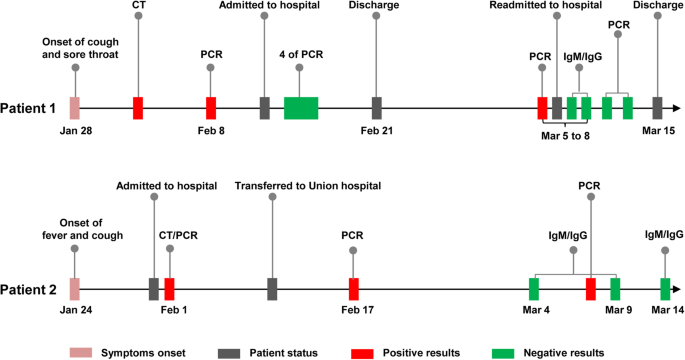
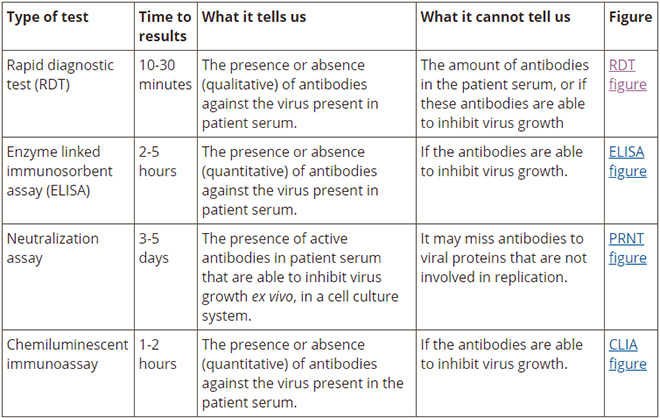
antibodies coronavirus test california
|
Monoclonal Antibody Treatment
available for COVID -19 health care providers and facilities – including pharmacists and pharmacies – regarding monoclonal antibody treatment and ordering monoclonal antibodies: • Monoclonal Antibody Treatment Information for Providers and Facilities • Direct Ordering of Monoclonal Antibodies |
|
DHCS COVID-19 Virus and Antibody Testing Guidance
25 août 2020 · This includes all medically necessary viral and serologic testing as well as serologic (antibody) tests ordered for infection control purposes ( |
|
What to Know for COVID-19 Testing
COVID-19 Testing – What You Need to Know When should you test? If you feel sick and have symptoms of COVID-19 • Test right away If your first test is negative test again 1-2 days later After exposure • Even if you don’t feel sick test after exposure and again 3-5 days after last exposure Before visiting those at high risk |
|
COVID-19 Antibody Testing Final 12-1-2020pdf
COVID-19 Viral and Serologic (Antibody) Testing DHCS covers both COVID-19 viral and serologic (antibody) tests at no cost to Medi-Cal beneficiaries This includes all medically necessary viral and serologic testing as well as serologic (antibody) tests ordered for infection control purposes (e g pre-operative |
|
COVID-19 Antibody Testing in a Pandemic
