ap chemistry electrochemistry free response
|
A P Chemistry 2014 Free-Response Questions
Directions: Questions 1–3 are long free-response questions that require about 20 minutes each to answer and are worth 10 points each Questions 4–7 are short free-response questions that require about 7 minutes each to answer and are worth 4 points each Write your response in the space provided following each question |
|
AP REVIEW ELECTROCHEMISTRY
G = Gibbs free energy [Reaction is thermodynamically favorable if ΔG is negative] n = number of moles of electrons F = Faraday constant 9 6485 × 10 4 J/V |
|
AP REVIEW QUESTIONS – Electrochemistry
(e) Write balanced net-ionic equation for the overall chemical reaction occurring in the cell (f) In the cell the concentration of Sn2+ is changed from 1 0 M |
|
The Advanced Placement Examination in Chemistry Electrochemistry
The Advanced Placement Examination in Chemistry Part II - Free Response Questions Answers 1970 to 2006 Electrochemistry Teachers may reproduce this |
|
AP* Electrochemistry Free Response Questions
(a) Identify M and M2+ in the diagram and specify the initial concentration for M2+ in solution Electrons flow from the anode to the cathode in a voltaic |
|
AP* Electrochemistry Free Response Questions
AP* Electrochemistry Free Response Questions page 1 Galvanic (Voltaic) Cells 1988 Average score = 5 02 two points Sn ---> Sn2+ + 2e ̄ Ag+ + e ̄ ---> Ag two points 2 Ag+ + Sn ---> 2 Ag + Sn2+ E° = [0 80 - (- 0 14)] V = 0 94 V two points = (0 0591 ÷ n) log K (or - nFE = - RT ln K) log K = (0 94 x 2) ÷ 0 0591 = 31 8 = 6 x 1031 two points |
|
2004 AP Chemistry Free Response Questions
The required essay was designed to assess students' knowledge of electrochemistry and to integrate a variety of chemical principles Students were expected |
|
2006 APR CHEMISTRY FREE-RESPONSE QUESTIONS (Form B)
2006 APR CHEMISTRY FREE-RESPONSE QUESTIONS (Form B) Answer EITHER Question 2 OR Question 3 below Only one of these two questions will be graded If you |
|
AP® CHEMISTRY 2012 SCORING GUIDELINES
This electrochemistry question was divided into three parts The first component asked students to consider observations of electrochemical reactions to |
|
AP Chemistry 2018 Free-Response Questions
In your drawing represent oxygen atoms and nitrogen atoms as indicated below 2018 AP® CHEMISTRY FREE-RESPONSE QUESTIONS The student reads in a reference text |
|
AP Chemistry 2022 Free-Response Questions
QUESTION 1 on this page CHEMISTRY SECTION II Time—1 hour and 45 minutes 7 Questions YOU MAY USE YOUR CALCULATOR FOR THIS SECTION Directions: Questions 1–3 are long free-response questions that require about 23 minutes each to answer and are worth 10 points each |
|
AP Chemistry 2023 Free-Response Questions
1 Answer the following questions related to manganese compounds (a) Manganese has several common oxidation states Write the complete electron configuration for an Mn atom in the ground state When manganese forms cations electrons are lost from which subshell first? Identify both the number and letter associated with the subshell |
How many free responses are there in AP Chemistry?
The AP® Chemistry exam includes 60 multiple-choice questions and 7 free-response questions, including 3 long-answer questions and 4 short-answer questions.
Each section represents 50% of your score.What percent is a 5 on AP Chem?
It is also curved similar to other AP exams.
In the case of AP chem, you will need to get roughly 75% of the questions correct to score a 5.What is the hardest unit in AP Chemistry?
1.
Thermodynamics (Unit 6) - This is considered the hardest unit for many students because it involves complex topics like entropy, enthalpy, and Gibbs free energy.
Calculations can be challenging, and understanding the conceptual aspects requires a good grasp of energy and spontaneity.

