ap chemistry redox reactions
|
Balancing Redox Reactions Worksheet
Balancing Redox Reactions Worksheet 1 Balance each redox reaction in acid solution Mn 2+ + BiO3 - MnO4 + Bi 3+ - Æ MnO4 - + S2O3 2- Æ S4O6 2- + Mn 2+ ClO3 - + Cl - Æ Cl2 + ClO2 P + Cu 2+ Æ Cu + H2PO4 - - - PH3 + I2 Æ H3PO2 + I NO2 Æ NO3 - + NO Basic Solutions MnO4 - + C2O4 2- Æ MnO2 + CO2 |
|
Oxidation-Reduction Extra Practice
Answer: 10H+ + 4Zn + NO3 - → 4Zn2+ + NH4 + + 3H2O 2- 3 Cr2O7 + C2H4O C2H4O2 + Cr3+ Answer: 8H+ + 2- Cr2O7 + 3C2H4O 3C2H4O2 + 2Cr3+ + 4H2O 4 H3PO2 + Cr2O7 2- → H3PO4 + Cr3+ |
How do you balance redox reactions?
Balancing Redox Reactions Worksheet 1 Balance each redox reaction in acid solution. + Cu 2+ Balance each redox reaction in acid solution. Cr2O7 2- I2 Balance each redox reaction in basic solution. TeO3 2- H2O 4H+ + 4ReO4 - + 8H+ + 5PbO2 + 12H2O + 8As Æ 2Cr3+
What are examples of redox reactions?
Redox reactions are all around us: the burning of fuels, the corrosion of metals, and even the processes of photosynthesis and cellular respiration involve oxidation and reduction. Some examples of common redox reactions are shown below. CH A 4 ( g) + 2 O A 2 ( g) → CO A 2 ( g) + 2 H A 2 O ( g) ( combustion of methane)
What are oxidation reduction reactions?
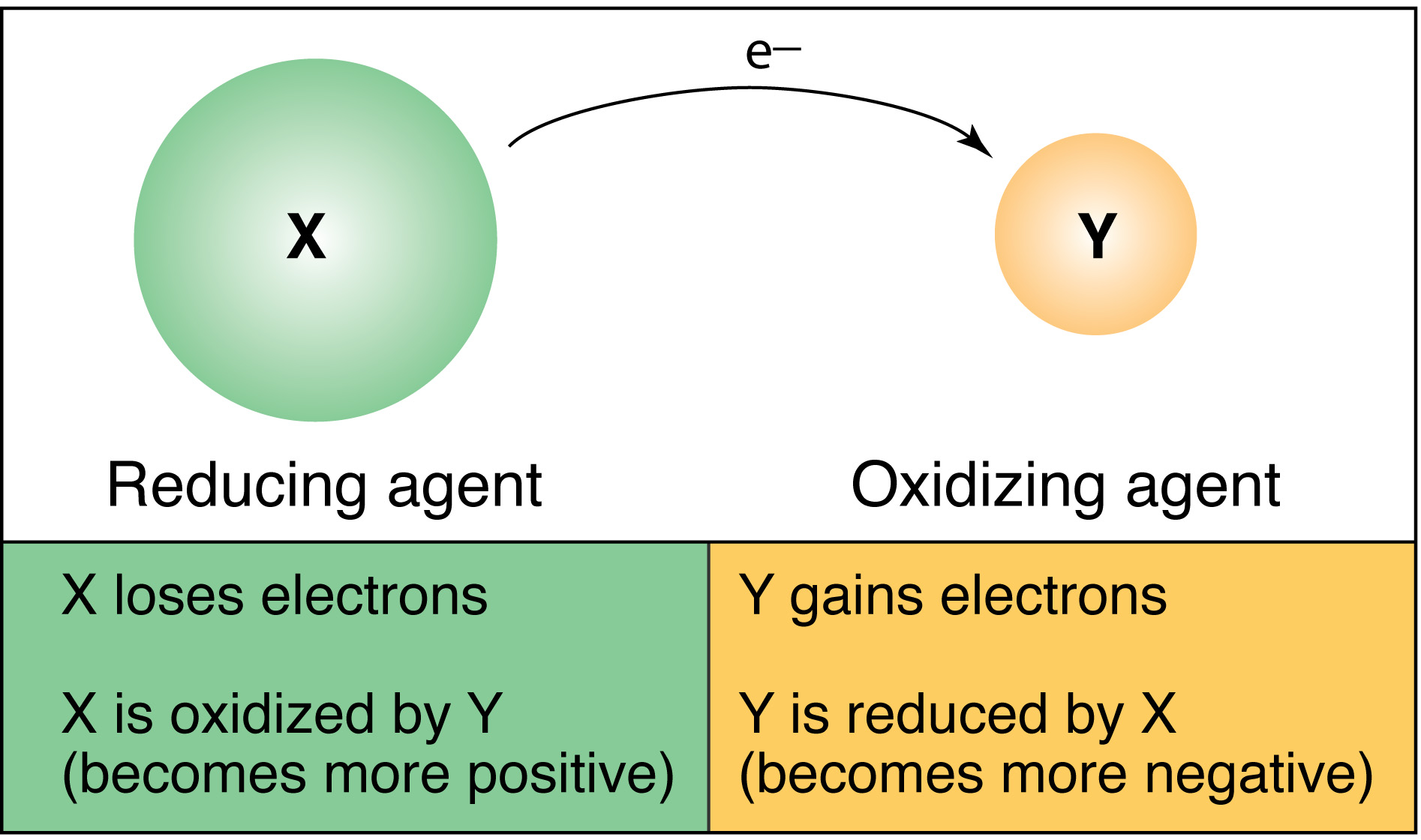
Oxidation–reduction reactions, commonly known as redox reactions, are reactions that involve the transfer of electrons from one species to another. The species that loses electrons is said to be oxidized, while the species that gains electrons is said to be reduced.
Can oxidation numbers identify redox reactions?
In other cases, however, it is not as obvious, particularly when the reaction in question involves only nonmetal substances. To help identify these less obvious redox reactions, chemists have developed the concept of oxidation numbers, which provides a way to track electrons before and after a reaction.

Redox titrations Chemical reactions AP Chemistry Khan Academy

Worked example: Balancing a simple redox equation Chemical reactions AP Chemistry Khan Academy

Worked example: Balancing a redox equation in acidic solution AP Chemistry Khan Academy
|
Unit 2 Redox and Electrochemistry Notes
Steps in balancing redox reactions. 1. Split the reaction into 2 half Balancing Redox Example #2 (shorter balancing and more typical for AP Chemistry). |
|
Worksheet # 5 Balancing Redox Reactions in Acid and Basic
WS #5 Balancing Redox Reactions in Acid and Basic Solution. Balance each redox equation. Assume all are spontaneous. Use the half reaction method. 1. 2O2-. +. |
