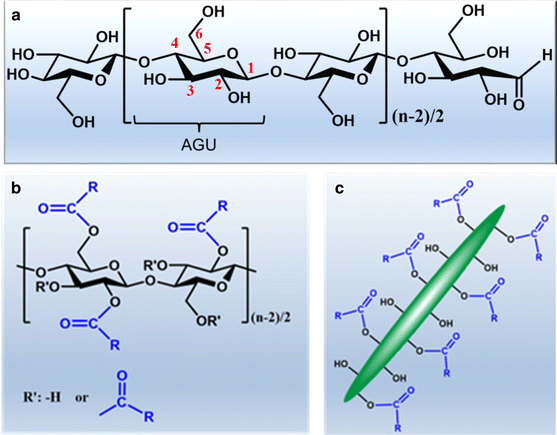
the fischer esterification mechanism
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification wherein a Make sure that the system to be heated is not a closed system!! Have |
What is the general reaction for Fischer esterification?
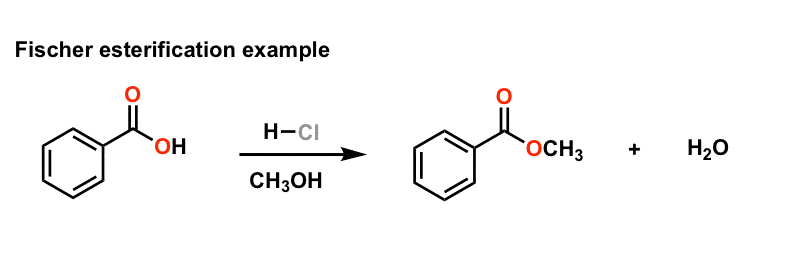
In this reaction there is an equilibrium between the starting materials (carboxylic acid + alcohol) and the products (ester + water).
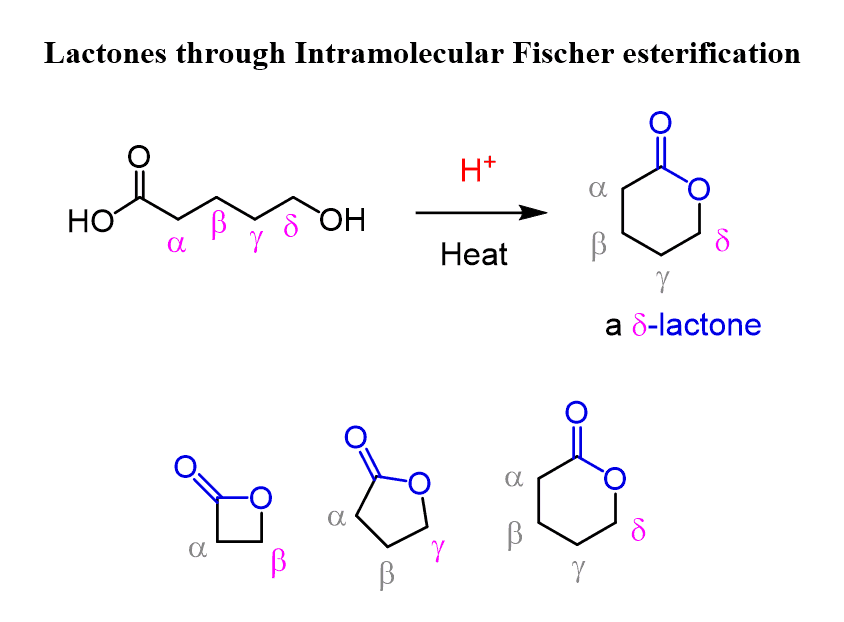
Cyclic esters can be formed under these conditions, which are known as lactones.16 nov. 2022What is the mechanism of action of esterification?
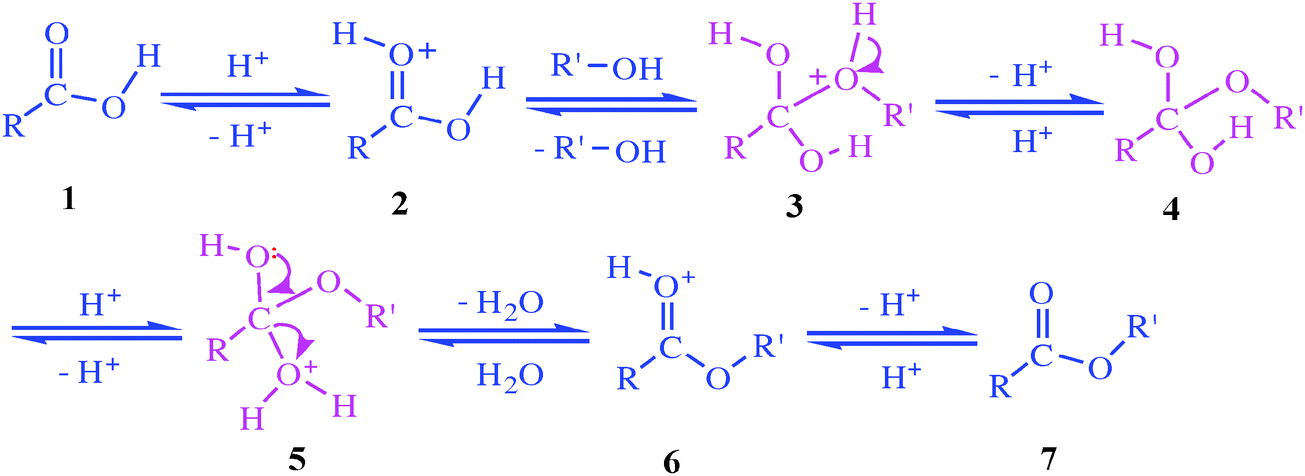
The mechanism of esterification reactions is the oxygen in an alcohol interacting with a carbocation in an acid.
