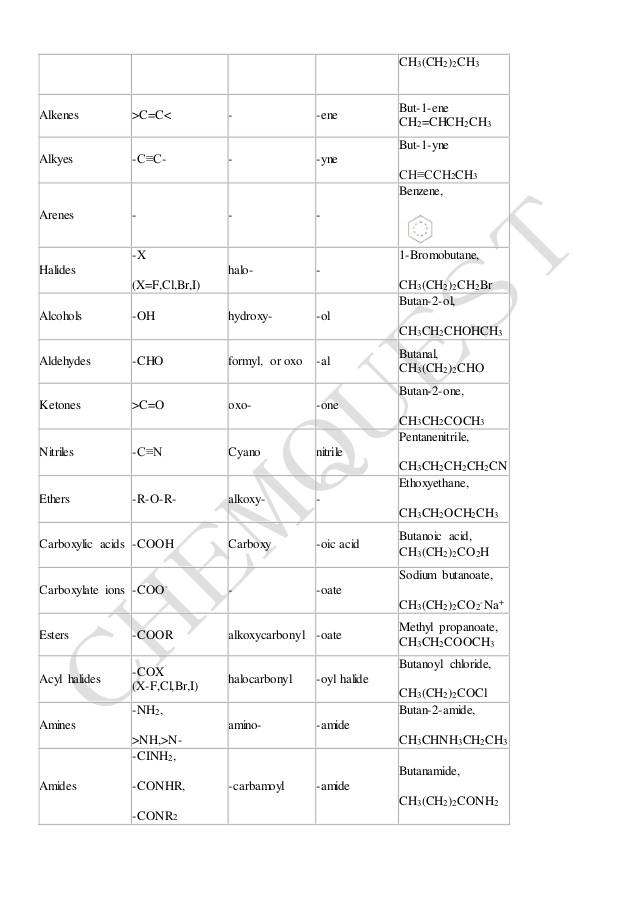
Substitution of amides is expressed by prefixes; numerical N and N′ locants are used as required N-Substitution of amides follows the substitution rules for carboxylic acids described in P-65 1 2 4 Examples: 3-chloropropanamide (PIN) (not 3-chloropropionamide; no substitution on propionamide) 4-chloro-NN-dimethylbutanamide (PIN) |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Nomenclature Primary amines are named in systematic (IUPAC) nomenclature by replacing the -e of the corresponding parent alkane with -amine In common nomenclature they are named as alkylamines Simple secondary and tertiary amines are named in common nomenclature by designating the organic groups separately in front of the word amine |
|
Classification and Nomenclature of Amines
