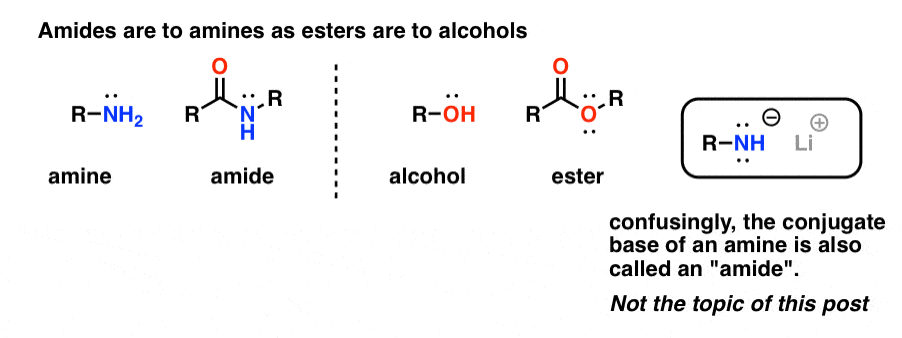
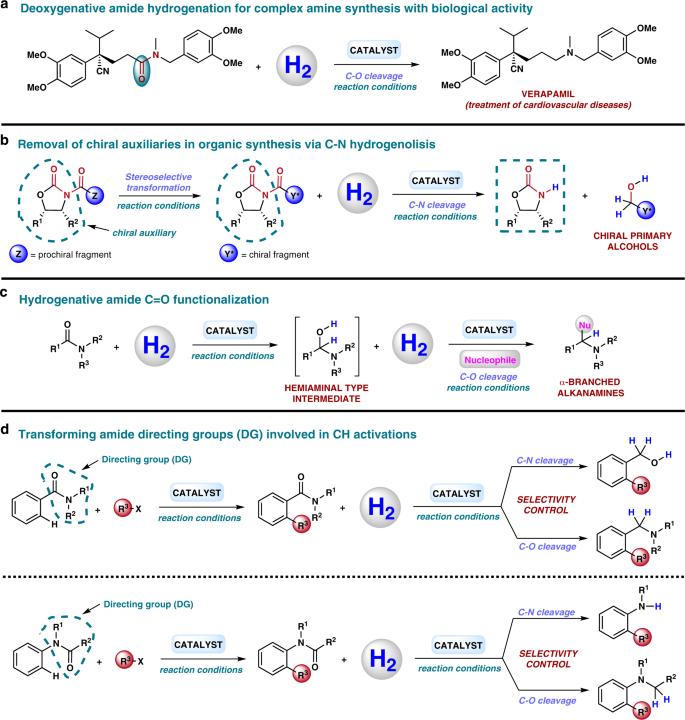
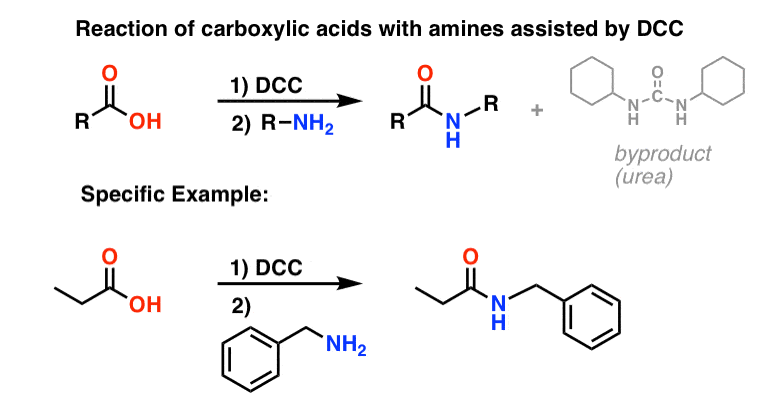
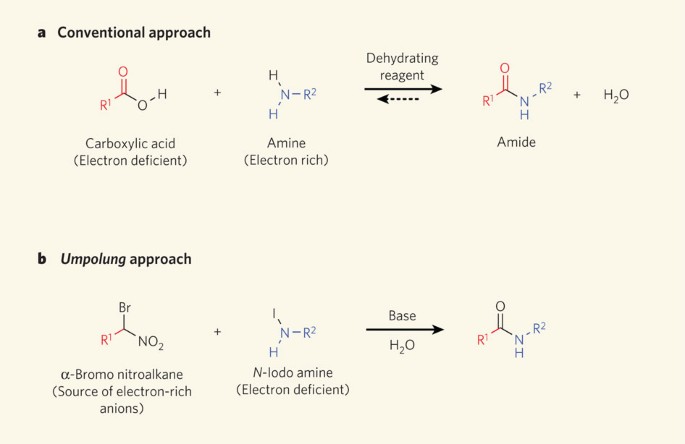
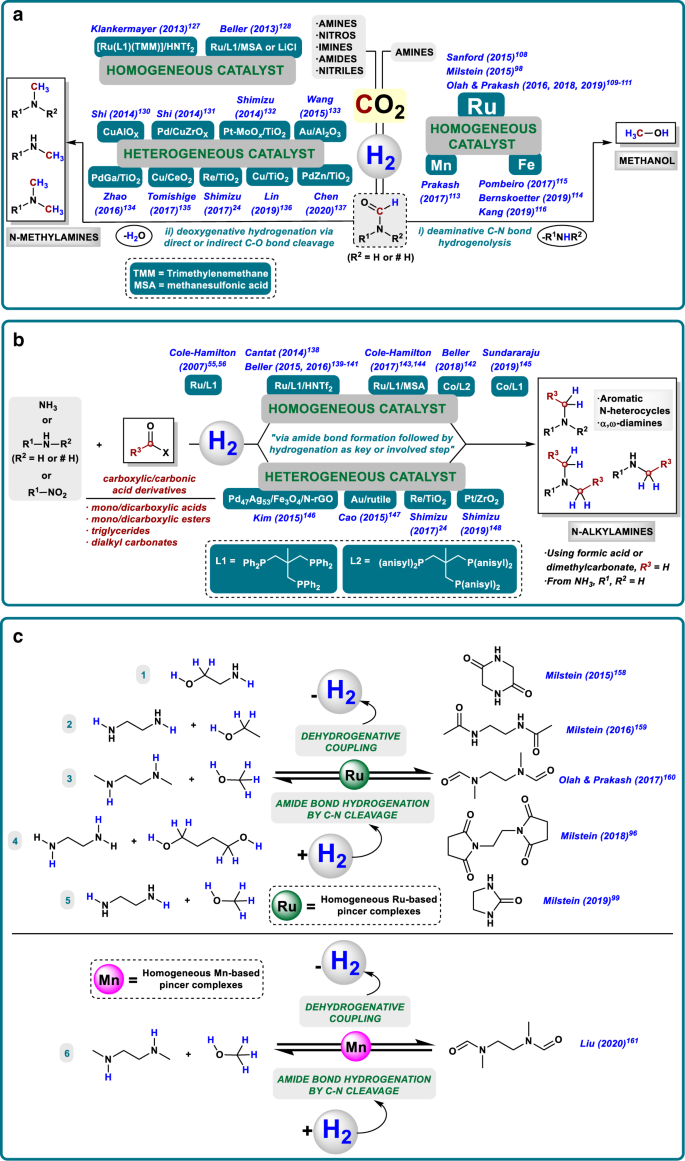
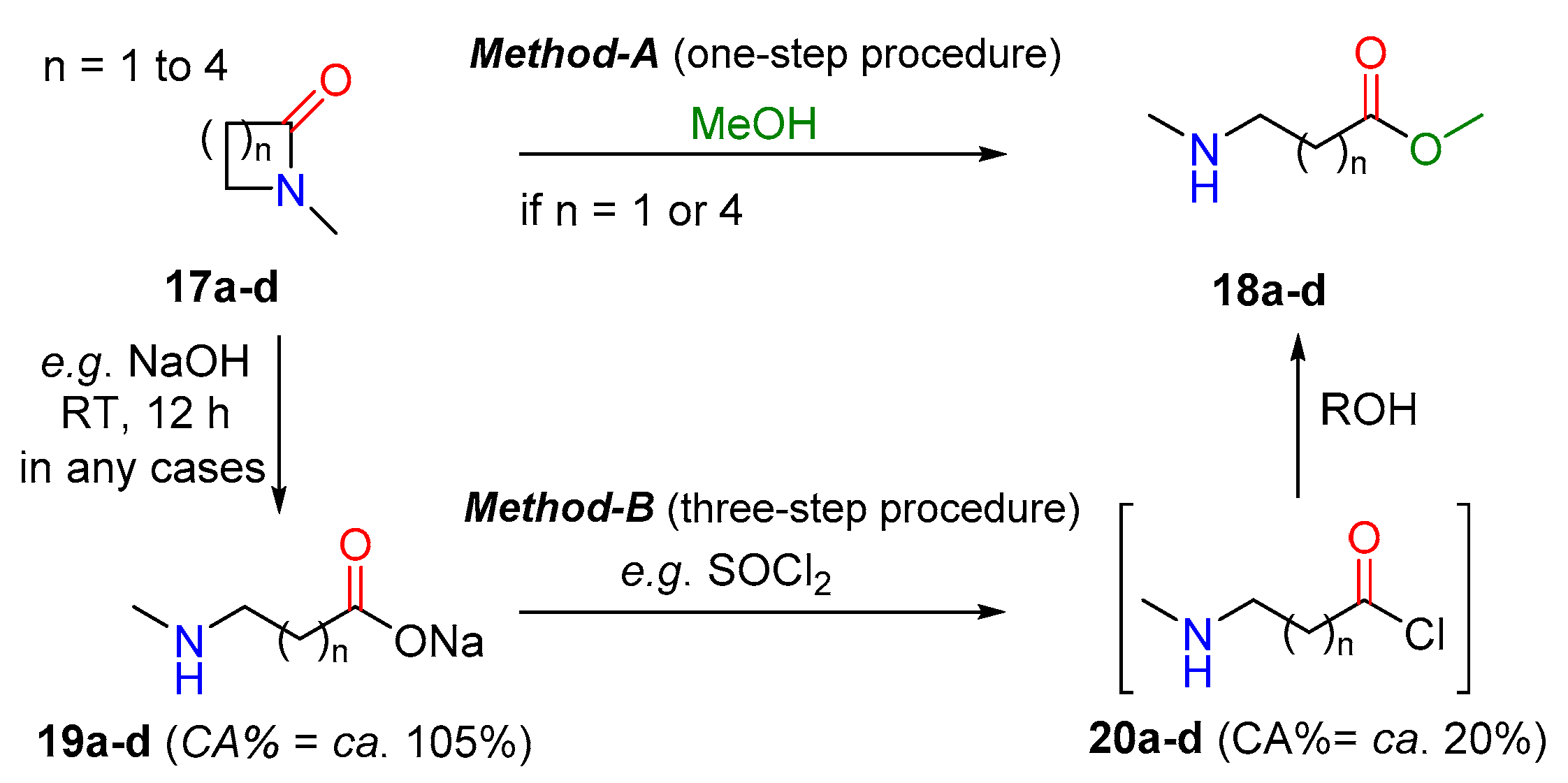
properties of amides
The characteristic reaction of covalent amides is hydrolysis (a chemical reaction with water), by which they are converted to acids and amines; this reaction ordinarily is slow unless it is catalyzed by a strong acid, an alkali, or an enzyme.