|
Unit 2 Redox Electrochem
Notice that in the final step that half reactions are combined and the electrons canceled. Page 21. AP Chemistry. Balance the following reaction. MnO. |
|
Chapter 20 Worksheet: Redox I. Determine what is oxidized and
28 Jun 2010 Determine what is oxidized and what is reduced in each reaction. Identify the oxidizing agent and the reducing agent also. 1. 2Sr + O2. 2SrO. 2 ... |
|
Exercise 4.11 - Oxidation Numbers and Redox Reactions - Answers
AP Chemistry. 1. Exercise 4.11. Oxidation Numbers and Redox Reactions. Name DIRECTIONS: Identify whether the following equations describe redox reactions or ... |
|
AP Chemistry 2018 Free-Response Questions
AP® CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following oxidation-reduction reaction represented by the balanced equation above. (a) ... |
|
AP Chemistry 2021 Free-Response Questions
AP® CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following (e) Is the reaction a redox reaction? Justify your answer. (f) The reaction ... |
|
Title: Redox reactions
Title: Electrochemistry. Target Audience: AP Chemistry students (possibly Honors). Time: Three class periods (90 minutes each). Prior Knowledge: Students |
|
AP® Chemistry 2007 Free-Response Questions
ADVANCED PLACEMENT CHEMISTRY EQUATIONS AND CONSTANTS energy velocity OXIDATION-REDUCTION; ELECTROCHEMISTRY cell cell cell. . [C] [D] where A. B. C. D. [A] [B]. |
|
Untitled
AP Chemistry. 3. Half-Reactions. Name. OXIDATION-REDUCTION EQUATIONS. The Half-Reaction Method: 1. 2. 3. 4. 5. Write the equation as two half-reactions. Include |
|
AP Chemistry 2021 Free-Response Questions
AP Central is the official online home for the AP Program: AP® CHEMISTRY EQUATIONS AND CONSTANTS ... (e) Is the reaction a redox reaction? |
|
Unit 2 Redox and Electrochemistry Notes
information about redox reactions needs to be understood. REDOX Balancing Redox Example #2 (shorter balancing and more typical for AP Chemistry). |
|
AP® Chemistry 2007 Free-Response Questions
The following questions relate to the synthesis reaction represented by the chemical equation in the box above. (a) Calculate the value of the standard free |
|
A.P. Chemistry Practice Test - Ch. 17: Electochemistry MULTIPLE
20 févr. 2018 ) Which of the following reactions is a redox reaction? (a) K2CrO4 + BaCl2 ¬ BaCrO4 + 2KCl. (b) Pb22+ + 2Br- ¬ PbBr. (c) Cu + ... |
|
2021 AP Exam Administration Student Samples: AP Chemistry Free
AP®. Chemistry. Sample Student Responses and Scoring Commentary must determine with evidence |
|
Oxidation-Reduction Reactions
