normality and molarity
What is molarity in chemistry?
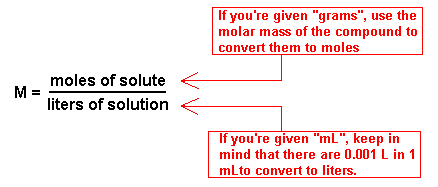
Molarity is the most commonly used measure of concentration. It is expressed as the number of moles of solute per liter of solution. For example, a 1 M solution of H 2 SO 4 contains 1 mole of H 2 SO 4 per liter of solution. H 2 SO 4 dissociates into H + and SO 4- ions in water.
How do you calculate normality from molarity?
Alternatively, you can use this equation to make solutions with different volumes: Initial Normality (N 1) × Initial Volume (V 1) = Normality of the Final Solution (N 2) × Final Volume (V 2) It’s easy to calculate normality from molarity for an acid or base solution if you know the number of hydrogen (acid) or hydroxide (base) ions produced.
What is normality in chemistry?
Normality is also called equivalent concentration. It is indicated by the symbol “N” or “eq/L” (equivalents per liter). To find the gram equivalent weight, you need to know how many hydrogen ions (H + or H 3 O + ), hydroxide ions (OH – ), or electrons (e –) are transferred in a reaction or you need to know the valence of the chemical species.
What is normality in titration?
Normality can be defined as the gram equivalent of solute dissolved in one cubic decimeter or one litre of solution. The symbol of normality is N. It is most preferred during titration calculations. Normal solutions have normality equal to unity. \\ (\\begin {array} {l}Normality =\\frac {Number\\ of\\ mole\\ equivalent} {1 L\\ of\\ solution}\\end {array} \\)

How To Calculate Normality & Equivalent Weight For Acid Base Reactions In Chemistry
![Molarity Normality and Molality [Tricks] Mole Concept in Solutions Molarity Normality and Molality [Tricks] Mole Concept in Solutions](https://pdfprof.com/FR-Documents-PDF/Bigimages/OVP.Bd6uVhZdyNglcdxeJm55bgHgFo/image.png)
Molarity Normality and Molality [Tricks] Mole Concept in Solutions

How to calculate normmality in chemistry?
|
Molarity and Normality
Normality is always a multiple of molarity. It describes the “equivalent” moles of reactants involved in chemical reactions. An “equivalent” is an older |
|
TITRIMETRIC ANALYSIS
is the molarity (M). Therefore N = a M … (vii). Equation (vii) is the expression for the relationship between normality and molarity. By using equation (vii) |
|
MOLARITY(M)MOLALITY (m)& NORMALITY(N).pdf
MOLALITY%20(m)&%20NORMALITY(N).pdf |
|
Unit 1 Units of measurements of solutes in solution e.g. normality
A standard solution contains accurately known concentration of solute. We can express the concentration in molality molarity |
|
Molarity Molality and Normality
12-Jan-2006 Molarity is the number of moles of a solute dissolved in a liter of solution. A molar solution of sodium chloride is made by placing 1 mole of a ... |
|
Molarity Molality and Normality
12-Jan-2006 Molarity is the number of moles of a solute dissolved in a liter of solution. A molar solution of sodium chloride is made by placing 1 mole of a ... |
|
Untitled
➢Both Normality and Molarity are measures of concentration . ➢Molarity is a measure of the no. of moles per liter of solution . ➢Normality changes depending |
|
Concentrations of Common Commercial Acids and Bases
Gravity Molarity Normality. Reagent. Percent. (w/w). To Prepare. 1L of 1 Molar. Solution. Acetic Acid Glacial (CH3COOH) 60.05. 1.05. 17.4. 17.4. 99.7%. 57.5mL. |
|
UNITS OF CONCENTRATION.pdf
Some of these are listed below. Molarity M = moles solute/liter of solution. Normality |
|
Molarity & Normality
contains 0.25 mol of sodium hydroxide in every litre of solution. Page 25. Calculation of Molarity. To calculate the molarity of a solution |
|
Molarity Molality and Normality
12 jan 2006 · The first three: molality molarity and normality are dependant upon the mole unit The last two: percent by volume and percent by weight have |
|
Both Normality and Molarity are
?Both Normality and Molarity are measures of concentration ?Molarity is a measure of the no of moles per liter of solution |
|
Some Basic Concepts of Chemistry
Normality (N) It is the number of gram equivalent of solute present in one litre of solution : N V = Eq in litres ( ) (i) Molarity (M) It is the moles |
|
MOLARITY(M)MOLALITY (m)& NORMALITY(N)pdf
MOLALITY%2520(m)%26%2520NORMALITY(N).pdf |
|
Lectures-1-2pdf - Cal State LA
Molarity (M) = # moles solute/#liters of solution The normality of permanganate ion is five times its molarity because MnO4 - ion accepts 5 electrons |
|
1)Solution: A homogenous mixture of two or more - VG Vaze College
ii) Molarity iii) Molality iv) Mole fraction Ans: i) Normality: It is defined s the number of gram equivalents of the solute dissolved in one dm |
|
Molarity Molality Normality and Mass Percent Worksheet IIpdf
Molarity = Moles of solute / Liters of Solution (abbreviation = M) Molality = Moles of solute / Kg of Solvent (abbreviation = m) Normality = number of |
|
Molarity, Molality and Normality
12 jan 2006 · substance equals the gram-formula mass or the gram-molar mass This equals the molarity and normality are dependant upon the mole unit |
|
IMPORTANT CHEMICAL CONCEPTS: SOLUTIONS - Cal State LA
The definition is selected so that the formula weight (in amu) and the molar mass ( The normality of permanganate ion is five times its molarity, because MnO4 |
|
Molarity & Normality
➢Both Normality and Molarity are measures of concentration ➢Molarity is a measure of the no of moles per liter of solution ➢Normality changes depending |
|
Solutions - UTC
Find Molarity (mol/L) of ethanol solution Note: In very dilute aqueous solution the molarity ~ molality Normality = equivalents of solute/ L of solution |
|
Concentration units
Molarity (M) Normality (N)? • N = (no of gram equivalent / Volume ) • N = * • N = M * Valency • Normality = Molarity * valency Wt of solute M wt of solute |
|
Stoichiometric calculations
Molarity • Molarity is M= number of moles per liter (mole/L) or millimoles per Normality and equivalents dependent as molar and normal concentrations |
|
1 GENERAL BIOCHEMISTRY - NIOS
Equivalent weight 2 Molarity 3 Molality 4 Normality 5 Percent solution ( weight/weight) 6 Molarity is based on the volume of solution containing the solute |
|
DRPUSALKAR PROBLEMS BASED ON - VG Vaze College
5 2) Explain the following terms: i) Normality ii) Molarity iii) Molality iv) Mole fraction Ans: i) Normality: It is defined s the number of gram equivalents of the solute |


:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)

































![Normality n Molarity - [PDF Document] Normality n Molarity - [PDF Document]](https://doubtnut-static.s.llnwi.net/static/ss/web/292257.jpg)