Antibody testing (serologic testing) is a way to test if an individual has developed antibodies for a certain virus (in this case SARS- CoV-2) during the later |
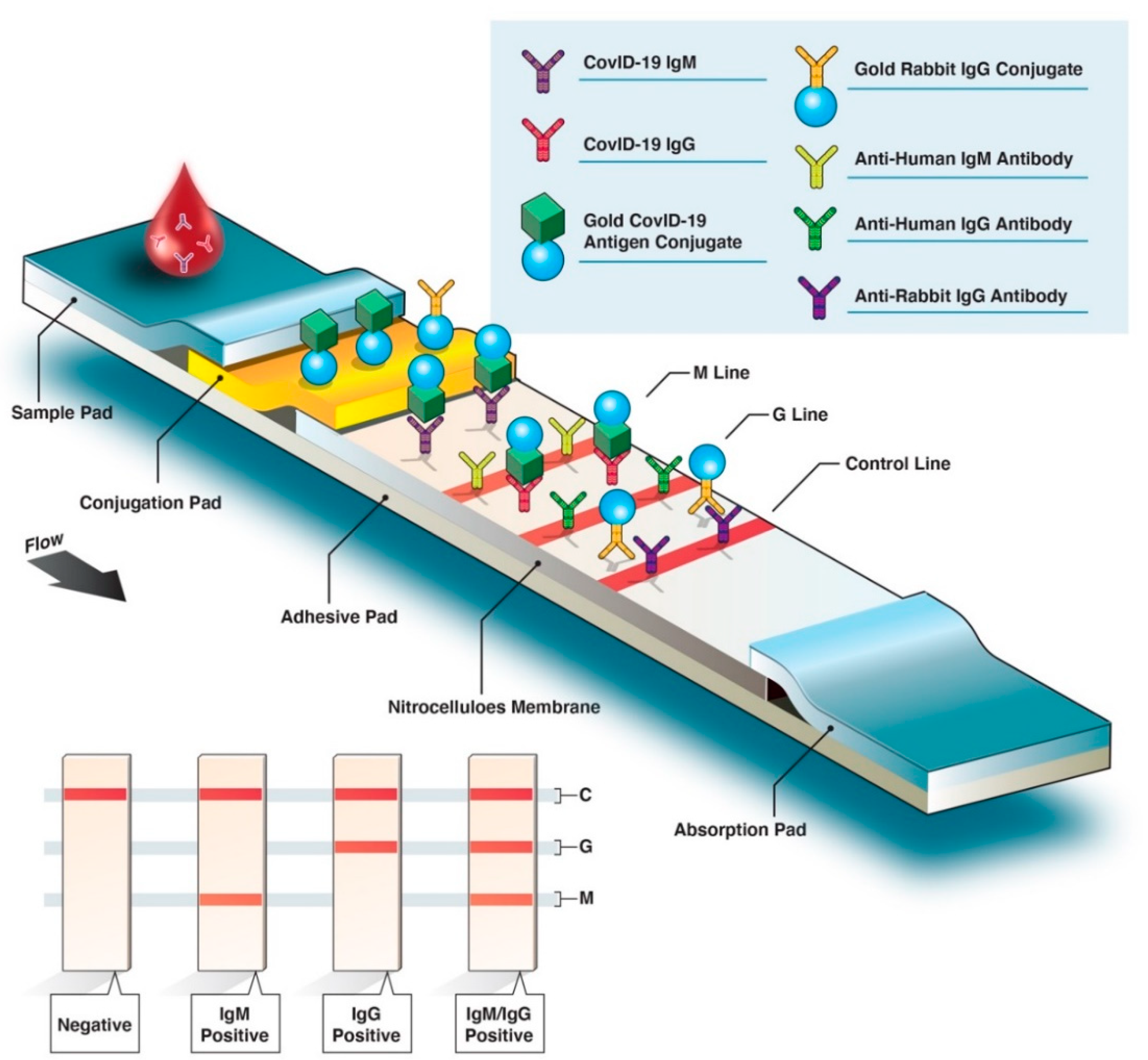
The COVID-19 IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma) is a lateral flow immunoassay intended for the qualitative detection and differentiation of IgM and IgG antibodies to SARS-CoV-2 in human venous whole blood (lithium heparin, dipotassium EDTA and sodium citrate), plasma (lithium heparin, dipotassium
What test checks antibody levels for Covid?
Antibody testing, also known as serology testing, is usually done after full recovery from COVID-19 .
Eligibility may vary, depending on the availability of tests.
A health care professional takes a blood sample, usually by a finger prick or by drawing blood from a vein in the arm.
Can you test positive if you have Covid antibodies?
False positive result
It's possible to get a “false positive” if you have antibodies but had a different kind of coronavirus, or the test may simply be inaccurate.
The risk here is having a false sense of security that you're protected from COVID-19.
Can you do an antibody test for Covid?
SARS-CoV-2 antibody tests can help identify people who may have been infected with the SARS-CoV-2 virus or have recovered from COVID-19.
Antibody tests should not be used to tell you if you have an active COVID-19 infection.
Scientists continue to learn more about COVID-19 and COVID-19 immunity.