• Learn to recognize the amine and amide functional groups • Learn the IUPAC system for naming amines and amides • Learn the important physical properties of the amines and amides • Learn the major chemical reactions of amines and amides and learn how to predict the products of amide synthesis and hydrolysis reactions |
|
Naming Amines and Amides (rules)
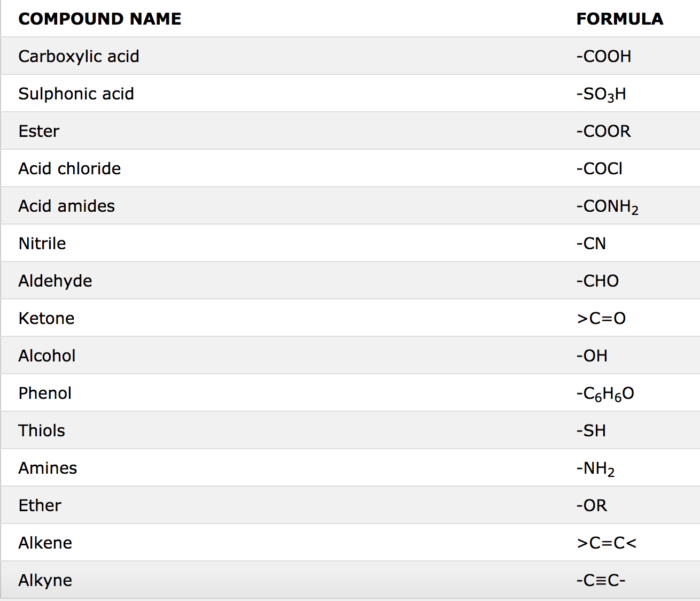
Amides Classification of amides by the number of alkyl chains: Amides are named as alkanamides IUPAC replaces –oic acid ending with –amide Common names replace -ic acid ending with –amide O Methanamide (IUPAC) H—C—NH2 Formamide (common) O |
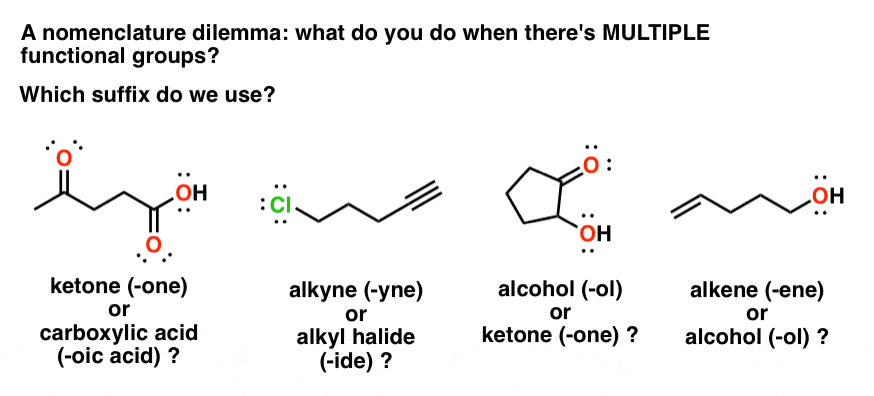
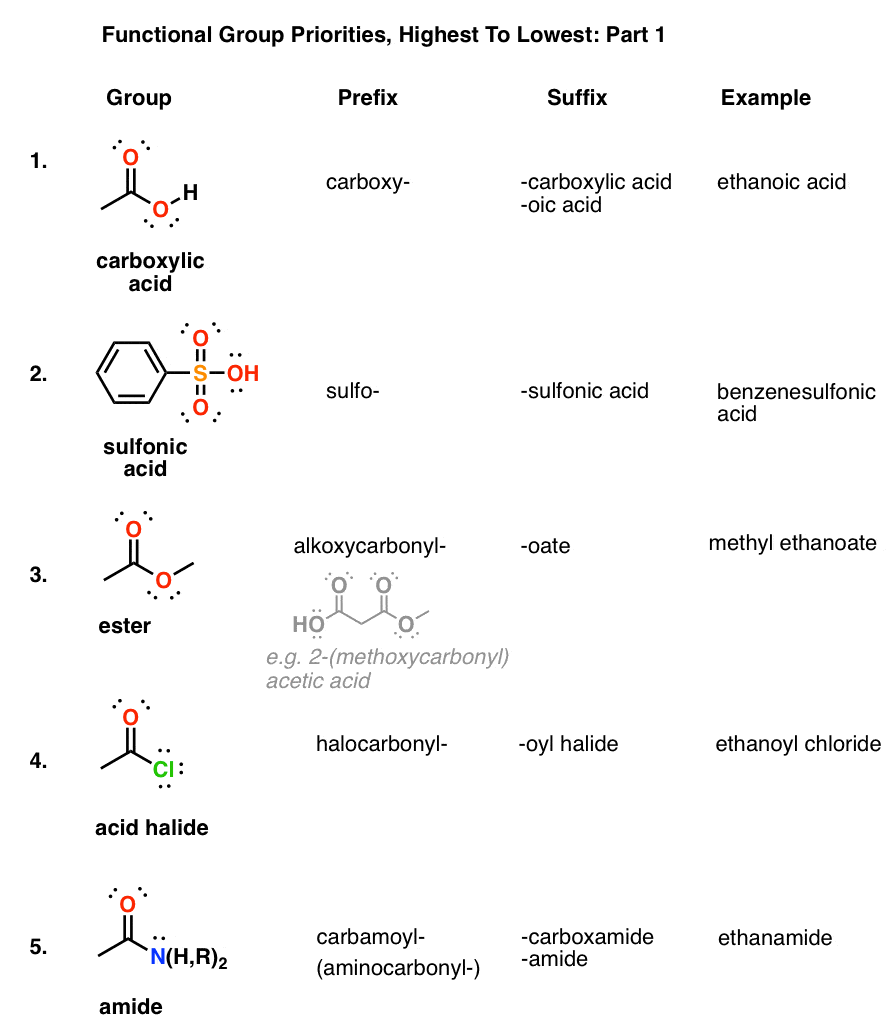
How are amides ranked in IUPAC nomenclature?
Preferred IUPAC names for these two acids and their derivatives are formed systematically. Amides are ranked in the same way as the corresponding acids (see P-42). Thus, in substitutive nomenclature, amides from carboxylic acids, including formamide, are senior to urea.
