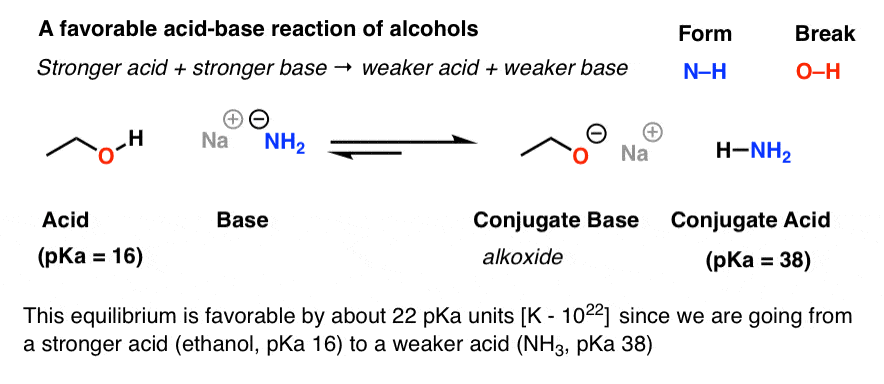strong acids and bases practice problems
|
Organic chemistry i practice problems for bronsted-lowry acid-base
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY Compare the strengths of the conjugate bases and remember that the weaker the base the stronger the |
What are the 5 strongest acids?
you'd wind up with a pH-neutral substance.
Mixing a strong acid and a strong base in solution results in a neutral solution.What are some examples of strong acids bases?
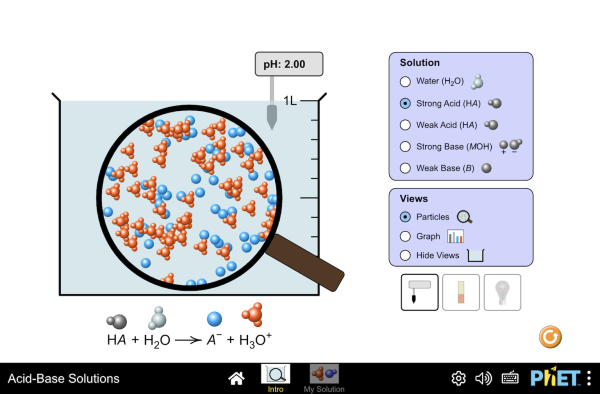
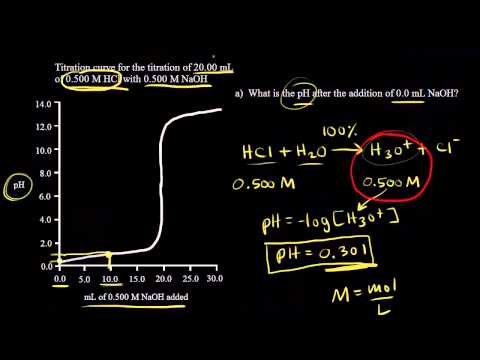
If the concentration of the acid is known, then measurement of the pH using a pH meter (or similar) will tell us whether or not an acid is strong or weak.
Stronger acids have lower pH values for the same concentration.How do you test for a strong acid?
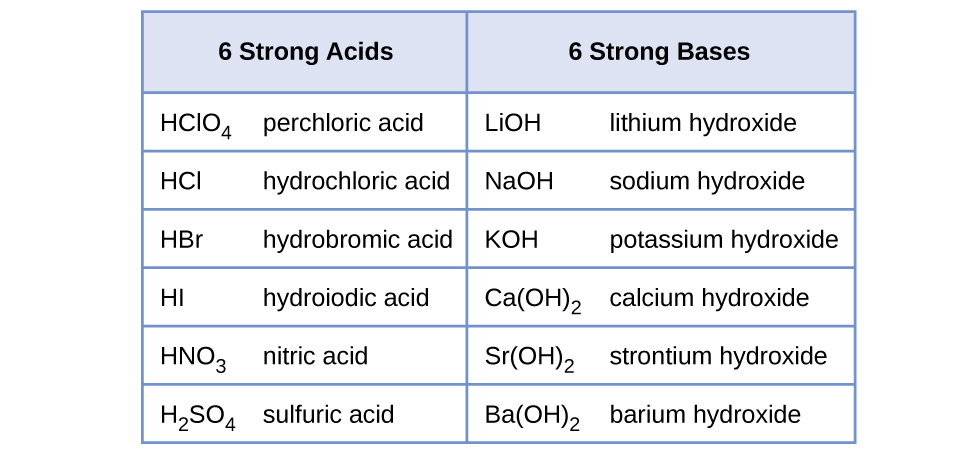
Strong acids include:
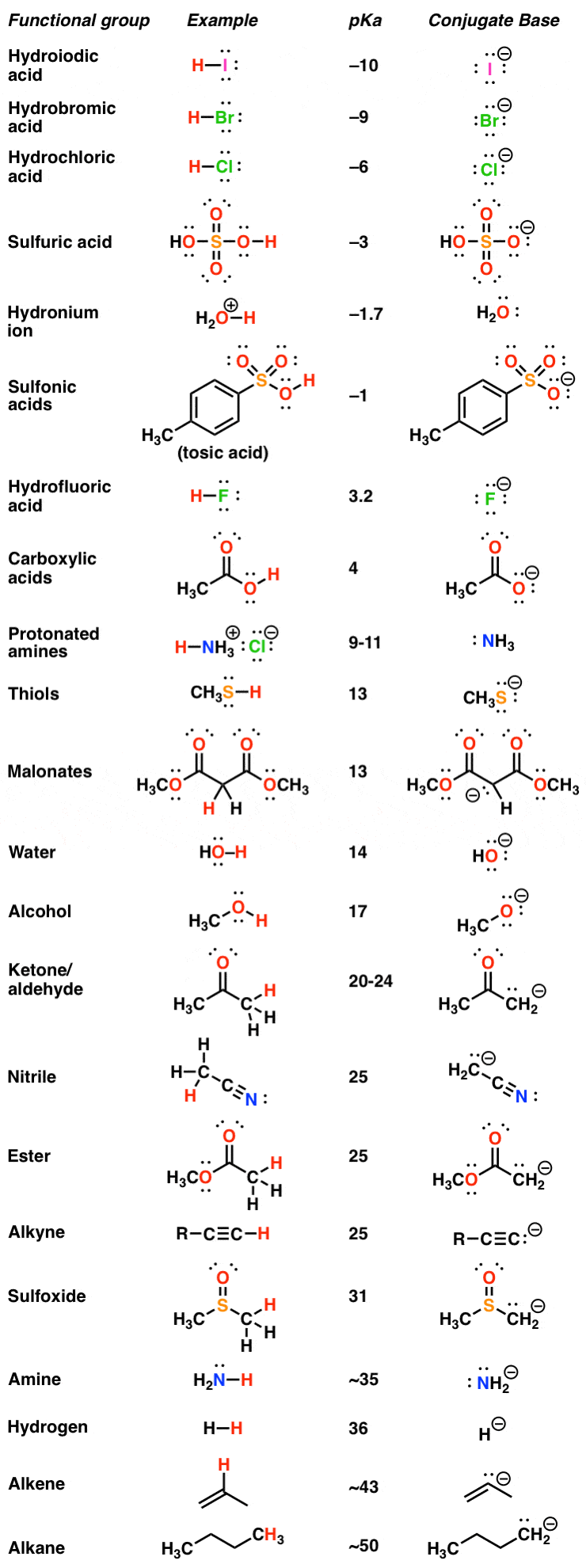
Hydrochloric acid (HCl): pKa -7.Sulfuric acid (H2 SO4: pKa -3 to -2.Nitric acid (HNO3): pKa 1.3.Hydroiodic acid.Chloric acid.Hydrobromic acid.Perchloric acid.
|
Test2 ch17a Acid-Base Practice Problems
Extra Practice Problems Recognizing Strong versus Weak Acids; ... In the Brønsted–Lowry definition of acids and bases an acid ______. |
|
Chapter 11 - Acids and Bases - Practice Problems - Section 11.1
An Arrhenius acid produces H and an Arrhenius base produces OH in aqueous solutions Goal: Write equations for the dissociation of strong and weak acids; ... |
|
Chapter 11 – Acids and Bases – Practice Problems Section 11.1
An Arrhenius acid produces H+ and an Arrhenius base produces OH- in aqueous Goal: Write equations for the dissociation of strong and weak acids; ... |
|
Ch 7 Practice Problems
Identify the Brønsted acids and bases in the following equation (A The acids HC2H3O2 and HF are both weak but HF is a stronger acid than HC2H3O2. |
|
1. For each of the species below identify the most acidic proton and
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY Which is the stronger acid and what accounts for this large difference in relative acidity? |
|
Untitled
Calculating pH pOH |
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the Base. Base. Strength. 1. Strong Acids. H-Cl H2SO4. |
|
1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