Summary of Recent Changes
Updates as of December 16, 2022 •Added introduction to antibodies and COVID-19. cdc.gov
Key Points:
Antibody tests have public health value for monitoring and evaluating population levels of immunity, as well as clinical utility for patients. •Antibody tests that have received Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA) may be used for both public health and clinical purposes. Individual performance characteristics for each test can be found in the test’s instructions for use (IFU). •Antibody testing should not be used to determine whether someone is currently infected with SARS-CoV-2. Viral tests detect current infection. •Antibody testing is not currently recommended to assess for immunity to SARS-CoV-2 following COVID-19 vaccination or to assess the need for vaccination in an unvaccinated person. cdc.gov
Introduction to COVID-19 Antibodies
Even if a person does get sick, having antibodies can protect them from getting severely ill because their body has some experience in fighting that disease. How long this protection lasts can be different for each disease, each person, or influenced by other factors. Antibodies are just one part of a persons’ immune response. Antibodies to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19, can be detected in the blood of people who have recovered from COVID-19 or people who have been vaccinated against COVID-19. It is important to remember that some people with antibodies to SARS-CoV-2 may become infected after vaccination (vaccine breakthrough infection) or after recovering from a past infection (reinfection). For many diseases, including COVID-19, antibodies are expected to decrease or “wane” over time. As their antibodies wane, a person may become more vulnerable to SARS-CoV-2 infection. Even after a person’s antibodies wane, their immune system may have cells that remember the virus and that can act quickly to protect the person from severe illness if they become infected. cdc.gov
Recommendations for Use of Antibody Tests
Antibody tests can be used to: •Determine if a person has COVID-19 antibodies, which suggests past infection or vaccination.* •Aid in the diagnosis of multisystem inflammatory syndrome in children (MIS-C) and in adults (MIS-A). •Monitor and evaluate population levels of immunity. *Antibody tests are not recommended or authorized by the FDA to assess someone’s immunity after COVID-19 vaccination or determine if they need to be vaccinated. However, EUA indications do not preclude use of antibody tests in vaccinated individuals in certain situations. Antibody tests should not be used to: •Diagnose current infection.** •Determine if someone can return to work or school. •Group people together in settings such as schools, dormitories, and correctional facilities; or to exempt someone from screening testing. •Exempt a person who wears personal protective equipment (PPE) at work from following site-specific requirements **Acute infection from SARS-CoV-2 is determined best by diagnostic testing using a nucleic acid amplification test (NAAT) or antigen test. Choice of antibody test and testing strategy For all clinical and public health purposes, it is recommended to use one of the numerous antibody tests for SARS-CoV-2 that have been authorized by FDA. The list of qualitative and semi-quantitative SARS-CoV-2 antibody tests granted an EUA by the FDA can be found on FDA’s website. Antibody tests with very high sensitivity and specificity are preferred since they are more likely to exhibit high positive (probability that the person testing positive actually has antibodies) and negative predictive values (probability that the person testing negative actually does not have antibodies) when administered at least 3 weeks after the onset of illness. Additional considerations when selecting an antibody test include: •IgG levels appear to decrease more slowly over time than levels of other classes of antibody. Therefore, assays that measure total antibody or IgG could have higher sensitivity than IgM assays as more time passes since a person’s last infection. •IgM antibody can persist for weeks to months following infection, though its persistence appears to be shorter than