Amides also can be dehydrated to nitriles.
Are amides water soluble?
Amides having low molecular weights readily dissolve in water because hydrogen bonds form between the amide group and water.
Even low molecular weight tertiary amides dissolve in water because the carbonyl oxygen atom can form hydrogen bonds to the hydrogen atoms of water.
Which of the following are characteristics of amides?
The amides are polar and hence can form hydrogen bonds.
Due to this property, amides have high boiling and melting point.
Amides are classified into three groups based on the substituents/groups attached to the nitrogen atom of amide.
|
Chapter 6 Amines and Amides
Learn the important physical properties of the amines and amides. • Learn the major chemical reactions of amines and amides and learn how to predict the. |
|
How Do Amides Affect the Electronic Properties of Pyrene?
09-Oct-2018 Herein we demonstrate how the different modes of attaching an amide to a pyrene chromophore affect the electrochemical and optical properties ... |
|
Properties of amides in aqueous solution. I. Viscosity and density
An extended study of the physical properties of substituted amide-water systems partial molar volumes of the amides at infinite dilution have been ... |
|
The comparative roles of the proton-acceptor properties of amide
The O(amide) is a significantly stronger proton acceptor than the O(carboxyl). Thus The packing characteristics of 16 N-acylated amino acids I have. |
|
How Do Amides Affect the Electronic Properties of Pyrene?
09-Oct-2018 Herein we demonstrate how the different modes of attaching an amide to a pyrene chromophore affect the electrochemical and optical properties ... |
|
The structural properties of amides and imides as hydrogen storage
The structural properties of amides and imides as hydrogen storage materials. Takayuki Ichikawa* and Shigehito Isobe. Institute for Advanced Materials |
|
Optimized intermolecular potential functions for amides and
Optimized Intermolecular Potential Functions for Amides and Peptides. Structure and Properties of Liquid Amides. William L. Jorgensen* and Carol J. Swenson. |
|
Synthesis and spectroscopic properties of bis (trimethylsilyl) amides
Synthesis and Spectroscopic Properties of Bis (trimethyIsily 1)amides of the. Alkaline-Earth Metals Magnesium Calcium |
|
Substituted Amines and Amides Containing an Additional
Synthesis and Structural Properties of Adamantane-. Substituted Amines and Amides Containing an Additional. Adamantane Azaadamantane or Diamantane Moiety. |
|
Chapter 17: Amines and Amides
The functional group of an amide is an acyl group bonded to a trivalent nitrogen The basis for an amide is the -CONH2 group - known as an amide or acid amide because it is derived from a carboxylic acid |
|
Chapter 16 – Amines and Amides
20 jan 2002 · Amides with two hydrogen atoms bound to the nitrogen are called simple amides Those with one or two aliphatic or aromatic groups are called amides of amines The hydrogen bonding in these molecules is amongst the strongest observed |
|
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides
Physical Properties of Amides Amides • that are primary (−NH 2 ) or secondary (−NH−) form hydrogen bonds • that are primary have higher melting points |
|
PHYSICOCHEMICAL PROPERTIES OF ORGANIC MEDICINAL
Typically amides are less soluble than comparable amines and carboxylic acids since these compounds can both donate and accept hydrogen bonds, and can |
|
Esters, Amides, and Related Molecules - Organic Chemistry
Reactivity and Properties Acid halides are reactive compounds that serve as precursors to all of the other compounds with the structure R-C(=O)-Z (Figure 15 09 |
|
Synthesis and properties of amides of 2-substituted 3-aryl-and 3
amides of pyrido[1,2-a]pyrimidine-3-carboxylic acid, which react with hydrazine hydrate to In order to compare the properties of amides I-III, we obtained the |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be able to give the of physical and chemical properties Carboxylic |