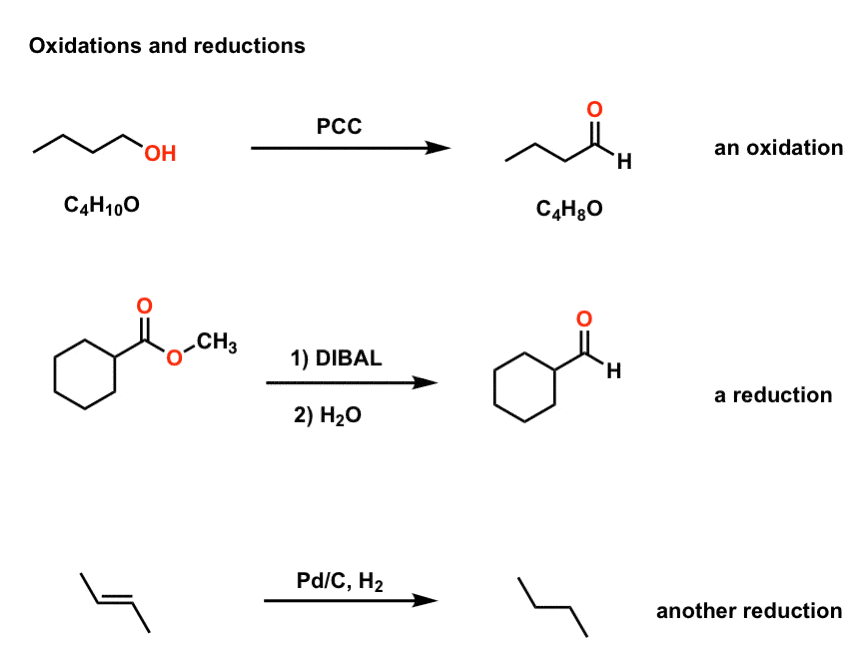
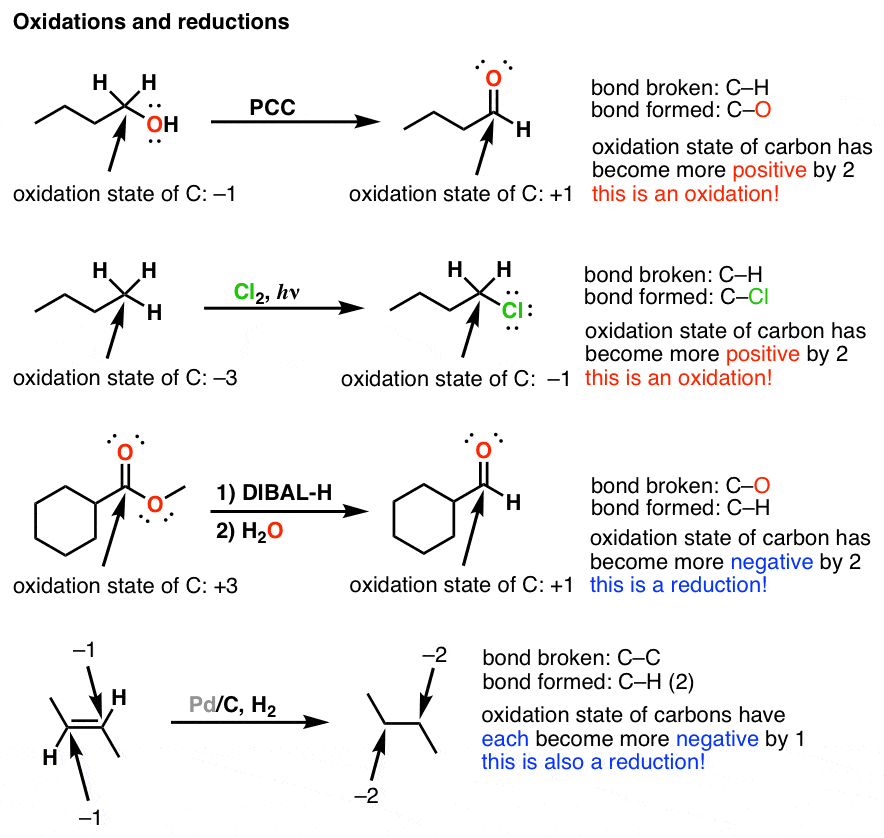
ap chem redox reactions
|
AP* Chemistry ELECTROCHEMISTRY
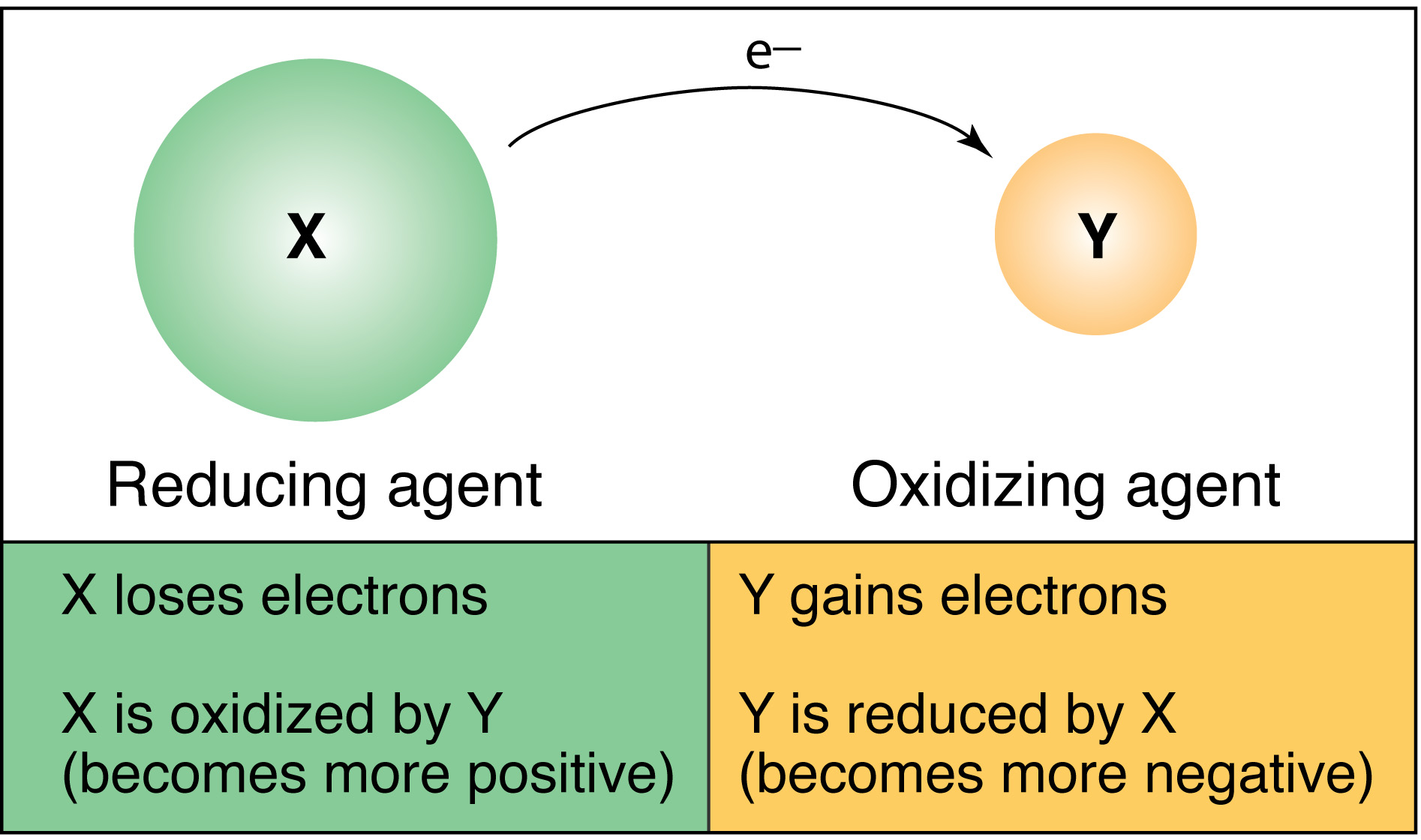
neutral salt Electron flow – ALWAYS through the wire from anode to cathode (alphabetical order) Voltmeter – measures the cell potential (emf) in volts Examine the diagram above Take note of the following mnemonic devices (easy ways to remember “stuff”): |
|
Oxidation-Reduction Extra Practice
Oxidation-Reduction Balancing Additional Practice Problems Acidic Solution 1 Ag + NO 3-→ Ag+ + NO 2 Zn + NO 3-→ Zn2+ + NH4 + 3 Cr 2O 7 2-+ C2H 4O → C 2H 4O 2 + Cr |
|
AP Chemistry 2016 Free-Response Questions
The student observes the formation of bubbles and that the flask gets cooler as the reaction proceeds (a) Identify the reaction represented above as an acid-base reaction precipitation reaction or redox reaction |
What is the scope of redox reactions?