IgG’s; therefore, detection of IgM could suggest relatively recent infection. •Detection of persistent antibodies varies by the test used. FDA has issued an EUA for surrogate neutralization tests, which are qualitative binding assays that detect antibodies that block the interaction between the virus and the cellular virus receptor (ACE-2) without using cells or infectious virus. Plaque reduction neutralization assays are considered the gold standard for detection of neutralizing antibodies, but require cells, infectious virus, and are difficult to standardize. Although the surrogate neutralization test exhibits correlation to a plaque reduction neutralization test, the clinical or public health applicability has not been established. The clinical applicability of semi-quantitative tests has not been established. cdc.gov
Antibody Testing in the United States
Antigenic targets The choice of antigenic targets might help address different aspects of immune response. Antibody detection against receptor-binding domain (RBD) is considered to have higher correlation with functional aspects like ability to neutralize virus (6). Differential reactivity of S and N specific antibodies might be used to help differentiate previous infection from vaccination in serologic studies, particularly for vaccines that produce antibodies only against S protein (1, 25, 40). Multiple forms of S protein—full-length (S1+S2) or partial (S1 domain or RBD)—are used as antigens for antibody tests. S protein is essential for virus entry into cells and is present on the viral surface. Within the S protein, the RBD is more conserved than S1 or full-length S. N protein is the most abundantly expressed immunodominant protein and is more conserved across coronaviruses than S. Types of antibody testing These tests use purified proteins of SARS-CoV-2, not viable virus, and can be performed in lower biosafety level laboratories (e.g., BSL-2). With specific reagents, individual antibody types, like IgG, IgM, and IgA, can be differentiated. Both SARS-CoV-2 IgM and IgG antibodies may be detected around the same time after infection. However, while IgM is most useful for determining recent infection, it usually becomes undetectable weeks to months following infection; in contrast, IgG is usually detectable for longer periods. IgA is important for mucosal immunity and, in addition to blood, can be detected in mucous secretions like saliva. Depending on their complexity, some binding antibody tests can be performed rapidly (in fewer than 30 minutes) in a field setting or in a few hours in a laboratory. Tests that detect binding antibodies These types of tests fall into two broad categories: •Point-of-care (POC) tests are diagnostic tests performed at or near the place where a specimen is collected and can provide results within minutes rather than hours. Antibody POC tests generally are lateral flow devices that detect IgG, IgM, or total antibody in fingerstick whole blood. •Laboratory tests use lateral flow, ELISA, or chemiluminescent immunoassay (CIA) methods for antibody detection in serum, plasma, whole blood, and dried blood spots, which, for some assays, might require trained laboratory scientists and specialized instruments. Based on the test, total antibody (Ig) can be detected, or IgG and IgM can be detected separately. While most tests detect antibodies against either S or N proteins, some tests can detect antibodies against both immunodominant proteins (multiplex assays). •Virus neutralization tests (VNT), such as the plaque-reduction neutralization test (PRNT) and microneutralization, use SARS-CoV-2 or recombinant SARS-CoV-2 expressing reporter proteins. These tests may take up to 5 days to complete. •Pseudovirus neutralization tests (pVNT) use recombinant pseudoviruses (like vesicular stomatitis virus [VSV] or lentiviruses) that incorporate the S protein of SARS-CoV-2. These reporter-based tests can be performed in BSL-2 laboratories depending on the virus strain used. •Surrogate neutralization tests have also been developed, and some have been authorized under an EUA by the FDA. These are binding antibody tests designed to qualitatively detect potentially neutralizing antibodies, often those