AP Chemistry Unit 9 Review: Electrochemistry

2021 Live Review 7 AP Chemistry Everything You Need to Know about Electrochemistry

2021 Live Review 8 AP Chemistry Free-Response Medley & Exam Strategies
|
AP* Electrochemistry Free Response Questions
Indicate which half-reaction occurs at the anode. (b) Write the balanced chemical equation for the overall spontaneous cell reaction that occurs when the. |
|
AP Chemistry 2018 Free-Response Questions
2018 AP® CHEMISTRY FREE-RESPONSE QUESTIONS. D. 1. A student performs an experiment to determine the value of the enthalpy change ?Hrxn |
|
AP* Electrochemistry Free Response Questions
1 point for the correct concentration of M2+ (Zn2+). Page 7. Page 8. AP® CHEMISTRY. 2002 SCORING GUIDELINES (Form B). Copyright © 2002 by College Entrance |
|
AP REVIEW QUESTIONS – Electrochemistry - Answers
An electrochemical cell is constructed with an open switch (e) Write balanced net-ionic equation for the overall chemical reaction occurring in the ... |
|
2004 AP Chemistry Free Response Questions
The required essay was designed to assess students' knowledge of electrochemistry and to integrate a variety of chemical principles. Students were expected to |
|
AP® Chemistry 2013 Free-Response Questions
ADVANCED PLACEMENT CHEMISTRY EQUATIONS AND CONSTANTS OXIDATION-REDUCTION; ELECTROCHEMISTRY ... 2013 AP® CHEMISTRY FREE-RESPONSE QUESTIONS. |
|
Read Online Ap Chemistry Electrochemistry Answers (PDF
Yeah reviewing a ebook Ap Chemistry Electrochemistry Answers could ensue your near friends AP Chemistry 2021 Free-Response Questions - College Board. |
|
AP Chemistry 2016 Free-Response Questions
AP. ®. CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following symbols have the definitions specified unless otherwise noted. |
|
A P Chemistry 2014 Free-Response Questions
2014 AP® CHEMISTRY FREE-RESPONSE QUESTIONS. © 2014 The College Board. Visit the College Board on the Web: www.collegeboard.org. GO ON TO THE NEXT PAGE. -5-. |
|
AP Chemistry 2015 Free-Response Questions
AP. ®. CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following symbols have the definitions specified unless otherwise noted. |
|
AP* Electrochemistry Free Response Questions - Waukee
