surface chemistry class 12 pdf
|
Chemistry Notes for class 12 Chapter 5 Surface
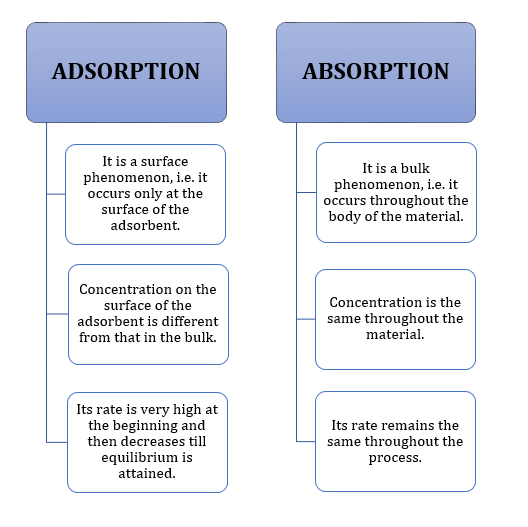
The molecular species accumulates at the surface is termed as adsorbate and the material on the surface of which the adsorption takes place is called adsorbent |
|
CBSE NCERT Solutions for Class 12 Chemistry Chapter 7
Solution: Chemisorption is highly specific in nature In chemisorption atoms and molecules are attached to the solid surface through chemical bonds (Covalent |
|
Surface chemis ace chemis ace chemistry
23 avr 2018 · (iii) Hydrogen on finely divided nickel (iv) Oxygen on metal surface 23/04/18 Page 3 Surface Chemistry 65 12 On the basis of data given |
|
Surface Chemistry Unacademy
The phenomenon of adherence of liquid or gaseous molecules on the surface of a solid by physical or chemical processes is termed as adsorption Concept Ladder |
| Surface Chemistry |
|
Unit 5 surface chemistrypdf
SURFACE CHEMISTRY CLASS-12 (CBSE) 1 no com 4ono com 4ono com 4ono com 4on S No QUESTION YEAR Note: The yellow coloured questions are from the reduced |
|
Formulae For SURFACE CHEMISTRY
When the catalyst and the reactants are in different phases the catalytic process is said to be heterogeneous catalysis 6 Activity of catalyst The abiloity |
Which book is surface chemistry class 12?
NCERT Book Class 12 Chemistry-I Chapter 05: Surface chemistry.
The important topics in surface chemistry are adsorption, catalysis, colloids and emulsions.
What is surface area in chemistry class 12?
The surface area of a solid reactant can be increased which leads to increase the rate of a chemical reaction.
Cutting the substance into small pieces or grinding it into a powder accomplishes this.
What is surface chemistry class 12?
What Is Surface Chemistry? It is the study of the chemical phenomena that occur at the interface of two surfaces which can be solid-liquid, solid-gas, solid-vacuum, liquid-gas, etc.
Some applications of surface chemistry are known as surface engineering.
|
Lech105.pdf
features of surface chemistry such as adsorption 12. 22. 11. 2. Solution. Both the reactants and the catalyst are in the same phase. |
|
Surface chemistry
features of surface chemistry such as adsorption 12. 22. 11. 2. Solution. Both the reactants and the catalyst are in the same phase. |
|
Chemistry Notes for class 12 Chapter 5 Surface Chemistry
www.ncerthelp.com (Visit for all ncert solutions in text and videos CBSE syllabus |
|
CBSE NCERT Solutions for Class 12 Chemistry Chapter 7
Solution: Chemisorption is highly specific in nature. In chemisorption atoms and molecules are attached to the solid surface through chemical bonds (Covalent |
|
CHEMISTRY (043) Class XI
4. Chemical. Kinetics. Concept of collision theory (elementary idea no mathematical treatment) |
|
UNIT 4 SURFACE CHEMISTRY
12. For adsorption from solution pressure (P) is replaced by equilibrium concentra- tion (C). 13. A catalyst is a |
|
Formulae For SURFACE CHEMISTRY
XII Chemistry. CHAPTER 5 - SURFACE CHEMISTRY FORMULAS- SURFACE CHEMISTRY ... 12. Distinction between true solution colloids and Suspension. |
|
CHEMISTRY (043) SYLLABUS FOR SESSION 2021-22 CLASS XI
1 Some Basic Concepts of Chemistry. 10. 11. 2 Structure of Atom. 12 Surface Chemistry: Adsorption - physisorption and chemisorption factors affecting ... |
|
Deleted portion from Class XII syllabus-Chemistry Unit Deleted
Temperature Dependence of the Rate of a Reaction. • Collision theory of chemical reactions. Unit V: Surface Chemistry. • Catalysis. • Emulsions. Unit 6 General. |
|
Unit 5.pmd
23 ?????? 2018 Physical adsorption of a gaseous species may change to chemical adsorption ... Surface Chemistry. 65. 12. On the basis of data given below ... |
|
Surface chemistry - NCERT
Surface chemistry deals with phenomena that occur at the surfaces (viii) Froth floatation process: A low grade sulphide ore is concentrated by separating it 12 22 11 2 Solution Both the reactants and the catalyst are in the same phase |
|
Chemistry Notes for class 12 Chapter 5 Surface Chemistry - Ncert Help
Adsorption Due to unbalanced attraction forces, accumulation of molecular species at the surface rather than in the bulk of a solid or liquid is termed as |
|
CBSE NCERT Solutions for Class 12 Chemistry Chapter 7
Solution: Chemisorption is highly specific in nature In chemisorption, atoms and molecules are attached to the solid surface through chemical bonds (Covalent |
|
CHEMISTRY CLASS-XII - edudel
Unit III Electrochemistry 12 23 Unit IV Chemical Kinetics 10 Unit V Surface Chemistry 08 Unit VI General Principles and Processes of Isolation of Elements |
|
UNIT -5 SURFACE CHEMISTRY - PUE
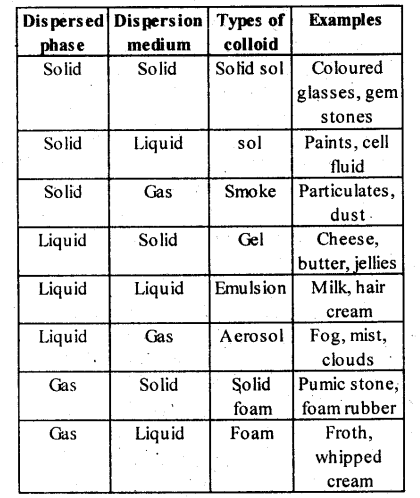
12 What is the dispersion medium in gel? Solid 13 Between Na2SO4 and Na3PO4 which has greater power to coagulate a positively charged |
|
Surface Chemistry
Surface Chemistry Author : 24Tutors Download the 2019 version of NCERT solutions for class 12 Chemistry for your College level exams and helpful for JEE |
|
Salwan Public School Class XII Assignment Surface Chemistry 1
Class XII Assignment Surface Chemistry 1 Differentiate between Adsorption and Absorption 2 Define the terms adsorbate and adsorbent 3 What is |
|
Chemistry Class 12 Chapter 5 NCERT Solution - Mywayteaching
www mywayteaching com Class XII Chapter 5 – Surface Chemistry Chemistry Page 1 of 18 Question 5 1: Write any two characteristics of Chemisorption |