that prevent interaction of the RBD with the ACE-2 receptor. The test mimics the interaction of the RBD with ACE-2 in an ELISA format (similar to the RBD on a virus particle binding to a cell surface ACE-2 receptor) and the ability of RBD specific antibodies to interfere with the interaction is detected using a decrease in signal based on the reporter-fused RBD. These tests can be conducted in BSL-2 laboratories because they do not require viable virus. Performance of antibody tests Independently evaluated test performance and the approval status of tests are listed on an FDA website. Most authorized tests are qualitative (providing a result that is positive, negative, or indeterminate) or semi-quantitative (providing a numerical result using a scale that is unique to that assay and not comparable to other assays); however, authorized quantitative assays (providing a measured and scaled assessment of antibody levels) are also available. FDA requires commercially marketed antibody tests for SARS-CoV-2 to receive Emergency Use Authorization (EUA) or approval. Multiple agencies—including FDA, the National Cancer Institute/National Institutes of Health (NCI/NIH), CDC, and the Biomedical Advanced Research and Development Authority (BARDA)—are collaborating with members of academia and the medical community to evaluate the performance of antibody tests independently using a well-characterized set of clinical specimens (serum and plasma) collected before and during the COVID-19 pandemic. The World Health Organization has developed international standards for SARS-CoV-2 antibody tests that can serve as the foundation for the calibration of tests that quantify antibodies. Both laboratory and point-of-care antibody tests have received EUA from the FDA. Antibody testing technologies include single-use lateral flow tests where the presence of antibody is demonstrated by a color change on a paper strip (similar to a pregnancy test) and laboratory-based immunoassays that allow for processing of many specimens at the same time. The EUA letter of authorization includes the settings in which each test is authorized, based on FDA’s determination of appropriate settings for use during the public health emergency. cdc.gov
Development of Antibodies and Immunity
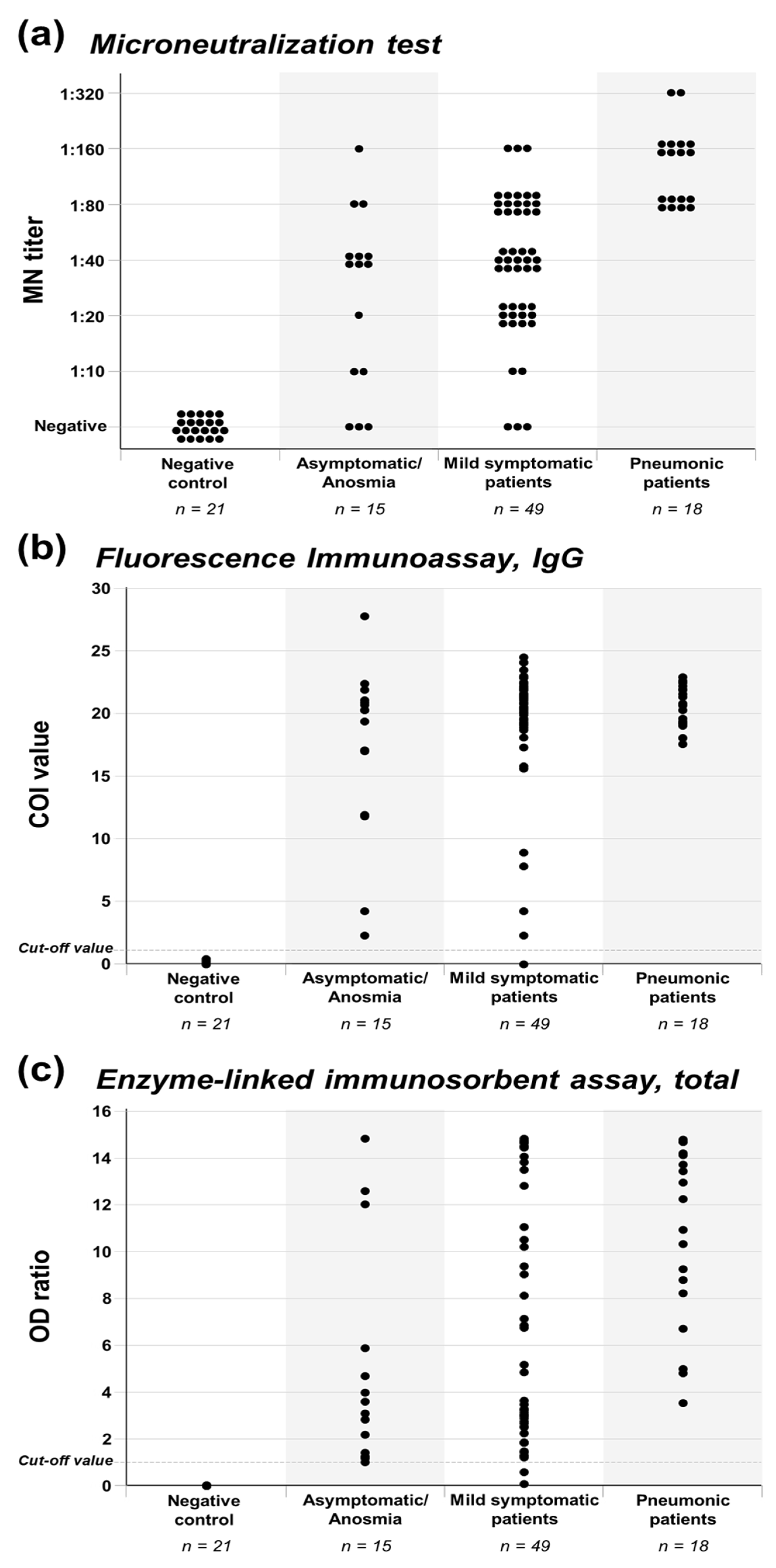
Infection Data indicate that nearly all immunocompetent persons develop an adaptive immune response following SARS-CoV-2 infection, triggering antiviral humoral and cellular immune responses via B and T cell-mediated immunity (4–6), respectively. In humans, the humoral response includes antibodies directed against S and N proteins. The S protein contains two subunits, S1 and S2. The S1 subunit contains the receptor-binding domain (RBD) that mediates binding of virus to susceptible cells. RBD is the main target for neutralizing antibodies. Antibodies—including IgM, IgG, and IgA—against S and its subunits can be detected in serum within 1-3 weeks after infection (7, 8). IgM and IgG antibodies can arise nearly simultaneously (7); however, IgM (and IgA) antibodies decay more rapidly than IgG (7, 9). The clinical significance of measuring serum IgA in SARS-CoV-2 infection is not known; however secretory IgA plays an important role in protecting mucosal surfaces against pathogens by neutralizing respiratory viruses, including SARS-CoV-2 (10). IgG antibodies, including IgG against the S and N proteins, persist for at least several months in most persons, but the precise duration of time that antibodies persist after infection is unknown (11). Loss of previously detectable SARS-CoV-2 antibodies (seroreversion) has been reported among persons with mild disease (12). Persons with more severe disease appear to develop a more robust antibody response with IgM, IgG, and IgA, all achieving higher titers and exhibiting longer persistence (12, 13). The observed persistence of antibodies can vary by assay (14), and some studies have found that approximately 5%–10% of people do not develop detectable IgG antibodies following infection (15, 16). Although neutralizing antibodies might not be detected among patients with mild or asymptomatic disease (17), the humoral immune response appears to remain