ap chemistry redox reactions and electrochemistry test
|
AP Chemistry 2018 Free-Response Questions
2018 AP ® CHEMISTRY FREE-RESPONSE QUESTIONS The student reads in a reference text that NO(g) and NO 2 (g) will react as represented by the equation below Thermodynamic data for the reaction are given in the table below the equation NO(g) + NO 2 (g) R N 2 O 3 (g) 0 87 kJ/mol rxn D D S D −40 4 kJ/mol rxn −138 5 J/(K mol rxn) H D D G D ⋅ |
|
AP Chemistry Practice Test
Choose the one alternative that best completes the statement or answers the question The gain of electrons by an element is called __________ oxidation sublimation reduction disproportionation fractionation __________ is reduced in the following reaction: Cr2O72- + 6S2O32- + 14H+ ¬ 2Cr3+ + 3S4O62- + 7H2O |
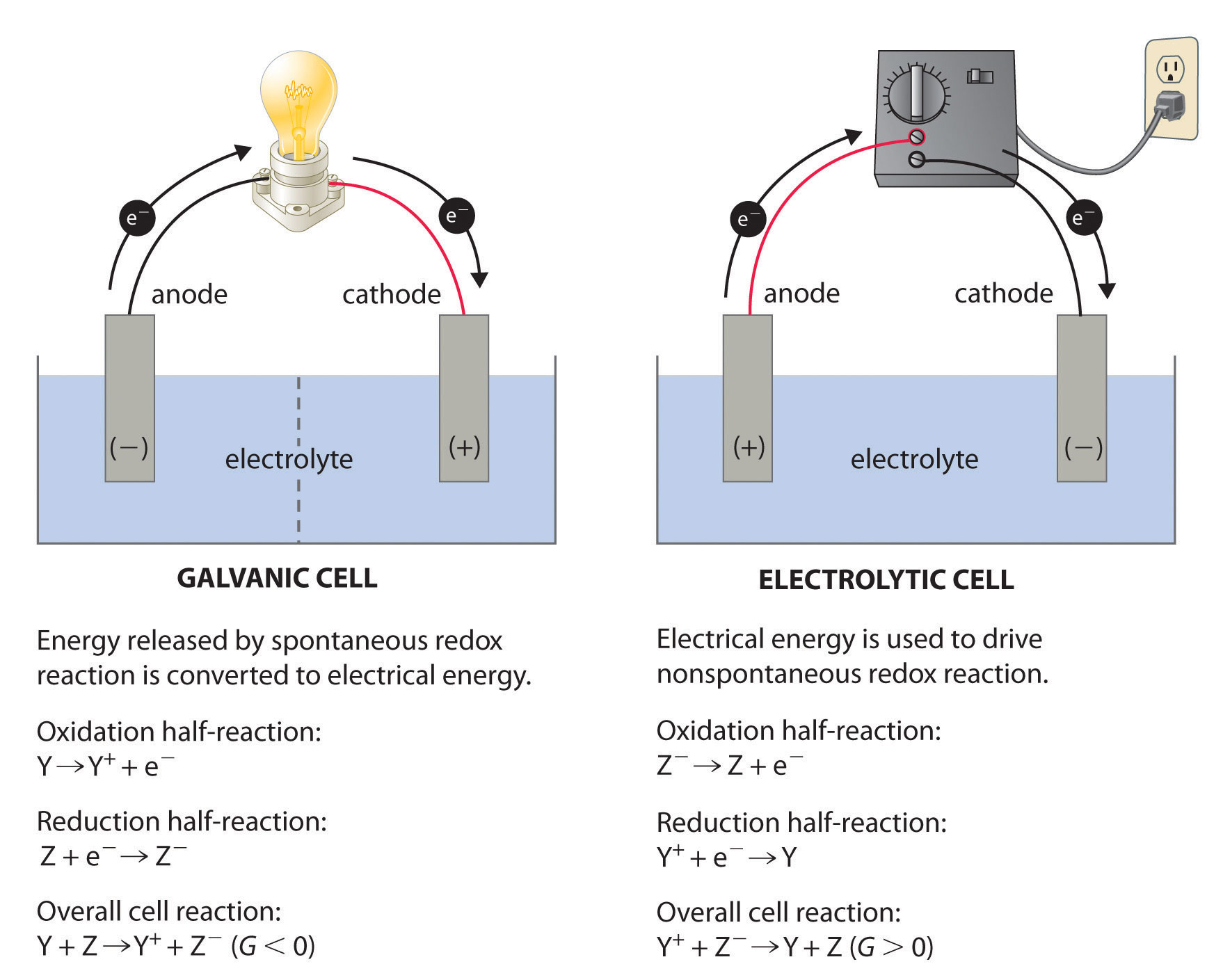
What is EMF in a redox reaction?