The scope of redox reactions in this unit is understanding how to balance their chemical equations. We come back to redox in the very final unit of this course: applications of thermodynamics. . Let's go over both scenarios. First, assign oxidation numbers to all atoms that take part in the reaction. Mg and O₂ are both of 0. MgO is made up of an
How do you calculate a thermodynamically favorable reaction?
thermodynamically favorable by calculating the value of E for the reaction. Br and Cl can react to form the compound BrCl. (c) The boiling point of Br2 is 332 K, whereas the boiling point of BrCl is 278 K. Explain this difference in boiling point in terms of all the intermolecular forces present between molecules of each substance.
What happens when a redox reaction loses an electron?
loses an electron, it’s oxidized, and its oxidation number increases. When a gains an electron, it’s reduced, and its oxidation number decreases. . Okay, wait...that was a lot of information in three sentences. Here is a quick recap of what you should understand about redox reactions so far: to another. . . to the substance that is reduced.
What is the net ionic reaction on the AP Chemistry exam?
The net ionic reaction is: SnCl2(aq) → Sn(s) + 2Cl−(aq) you’ll need to figure out if the ions are reacting or the water is reacting. There are basically only two questions we can ask about electrolysis on the AP Chemistry exam so, this may be viewed as “good news”!
AP® CHEMISTRY EQUATIONS AND CONSTANTS
Throughout the exam the following symbols have the definitions specified unless otherwise noted. L, mL = liter(s), milliliter(s) g nm atm = gram(s) = nanometer(s) = atmosphere(s) mm Hg = millimeters of mercury J, kJ V mol = joule(s), kilojoule(s) = volt(s) = mole(s) CHEMISTRY apcentral.collegeboard.org
GO ON TO THE NEXT PAGE.
Use a pencil or pen with black or dark blue ink only. Do NOT write your name. Do NOT write outside the box. collegeboard.org. Continue your response to QUESTION 1 on this page. (c) In the box below, complete the Lewis electron-dot diagram for HCOOH. Show all bonding and nonbonding valence electrons. HNNH ( aq ) H O(l) apcentral.collegeboard.org
H2NNH2
with HCOOH. (ii) Is the resulting solution acidic, basic, or neutral? Justify your answer. GO ON TO THE NEXT PAGE. Use a pencil or pen with black or dark blue ink only. Do NOT write your name. Do NOT write outside the box. Continue your response to QUESTION 1 on this page. When a catalyst is added to a solution of HCOOH(aq ), the reaction represent

Redox titrations Chemical reactions AP Chemistry Khan Academy

Worked example: Balancing a simple redox equation Chemical reactions AP Chemistry Khan Academy

Worked example: Balancing a redox equation in acidic solution AP Chemistry Khan Academy
|
Unit 2 Redox Electrochem
Name as many types of batteries as you can think of. Page 3. AP Chemistry. Redox Reactions. • Batteries are redox reactions |
|
AP Chemistry 2018 Free-Response Questions
AP® CHEMISTRY EQUATIONS AND CONSTANTS. Throughout oxidation-reduction reaction represented by the balanced equation above. ... A student investigates the ... |
|
Unit 2 Redox and Electrochemistry Notes
Assign the most electronegative element a negative oxidation number. Page 3. AP Chemistry. Unit 2 Student Notes. Examples-. 1. H2O |
|
AP Chemistry Sample Student Responses and Scoring Commentary
oxidation-reduction reaction represented by the balanced equation above. (a) Determine the oxidation number of CI in NaOCI. +1. 1 point is earned for the |
|
Exercise 18.1 - Balancing Redox Equations
AP Chemistry. Exercise 18.1. Balancing Redox Equations. DIRECTIONS: Balance the following redox equations. 3+. 11. Mn²++ BiO3 → MnO4¯ + Bi³* (in acidic |
|
AP Chemistry 2021 Free-Response Questions
(e) Is the reaction a redox reaction? Justify your answer. (f) The reaction occurs inarigid 4.3 L vessel at 25°C and the total pressure is monitored |
|
AP Chemistry Scoring Guidelines
(c) Using the balanced equation for the oxidation-reduction reaction and the information in the table above determine which reactant is the limiting reactant. |