intact, even with loss of specific antibodies over time, because of the persistence of memory B-cells (18). SARS-CoV-2 neutralizing antibodies that inhibit viral replication in vitro mainly target the RBD (5, 6). Efforts to better understand antibody kinetics, longevity of humoral immune responses, correlation of binding antibody levels to neutralizing antibodies, and serological surrogates of immune protection are dependent on wider availability of quantitative binding antibody assays that are standardized and traceable to an international standard (19). SARS-CoV-2 reinfection has been documented (20, 21); however, studies indicate that persons with SARS-CoV-2 antibodies are less likely to experience subsequent infection or clinical disease than persons without antibodies. Investigations of outbreaks among people on a fishing vessel and at a summer camp in the United States found that persons with pre-existing SARS-CoV-2 antibodies were correlated with protection from subsequent infection (22, 23). In sequential outbreaks among staff and residents of two British nursing homes, persons who tested antibody-positive following the first outbreak were approximately 96% less likely to become infected during the second outbreak four months later (24). In a British prospective cohort study of persons with and without SARS-CoV-2 antibodies, the adjusted incidence rate ratio for subsequent infection was 0.11 among persons followed for a median of 200 days after a positive antibody test, compared with those who tested negative for SARS-CoV-2 antibodies (2). Another British cohort study found an 84% reduction in SARS-CoV-2 infection incidence over a seven-month period among persons who had tested antibody positive for SARS-CoV-2 or had prior infection documented by reverse transcription polymerase chain reaction (RT-PCR) (1). A large study in the United States of commercial laboratory results linked to medical claims data and electronic medical records found a 90% reduction in infection among persons with antibodies compared with persons without antibodies (25), and another study of U.S. military recruits found that seropositive persons had an 82% reduction in incidence of SARS-CoV-2 infection over a 6-week period (26). Experiments on non-human primates support the above observations in humans. Experimentally infected rhesus macaques that developed humoral and cellular immune responses were protected against reinfection when re-challenged 35 days later (27). Another study found that transfer of purified IgG from rhesus macaques infected with SARS-CoV-2 was effective in protecting naïve rhesus macaques from infection, and the threshold titers for protection, based upon binding and neutralizing antibodies, were determined. Analyses of data from two vaccine trials found that higher titers of neutralizing and anti-S binding antibodies correlated with more effective protection from infection (28, 29). Taken together, these findings in humans and non-human primates suggest that SARS-CoV-2 infection and development of antibodies can result in some level of protection against SARS-CoV-2 reinfection. The extent and duration of protection have yet to be determined. While life-long immunity has not been observed with endemic seasonal coronaviruses (30), studies of persons infected with the SARS-CoV-1 and Middle East Respiratory Syndrome (MERS-CoV) coronaviruses demonstrated measurable antibody for 18–24 months following infection (31, 32), and neutralizing antibody was present for 34 months in a small study of MERS-CoV-infected patients (33). It is not known to what extent persons re-infected with SARS-CoV-2 might transmit SARS-CoV-2 to others or whether the clinical spectrum differs from that of primary infection. Vaccination cdc.gov
References