The emf can be thought of as being a measure of the driving force for a redox process. In an electrolytic cell an external source of energy is required to force the reaction to proceed. In order to drive the nonspontaneous reaction, the external emf must be greater than Ecell. 2) __________ is the oxidizing agent in the reaction below.
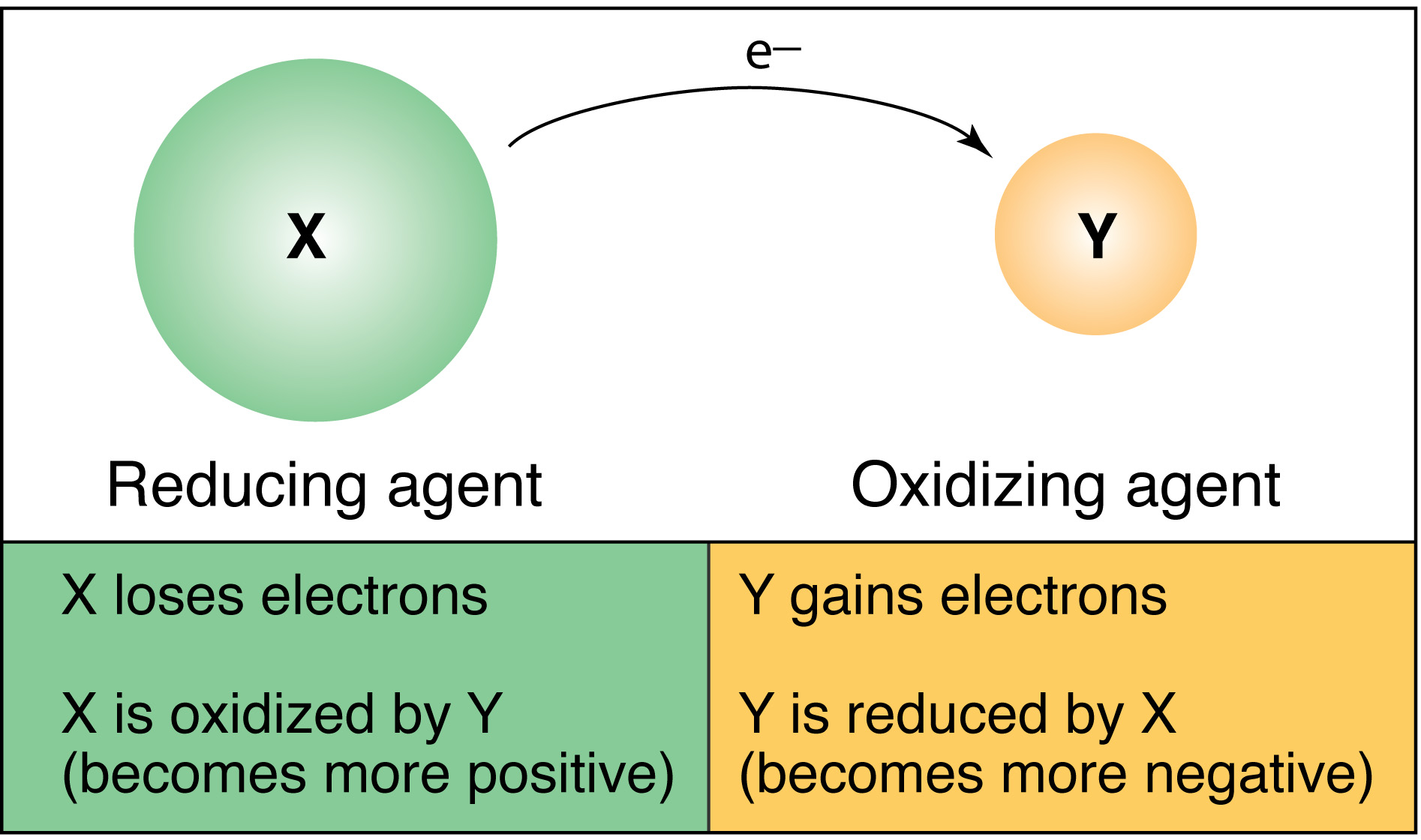
What are oxidation-reduction reactions?
called oxidation-reduction reactions (redox reactions). Oxidation involves loss of electrons (OIL). Reduction involves gain of electrons (RIG). Electrochemistry deals with relationships between electricity and chemical reactions. The oxidation numbers of Zn and H+ have changed. The ox# of Zn has increased from 0 to +2.
What is the difference between oxidation and reduction in chemistry?