Extra Practice Problems. General Types/Groups of problems: Answer: A buffer consists of a weak acid and its conjugate base in roughly equal amounts. |
|
1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
Extra Practice Problems. General Types/Groups of problems: Answer: A buffer consists of a weak acid and its conjugate base in roughly equal amounts. |
|
Where To Download Solutions Acids And Bases Review [PDF
Studies Practice of Chemistry essays |
|
Ch 17a Acid-Base Practice Problems
The base ionization constant Kb describes which of the following reactions for a weak base, B, in aqueous solution? (Note: often the base will be anionic rather |
|
Acid-Base Practice Problems
Acid-Base Practice Problems A Identify each chemical as either an “acid” or a “ base” in the following reactions, and identify “conjugate” Entry Class Structure Ka Acid Strength Base Base Strength 1 Strong Acids H-Cl, H2SO4 102 2 |
|
Chapter 11 – Acids and Bases – Practice Problems Section 111
An Arrhenius acid produces H+ and an Arrhenius base produces OH- in Goal: Write equations for the dissociation of strong and weak acids; identify the |
|
Solutions to Review Problems for Acid/Base Chemistry - CSUN
Solutions to Review Problems for Acid/Base Chemistry 1 Glacial acetic samples If 5 0 mL of 6 0 M HCl are added to one sample, and 5 0 mL of 6 0 M The added HCl is neutralized by the weak base and a new buffer is formed mol HCl |
|
Acid Base Practice Problems 1 Write the formula for the conjugate
When solving problems identification of the chemicals is very important Is the chemical a strong acid (ionizes 100 ), a weak acid (partial ionization and has a |
|
Unit 7J Practice Problems I Acids and Bases - Georgia Public
b) What is the molarity of the acid? 9 What is the word equation for the neutralization of a strong acid and strong base? 10 In a neutral solution, moles of equal the |
|
Acid Base Practice Test - Houston ISD
weak electrolytes d also strong acids ____ 15 Whose definition of acids and bases emphasizes the role of protons? a Brønsted |
|
AP Chemistry Practice Test: Ch 14, Acids and Bases
13 déc 2017 · 10) Sodium hydroxide is a strong base This means that ______ A) aqueous solutions of NaOH contain equal concentrations of H+ (aq) and |
|
Ch14 Acids Bases Practice Problems - keypdf
11) Which pair of acids would each react with barium hydroxide and have the net ionic equation: weak acil H + OH → H2O Stromo base a) HCl and HNO b) HBr |