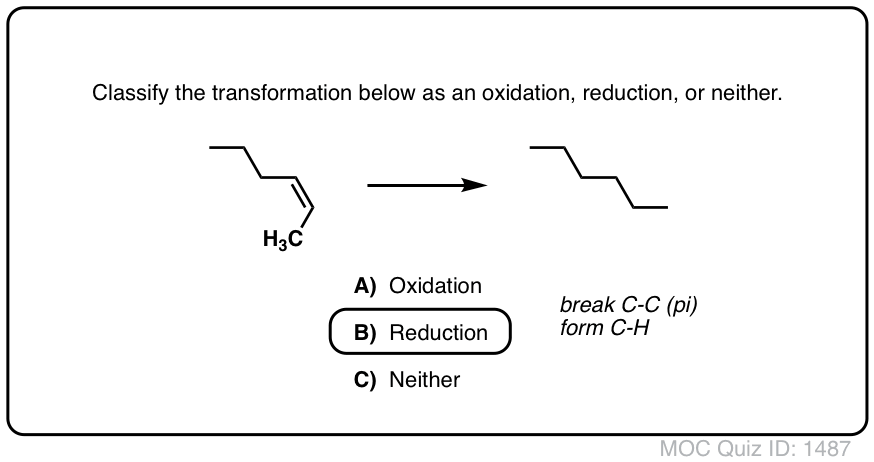
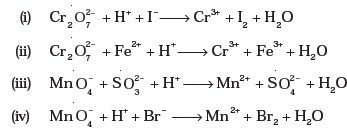ap chemistry redox multiple choice
|
CHAPTER 5 QUESTIONS Multiple-Choice Questions
One method to determine the exact concentration of H2O 2 in a bottle of hydrogen peroxide is to titrate a sample with a solution of acidified potassium permanganate This causes the following redox reactions to occur: Reduction: 8H+ + MnO4 – + 5e– → Mn2+ + 4H2O(l) Oxidation: H2O2(aq) → 2H+ + O2(g) + 2e− |
|
Worksheet &# 5 Balancing Redox Reactions in Acid and Basic
Balance each redox reaction in basic solution using the half reaction method 13 3O 2 + 8OH-+ 2Cr3+ -----> H 2O + 3H 2O 2 + Cr 2O 7 2-14 H 2O + Te + 4NO 3 - -----> TeO 3 2-+ 2OH-+ 2N 2O 4 15 7IO 3-+ 4OH- + 4Re -----> 4ReO 4-+ 7IO-+ 2H 2O 16 8OH- + 5Pb2+ + 2IO 3 - -----> 5PbO 2 + I 2 + 4H 2O 17 7H |
|
AP Chem: Chapter 4 Practice Multiple Choice Questions
Multiple Choice Identify the choice that best completes the statement or answers the question ____ What mass of silver nitrate AgNO3 is required to prepare 800 g of 3 50 solution of AgNO3? 24 6 g 26 7 g 27 0 g 25 5 g 28 0 g ____ 2 What mass of water is contained in 160 grams of 22 0 KCl solution? 125 g 86 8 g 35 2 g 130 g 112 g ____ 3 |
|
AP Chemistry Quiz: Redox Chemistry Name
quiz_redox_ap_ch_04 tst AP Chemistry Quiz: Redox Chemistry Name___________________________________ MULTIPLE CHOICE Choose the one alternative that best completes the statement or answers the question 1) __________ is reduced in the following reaction: Cr2O72- + 6S2O32- + 14H+ ¬ 2Cr3+ + 3S4O62- + 7H2O A) Cr2O72- |
How do you balance a redox reaction?
CuF2 + NH3 → Cu3N + NH4F + N2 Ca3(PO4)2 + SiO2 + C → P4 + CaSiO3 + CO Balance each redox equation. Assume all are spontaneous. Use the half reaction method. 2O2- Balance each half reaction in basic solution. 4. Cr2O7 2- 2K+ 5. NO 2e- Balance each redox reaction in acid solution using the half reaction method. 9.
What is a proposed mechanism for a reaction?
A proposed mechanism for the reaction consists of the three elementary steps shown below. Write the chemical formula of a species that behaves as a catalyst in the mechanism. is added to the reaction mixture, will the rate of the overall reaction increase, decrease, or remain the same? Justify your answer.
How do you convert oxidation to reduction chemistry?