Oxidation involves loss of electrons (OIL). Reduction involves gain of electrons (RIG). Electrochemistry deals with relationships between electricity and chemical reactions. The oxidation numbers of Zn and H+ have changed. The ox# of Zn has increased from 0 to +2. The ox# of H has decreased from +1 to 0.
|
Test4 ch19 Electrochemistry Practice Problems
General Chemistry II. Jasperse. Electrochemistry. Extra Practice Problems. Oxidation Numbers p1 Free Energy and Equilibrium p10. Balancing Redox; Electrons |
|
A.P. Chemistry Practice Test - Ch. 17: Electochemistry MULTIPLE
Feb 20 2018 ) The purpose of the salt bridge in an electrochemical cell is to ______. A) provide a source of ions to react at the anode and cathode. B) ... |
|
AP Chemistry 2018 Free-Response Questions
AP® CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following symbols have the definitions specified unless otherwise noted. |
|
AP Chemistry CHAPTER 20- Electrochemistry
called oxidation-reduction reactions (redox reactions). •Electrochemistry deals with relationships between ... AP Chemistry Chapter 20 Practice Test. |
|
AP Chemistry 2021 Free-Response Questions
AP® CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following symbols have the definitions specified unless otherwise noted. |
|
AP Chemistry 2019 Free-Response Questions
AP® CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following symbols have the definitions specified unless otherwise noted. |
|
Electrochemistry Electrochemistry
electrochemical cell that converts the chemical energy of a spontaneous redox reaction into electrical energy. In this device the Gibbs energy of. |
|
AP Chemistry 2016 Free-Response Questions
AP. ®. CHEMISTRY EQUATIONS AND CONSTANTS. Throughout the exam the following symbols have the definitions specified unless otherwise noted. |
|
AP Chemistry Student Sample Q7 from the 2017 Exam Administration
College Board Advanced Placement Program |
|
AP Chemistry Practice Exam from the 2018 Administration
NOTE: This is a modified version of the 2018 AP Chemistry Exam. This exam may not be posted (i) A student claims that the reaction is a redox reaction. |
|
Test4 ch19 Electrochemistry Practice Problems
Key Equations Given for Test: E˚cell=E˚reduction + E˚ Balancing Redox; Electrons Transferred; Oxidizing Agents; Reducing Agents 6 Balance the How many electrons are transferred in this redox reaction as written? 2CH3OH + 3O2 |
|
AP Chemistry Practice Test - Prospect Ridge Academy
20 fév 2018 · ) Which of the following reactions is a redox reaction? ) The purpose of the salt bridge in an electrochemical cell is to ______ A) provide a |
|
Redox and Electrochemistry Test Review Sheet
Regents Chemistry Redox and Electrochemistry Test Review Sheet Sample: Write the half reactions for the redox reaction: Fe + Cl2 → FeCl2 Answer: |
|
Redox and Electrochemistry Multiple Choice Review CONCEPT
Redox and Electrochemistry Multiple Choice Review CONCEPT REVIEW the oxidation-reduction reactions and the Activity Series Reference Table, predict which of the It changes electrical energy into chemical energy b It is a type of |
|
Redox Date: R Chem - Ms Huenger Practice Test Period
Practice Test Period: D) –2 to 0 4 Given the reaction that occurs in an electrochemical cell: In an oxidation-reduction reaction, the number of electrons lost |
|
Ch 17 Electrochemistry
Electrochemistry: The area of chemistry that examines the transformations between chemical Example 18 1 Half-Reaction Method of Balancing Aqueous Redox Equations in Acidic When run directly in a test tube • Cu metal plates out on |
|
Wizard Test Maker
CHEMISTRY Redox Practice Test 2008 Given the electrochemical cell reaction: Zn(s) + Ni2+ diagram of the chemical cell at 298 K and on the equation |
|
3202 guide work4
migrate to oppositely charged electrodes and chemical reactions take place Quantitative aspects of this oxidation-reduction reactions could be investigated Science Curriculum 214-14 construct and test a prototype of a device or Students should understand from the beginning that electrochemistry involves the study |
|
Unit 12: Electrochemistry
Unit 12: Electrochemistry-Lecture notes Regents Chemistry '14-'15 Mr Murdoch Page 9 of 61 Website upload 2015 Lecture notes Redox Reaction Type |
|
Chem 106 Laboratory Manual, Experiment 9 - UCCS
Electrochemistry I – Galvanic Cell Introduction: Chemical reactions involving the transfer of electrons from one reactant to another are called A galvanic cell or voltaic cell is a device in which a redox reaction, such as the one in equation (4) volts, test the galvanic cell a third time and record the potential immediately after |










![The Direct Electrochemical Synthesis of [(C6H5)3Ph]2[CoCl4 The Direct Electrochemical Synthesis of [(C6H5)3Ph]2[CoCl4](https://i0.wp.com/ytimg.googleusercontent.com/vi/AnIEenv0Ek4/hqdefault.jpg?resize\u003d650)