|
AP Chemistry Chief Reader Report from the 2018 Administration
In the second grouping parts (d) through (f) focused on the interpretation of calorimetry data for the redox reaction by determining the standard enthalpy |
|
AP Chemistry Course at a Glance Effective Fall 2019
7–9% AP Exam The Course at a Glance provides a useful visual organization of the AP Chemistry curricular ... (Redox) Reactions. 5. SAP. 3.1 Intermolecular ... |
|
AP Chemistry Quiz: Redox Chemistry
AP Chemistry Quiz: Redox Chemistry. Name___________________________________. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or |
|
Unit 2 Redox and Electrochemistry Notes
Balancing Redox Example #1 (longer balancing using all steps). Balance the following reaction. MnO4. – + Fe2+ ? Mn2+ + Fe3+. Page 5. AP Chemistry. |
|
A.P. Chemistry Practice Test - Ch. 17: Electochemistry MULTIPLE
20-Feb-2018 ) Which of the following reactions is a redox reaction? (a) K2CrO4 + BaCl2 ¬ BaCrO4 + 2KCl. (b) Pb22+ + 2Br- ¬ PbBr. (c) Cu + ... |
|
Unit 2 Redox Electrochem
AP Chemistry. Electrochemistry. • is the study of the interchange of chemical and electrical energy. • The key is an Oxidation-Reduction or redox reaction. |
|
Download Free Ap Lab Redox Titration
experiment Redox titration |
|
Download File PDF Ap Lab Redox Titration
reactions and electrochemistry |
|
AP Chemistry 2021 Free-Response Questions
AP Central is the official online home for the AP Program: AP® CHEMISTRY EQUATIONS AND CONSTANTS ... (e) Is the reaction a redox reaction? |
|
Read Free Ap Lab Redox Titration
Final AP Chem Project: Redox/Titration Lab by Natalie Thornton Redox Reactions: Crash Course Chemistry #10 Redox Titration AP Chemistry Lab #5 Analysis ... |
|
Exercise 18.1 - Balancing Redox Equations
AP Chemistry. Exercise 18.1. Balancing Redox Equations. 1. 2. The oxidation number of alkali metals in compounds is +1 and that of alkaline earths in |
|
Bookmark File PDF Ap Chemistry Electrochemistry Answers
02-Sept-2022 AP Chem: Electrochemistry-1: Galvanic. Cells and Reduction Potentials (3/4). Introduction to Oxidation Reduction. (Redox) Reactions AP ... |
|
Download Free Ap Chemistry Electrochemistry Answers
5 days ago Redox reaction from dissolving zinc in copper sulfate. Introduction to galvanic/voltaic cells. Electrodes and voltage of Galvanic cell. |
|
RedOx Reactions
Oxidation-reduction reactions are also known as redox reactions • Def: Redox reactions describe all chemical reactions in which there is a net change in |
|
REDOX REACTIONS - NCERT
balance chemical equations using (i) oxidation number (ii) half reaction method; • learn the concept of redox reactions in terms of electrode processes UNIT 8 |
|
Chapter 19: Redox Reactions
Oxidation numbers are tools that scientists use in written chemical equations to help them keep track of the movement of electrons in a redox reaction Like some of |
|
MECHANISMS OF REDOX REACTIONS OF SIMPLE CHEMISTRY
MECHANISMS OF REDOX REACTIONS OF SIMPLE CHEMISTRY H Taube The general subject includes as a proper part problems of specific chem- |
|
Chemistry 30 Redox Reactions Unit - Prairie Spirit Blogs
Chemistry 30 Redox Reactions Unit: Intro to Redox Reactions Oxidation and Reduction • Oxidation-reduction reactions are commonly known as REDOX |
|
Oxidation-Reduction Experiments
Chem 112 OXIDATION-REDUCTION EXPERIMENT INTRODUCTION An oxidation-reduction (redox) reaction involves the movement of electrons from one |
|
1 Lecture 11 Redox Chemistry Many elements in the periodic table
Redox reactions are characterized by the transfer of electrons between chemical species: For example, see the following full reaction of cupric ion with metallic |
|
Redox Reactions
With this, reduction and oxidation have become terminologically paired – to two types of chemical reactions, of which one represents the reverse of the other |
|
RedOx Chemistry
Check if the two are a RedOx pair If not, reverse the more negative half reaction III For each half reaction, balance elements other than oxygen and hydrogen |