Convert between K, G, and E using equations given on the AP Exam. Electrochemistry is all oxidation-reduction chemistry. Leo Ger OIL RIG Oxidation: loss of e; ✪ ox # increases Reduction: gain of e; ✪ ox # decreases example: Fe2+ + 2e ✪ Fe(s) (reduction) reducing agent gets oxidized. half-reaction method.
|
A.P. Chemistry Practice Test - Ch. 17: Electochemistry MULTIPLE
20-Feb-2018 4) Which of the following reactions is a redox reaction? ... Advanced Placement Chemistry: 1996 Free Response Questions. |
|
AP Chemistry Quiz: Redox Chemistry
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) ______ is reduced in the following reaction: Cr2O72- |
|
Redox and Electrochemistry Multiple Choice Review CONCEPT
The iron will be oxidized. 10. Which of the following is true for an electrolytic cell? a. It changes electrical energy into chemical energy. |
|
Untitled
Electrochemistry < 241. Use these questions to review the content of this chapter and practice for the AP Chemistry exam. First are 14 multiple-choice |
|
Read Book Ap Lab Redox Titration
20-Aug-2022 Provides techniques for achieving high scores on the AP chemistry exam and includes two ful- l-length practice tests a subject review for ... |
|
AP Chemistry Practice Exam from the 2018 Administration
NOTE: This is a modified version of the 2018 AP Chemistry Exam. This exam may not be posted (i) A student claims that the reaction is a redox reaction. |
|
Bookmark File PDF Ap Chemistry Electrochemistry Answers
02-Sept-2022 AP Chemistry: Electrochemistry Multiple. Choice Answers 14. Questions 14-17 The spontaneous reaction that occurs when. |
|
Read PDF Ap Lab Redox Titration
6 days ago Get ready to ace your AP Chemistry Exam with this easy-to-fol- low multi-platform study guide 5 Steps to a 5: AP Chemistry Elite. |
|
Multiple Choice Section: Please DO NOT WRITE or MAKE ANY
THE “OFFICIAL” CHEMISTRY 12 REDOX &ELECTROCHEMISTRY. STUDY GUIDE. SAHOTA. ? 03 Electrochemistry Study Guide - Multiple Choice - Page 1 of 22. |
|
2021 AP Exam Administration Student Samples: AP Chemistry Free
AP®. Chemistry. Sample Student Responses and Scoring Commentary must determine with evidence |
|
Redox and Electrochemistry Multiple Choice Review CONCEPT
c No reaction occurs d The iron will be oxidized 10 Which of the following is true for an electrolytic cell? a It changes electrical energy into chemical energy b It |
|
REDOX REACTIONS - NCERT
Which of the following is not an example of redox reaction? (i) CuO + H2 I Multiple Choice Questions (Type-I) Unit 8 Exemplar Problems, Chemistry |
|
AP Chemistry Practice Test - Ch 17: Electochemistry MULTIPLE
20 fév 2018 · Choose the one alternative that best completes the statement or answers the question 4) Which of the following reactions is a redox reaction? |
|
Multiple Choice Section - Ms Bunneys Classes
THE “OFFICIAL” CHEMISTRY 12 REDOX &ELECTROCHEMISTRY STUDY GUIDE SAHOTA Е 03 Electrochemistry Study Guide - Multiple Choice - Page 1 of |
|
Chem12 Oxidation/Reduction : Test - 80
Multiple choice questions 5) Which of the following represents a redox reaction ? In the redox equation above, the chemical species which is oxidized is : |
|
AP Chemistry Quiz: Redox Chemistry
MULTIPLE CHOICE Choose the one alternative that best completes the statement or answers the question 1) ______ is reduced in the following reaction : |
|
Unit 5E Practice Questions V Determining Oxidation Number
Multiple Choice – Circle The Best Answer 1 Oxidation-reduction (redox) reactions involve the loss and gain of: b electrochemistry d organic chemistry 18 |
|
MULTIPLE CHOICE QUESTIONS - PianetaChimica
NATIONAL HIGH SCHOOL CHEMISTRY EXAMINATION 2001 PART A - MULTIPLE CHOICE QUESTIONS (60 minutes) 1 A solution of ethanol in water Which one of the following reactions is NOT redox? A Br2 + H2O ⇔ HBr + HBrO |
|
AP Chem - Redox study guide answerspdf - Peoria Public Schools
First are 14 multiple-choice questions similar to what you will encounter in Section I of the AP Chemistry exam Following those is a multipart free-response |






![03 Electrochemistry Study Guide - Multiple Choice - [PDF Document] 03 Electrochemistry Study Guide - Multiple Choice - [PDF Document]](https://c2.staticflickr.com/4/3956/33563748222_0461038a06_o.png)