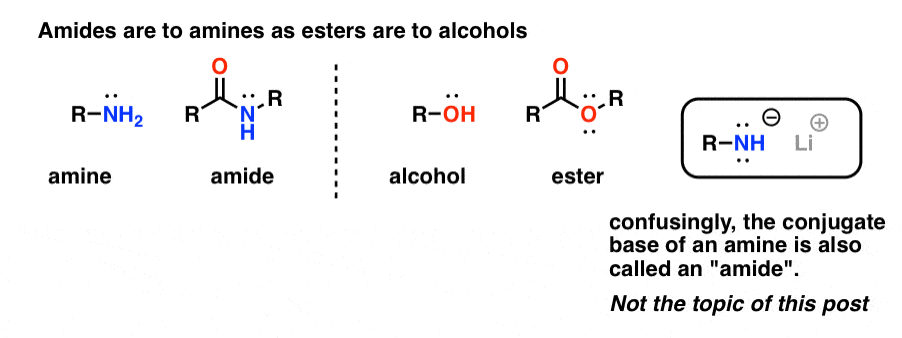
Are amines and amides the same?
Amines and amides come from ammonia (replacing the H’s). Are named as alkylamines. List the names of the alkyl groups bonded to the N atom in alphabetical order in front of amine. CH3 Amines are named as alkanamines. The –e in the alkane name of the longest chain is changed to –amine.
What are amides called?
Amides are named as alkanamides. IUPAC replaces –oic acid ending with –amide. Common names replace -ic acid ending with –amide. An alkyl group bonded to the N atom is named as N-alkyl in front of the amide name. H H • The amide of benzene is named benzamide.
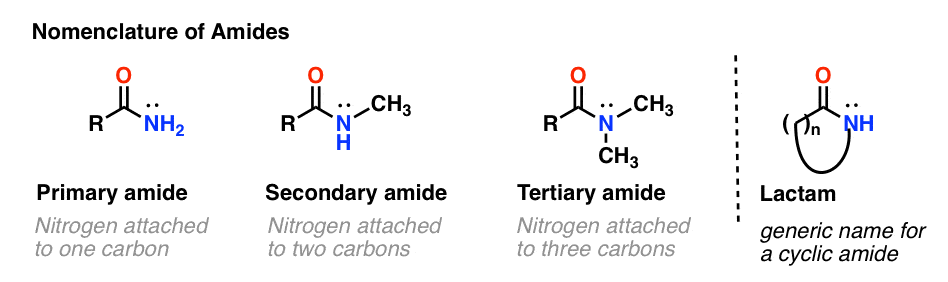
What naming conventions should be used for secondary and tertiary amides?
There are special naming conventions that need to be used for secondary and tertiary amides. Primary amides follow similar naming conventions to other functional groups. Secondary and tertiary amides use an italicised ‘N-‘ to locate the side chains in relation to the amide group.

Nomenclature and properties of amides Organic chemistry Khan Academy

Naming Amides

Naming Amines
|
NOMENCLATURE EN CHIMIE ORGANIQUE 1. Hydrocarbures (HC
NOMENCLATURE EN CHIMIE ORGANIQUE. La nomenclature permet de : a) Trouver le nom d'une molécule Groupe principal : Suffixe = -amide. -carboxamide. |
|
I. Connaissance sur les amides II. Les différentes classes damides
) remplace le groupe hydroxyle (–OH) de la molécule d'acide carboxylique. Page 2. III. Les composés du carbone. 131. III. Nomenclature des |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the products of amide synthesis and hydrolysis reactions. • Learn some of the |
|
Sch_206-amides.pdf
IUPAC Nomenclature of Amides. Primary Amides. 4. An amide is named based on the recognition that it is a composite of a carboxylic acid and ammonia or an |
|
Page 1 sur 14 Les Acides aminés et les protéines
1. Nomenclature. Les acides aminés sont des ions dipolaires (zwitterion) dans leur état naturel : En effet ils sont solubles dans l'eau |
|
Chapter 17: Amines and Amides
* Because the suffix here is -diamine which starts with a consonant the terminal -e of the alkane name is retained. Naming aromatic amines. 17.4 Isomerism for |
|
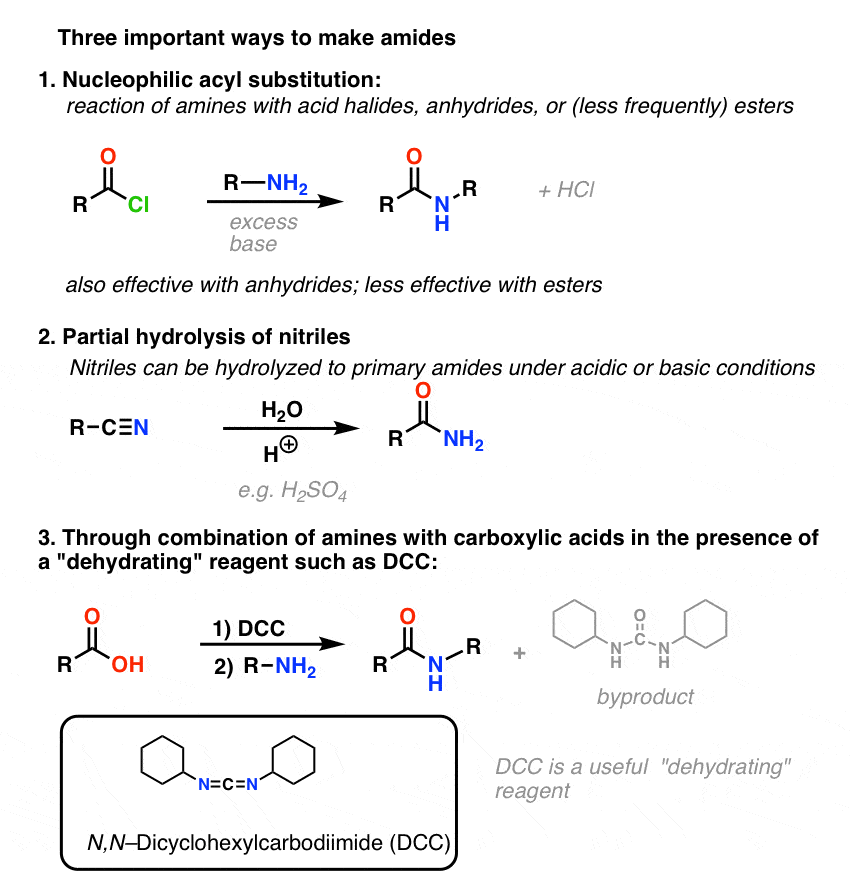
CHM 2520 Chimie organique II