Oxidation-reduction reactions are also known as redox reactions. • Def: Redox reactions describe all chemical reactions in which there is a net change in. |
|
Exercise 18.1 - Balancing Redox Equations
AP Chemistry. Exercise 18.1. Balancing Redox Equations. 1. 2. The oxidation number of alkali metals in compounds is +1 and that of alkaline earths in |
|
NIE Videos
AP Chem Net Ionic Equations 1 Balance and Redox Reactions I ... AP Chemistry: Balancing Redox Reactions from Rene McCormick http://vimeo.com/15026695. |
|
AP Chemistry Sample Student Responses and Scoring Commentary
College Board Advanced Placement Program |
|
AP Chemistry 2018 Free-Response Questions
AP Chemistry. Free-Response Questions AP® CHEMISTRY EQUATIONS AND CONSTANTS ... oxidation-reduction reaction represented by the balanced equation above. |
|
RedOx Reactions
Oxidation-reduction reactions are also known as redox reactions • Def: Redox reactions describe all chemical reactions in which there is a net change in |
|
REDOX REACTIONS - NCERT
balance chemical equations using (i) oxidation number (ii) half reaction method; • learn the concept of redox reactions in terms of electrode processes UNIT 8 |
|
Chapter 19: Redox Reactions
reduction are complementary—as an atom is oxidized, another atom is reduced 19 2 Balancing Redox Equations MAIN Idea Redox equations are balanced |
|
MECHANISMS OF REDOX REACTIONS OF SIMPLE CHEMISTRY
MECHANISMS OF REDOX REACTIONS OF SIMPLE CHEMISTRY H Taube Department of Chemistry, University of Chicago, Chicago, Illinois I Introduction |
|
RedOx Chemistry
Check if the two are a RedOx pair If not, reverse the more negative half reaction III For each half reaction, balance elements other than oxygen and hydrogen |
|
Oxidation-Reduction Reactions - An Introduction to Chemistry
convention, any chemical change in which an element loses electrons is called an oxidation (Figure 6 1) 208 Chapter 6 Oxidation-Reduction Reactions |
|
Organic Chemistry - University of California, Riverside
Oxidation and reduction reactions (redox reactions) involve the overall transfer of electrons from one species to another species The chemical species being |
|
1 Lecture 11 Redox Chemistry Many elements in the periodic table
Redox reactions are characterized by the transfer of electrons between chemical species: For example, see the following full reaction of cupric ion with metallic |
|
Oxidation-Reduction Experiments
Chem 112 OXIDATION-REDUCTION EXPERIMENT INTRODUCTION An oxidation-reduction (redox) reaction involves the movement of electrons from one |