1.Hall VJ, Foulkes S, Charlett A, Atti A, Monk EJM, Simmons R, et al. SARS-CoV-2 infection rates of antibody-positive compared with antibody-negative health-care workers in England: a large, multicentre, prospective cohort study (SIREN). Lancet. 2021 Apr 17;397(10283):1459-69. 2.Lumley SF, O’Donnell D, Stoesser NE, Matthews PC, Howarth A, Hatch SB, et al. Antibody status and incidence of SARS-CoV-2 infection in health care workers. N Engl J Med. 2020 Dec 23;384:533-40. 3.Abdool Karim SS, de Oliveira T. New SARS-CoV-2 Variants – Clinical, Public Health, and Vaccine Implications. N Engl J Med. 2021 May 13;384(19):1866-8. 4.Grifoni A, Weiskopf D, Ramirez SI, Mateus J, Dan JM, Moderbacher CR, et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell. 2020 Jun 25;181(7):1489-501 e15. 5.Robbiani DF, Gaebler C, Muecksch F, Lorenzi JCC, Wang Z, Cho A, et al. Convergent antibody responses to SARS-CoV-2 in convalescent individuals. Nature. 2020 Aug;584(7821):437-42. 6.Suthar MS, Zimmerman MG, Kauffman RC, Mantus G, Linderman SL, Hudson WH, et al. Rapid generation of neutralizing antibody responses in COVID-19 patients. Cell Rep Med. 2020 Jun 23;1(3):100040. cdc.gov
|
DHCS COVID-19 Virus and Antibody Testing Guidance
9 déc. 2020 State of California—Health and Human Services Agency ... DHCS covers both COVID-19 viral and serologic (antibody) tests at no cost to Medi- ... |
|
COVID-19 Antibody Seroprevalence in Santa Clara County California
11 avr. 2020 Methods. On 4/3-4/4 2020 |
|
COVID-19 Antibody Seroprevalence in Santa Clara County California
11 avr. 2020 In this section we describe our sampling and recruitment approaches specimen collection methods |
|
COVID-19 IgG/IgM antibody testing in Los Angeles County California
Developing a test that indicates past infection and possible immunity against the virus is important. We administered 244 antibody tests to three groups of high |
| Medicare Monoclonal Antibody COVID-19 Infusion Program Instruction |
|
COVID -19 Testing Locations
Notes: Four types of COVID tests are being offered (NOTE: There is a charge but insurance is accepted if available):. ·. Rapid COVID-19 Antibody Test – $99. |
|
COVID-19 Antibody Seroprevalence in Santa Clara County California
30 avr. 2020 Our protocol was informed by a World Health Organization protocol for population-level COVID-19 antibody testing.14 We conducted our study with ... |
|
COVID-19 Antibody Seroprevalence in Santa Clara County California
30 avr. 2020 We report both the weighted and unweighted prevalence of antibodies to SARS-CoV-2. We also adjust for test performance characteristics by ... |
|
SARS-CoV-2 antibody seroprevalence among firefighters in Orange
25 août 2021 Orange County California in August 2020. ? Among 57 firefighter/paramedics who self- reported a previous positive COVID-19 test. |
| CalScope FAQs |
|
Covid-19 Antibody Testing Guide - CSUSM
Kaiser Permanente is one of the leading health care providers/hospital networks in California with divisions in several other states including Hawaii You can get |
|
COVID-19 IgG/IgM antibody testing in Los - eScholarshiporg
1 fév 2021 · COVID-19 IgG/IgM antibody testing in Los Angeles County, Powered by the California Digital Library Keywords antibodies coronavirus |
|
Seroprevalence of antibodies to SARS - BMJ Open
12 fév 2021 · exposures; and symptoms consistent with COVID-19 infection Results We standing how different antibody tests for SARS- CoV-2 perform, and Cedars- Sinai Medical Center, Los Angeles, California, USA 4Division of |
|
COVID-19 Antibody Seroprevalence in Santa Clara - medRxiv
17 avr 2020 · The first two cases of COVID-19 in Santa Clara County, California were for population-level COVID-19 antibody testing 13 We conducted our |
|
On Testing for COVID-19 in Riverside County - COVID-19 Testing
Part of the plan on safely opening the State of California as well as the County of Antibody tests may have a false negative rate up to around 30 for many of |
|
Seroprevalence of SARS-CoV-2 IgM and IgG - IRIS PAHO Home
Asymptomatic infections; seroepidemiologic studies; coronavirus infections; asymptomatic infections; who tested positive for SARS-CoV-2 antibodies were contacted COVID-19 Antibody Seroprevalence in Santa Clara County, California |
|
COVID-19 Seroprevalance Study, Riverside County
22 jui 2020 · I'm a California resident under a “Shelter at home” order—can I still have COVID-19 antibodies even though the test results say you do not |
|
COVID-19 Antibody Seroprevalence in Santa Clara County - Govuk
30 avr 2020 · The first two cases of COVID-19 in Santa Clara County, California were for population-level COVID-19 antibody testing 14 We conducted our |