Indicate which half-reaction occurs at the anode (b) Write the balanced chemical equation for the overall spontaneous cell reaction that occurs when the circuit is |
|
AP* Electrochemistry Free Response Questions - Coach Cokers
1 point for the correct concentration of M2+ (Zn2+) Page 7 Page 8 AP® CHEMISTRY 2002 SCORING GUIDELINES (Form B) Copyright © 2002 by College |
|
AP CHEMISTRY NOTES 12-1 ELECTROCHEMISTRY - Azle ISD
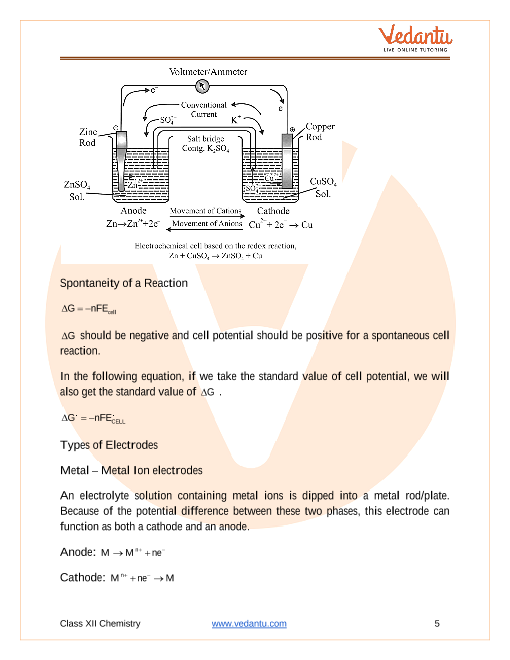
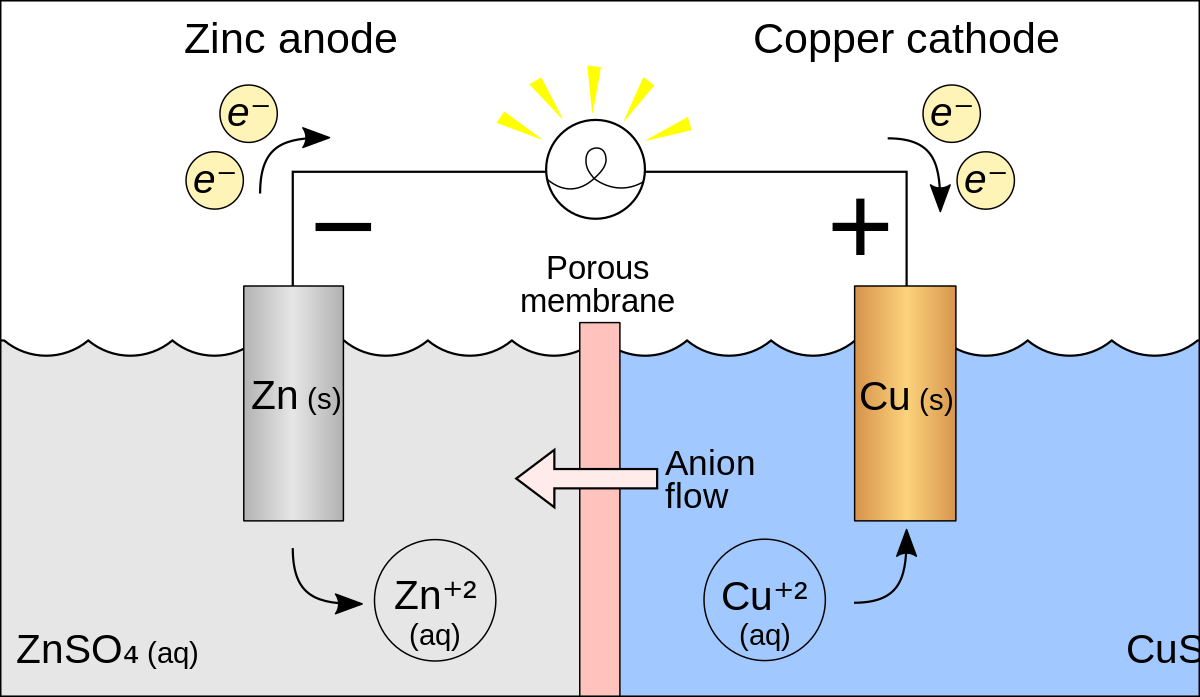
*These cells convert chemical energy into electrical energy (ie batteries) In an electrochemical cell, oxidation and reduction occur simultaneously The relationship between free energy and the spontaneity of an electrochemical cell can |
|
AP* Chemistry ELECTROCHEMISTRY
Draw a diagram of the galvanic cell for the reaction and label completely E°cell= 0 03 V CELL POTENTIAL, ELECTRICAL WORK FREE ENERGY It is time to |
|
Electrochemistry frq
Part II - Free Response Questions Answers Advanced Placement Examination in Chemistry (b) One half cell of an electrochemical cell is made by |
|
AP Chemistry 2019 Free-Response Questions - AP Students
2019 AP® CHEMISTRY FREE-RESPONSE QUESTIONS GO ON TO THE NEXT PAGE -5- CHEMISTRY Section II Time—1 hour and 45 minutes 7 Questions |
|
AP REVIEW QUESTIONS – Electrochemistry - Answers
An electrochemical cell is constructed with an open switch, as shown in the diagram above (e) Write balanced net-ionic equation for the overall chemical reaction (ii) On the basis of your response in (f) (i), will the cell potential be greater |
|
Electrochemistry - Higher Education Pearson
cal work done by an electrochemical cell and the free energy The Nernst equation chemical concentrations of oxidizable or reducible molecules, such as glucose, into electrical potentials, and thus In response to the membrane potential |
|
AP* Chemistry ELECTROCHEMISTRY - Lundquist Labs
Electrochemistry – the study of the interchange of chemical and electrical energy The following free responses are taken DIRECTLY from released AP exams, |




















![PDF) [2012] Electrochemical treatment of Methyl Orange dye PDF) [2012] Electrochemical treatment of Methyl Orange dye](https://www.mdpi.com/molecules/molecules-24-02122/article_deploy/html/images/molecules-24-02122-g014.png)