considérons la préparation et les réactions de quatre dérivés : 1. chlorures d'acide. 2. anhydrides. 3. esters. 4. amides. |
|
NOMENCLATURE ET ISOMERIE
L'atome de carbone du groupe -. COOH porte toujours le numéro 1; comme pour les aldéhydes et les amides l'indice 1 est omis. acide pentanoïque et NON acide |
|
La fonction amide.pdf
R R' et R" sont des atomes d'hydrogène ou des groupements alkyles. 2. NOMENCLATURE. Le nom de l'amide dérive de celui de l'alcane de même chaîne carbonée en |
|
Bi102-2017-acides amines.pdf
Thréonine Thr T. Nomenclature des acides aminés : On utilise un code soit à 3 lettres soit à 1 lettre. II-Structure des acides aminés. Figure 6 |
|
Amides
IUPAC Nomenclature of Amides Primary Amides 4 An amide is named based on the recognition that it is a composite of a carboxylic acid and ammonia or an |
|
Chapter 17: Amines and Amides
17 3 Nomenclature for Amines Atomic grouping Suffix -amine Prefix amino Position in chain anywhere General formula CnH2n+3N Rule 1: Select as the |
|
I Connaissance sur les amides II Les différentes classes damides
) remplace le groupe hydroxyle (–OH) de la molécule d'acide carboxylique Page 2 III Les composés du carbone 131 III Nomenclature des |
|
Nomenclature en chimie organique - UniNE
NOMENCLATURE EN CHIMIE ORGANIQUE La nomenclature permet de : a) Trouver le Amines aromatiques : benzènamines (nom courant : anilines) NH2 |
|
Esters, Amides, and Related Molecules - Organic Chemistry
of acid chlorides into carboxylic acids, esters, amides, or anhydrides (Figure We describe nomenclature, along with the physical and chemical properties of the |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be carboxylic acids is based on the hydrocarbon naming method |
|
Quelques exercices sur les amides - TuniSchool
- Disubstitué veut dire deux atomes d'hydrogène, liés à l'atome d'azote, sont remplacés par deux groupes alkyles R1 et R2 • Nomenclature : ❖ Amide non |
|
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides
Naming Amines Simple amines • are named as alkylamines • List the names of the alkyl groups bonded to the N atom in alphabetical order in front of amine |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Simple secondary and tertiary amines are named in common nomenclature by designating the organic groups separately in front of the word amine |