This complex then transfers the proton from the alcohol to another proton acceptor to act as a leaving group, leaving an -OR group where there was once a hydroxyl (-OH) group.What is the method of forming an ester in Fischer esterification?
One common way to make an ester is Fischer esterification, where a carboxylic acid and an alcohol react in the presence of an acid catalyst to form the ester and water.
The R group and the carbonyl come from the carboxylic acid, and the alkoxy or aryloxy group with the R' comes from the alcohol.Esters are produced when carboxylic acids are heated with alcohols in the presence of an acid catalyst.
The catalyst is usually concentrated sulphuric acid.
Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones where the carboxylic acid contains a benzene ring).
|
Exp 22 Fischer Esterification_2017
Another frequently used reaction to form esters is the Fischer esterification which Mechanism of the Acid Catalyzed Fischer Esterification. |
|
Synthesis of Isopentyl Acetate.pdf
When catalyzed by a strong acid usually sulfuric acid or p-toluenesulfonic acid |
|
Study of the kinetics and equilibria of the oligomerization reactions
Mar 15 2013 The existing chemical literature (Loudon |
|
215-216 HH W12-notes-Ch 15
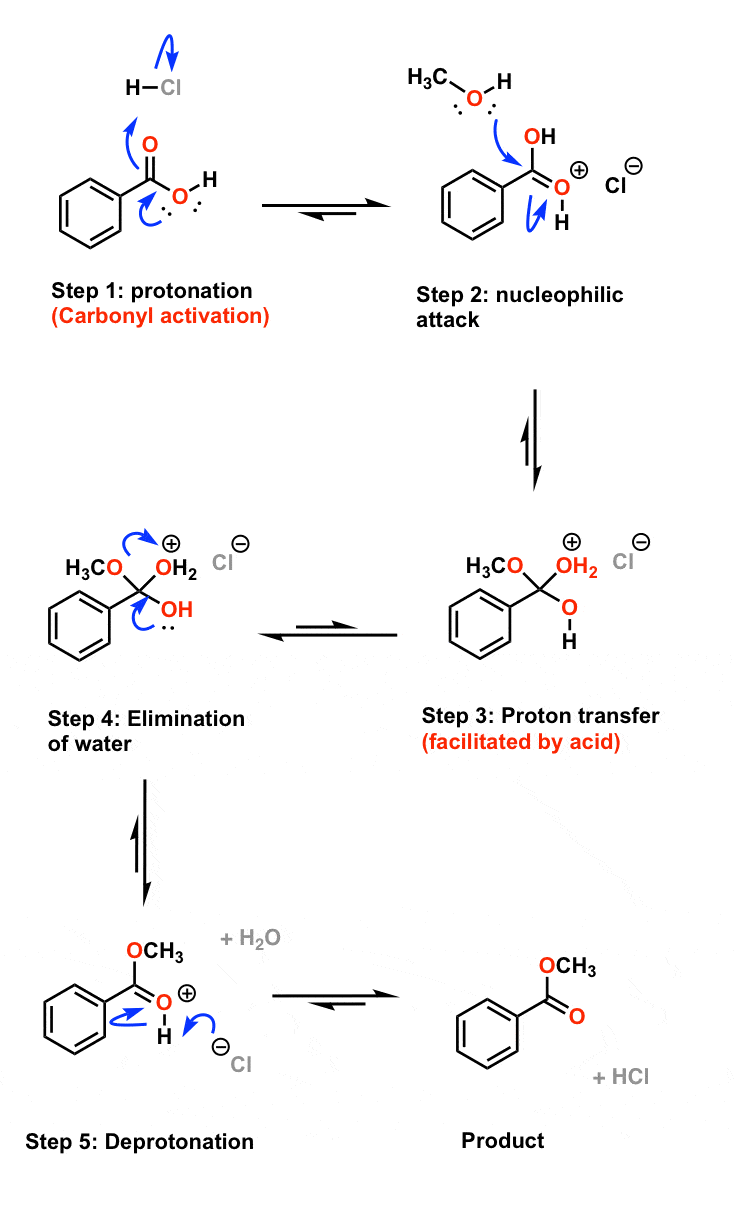
Oct 5 2012 (2) The mechanism for the acid-catalyzed esterification [Commonly referred to as the Fischer esterification: see pp 623-624 of the textbook] ... |
|
Example: esterification of benzoic acid to methyl benzoate.
1) Fischer Esterification: MECHANISM (Fischer esterification). The overall process of esterification is one involving an equilibrium among a variety of ... |
|
10. Fisher Esterification: Preparation of Banana Oil
The overall reaction for Fischer esterification. The overall mechanism for a general acid and alcohol is depicted in Figure 2. R. O. O. |
|
87 Steglich Esterification
277 The Steglich esterification is an ester coupling reaction (compare to the peptide (amide) coupling mechanism in Chapter 74 or the Fischer esterification |
|
Green and Efficient Esterification Method Using Dried Dowex H+/NaI
May 22 2019 mechanism might be possible. ? CONCLUSIONS. The study clearly and unambiguously highlights the straightforward nature of these kinds of ... |
|
Www.rsc.org/pccp
The acid-catalyzed carboxylic acid esterification and ester hydrolysis mechanism proposed by Ingold. Has long been known certain substituted benzoic acids |
|
Chapter 2. Structure and Properties
Preparation of Esters. ? Fischer esterification: acids & alcohol. ? mechanism: acid catalyst/equilibrium. ? lactones: cyclic esters |
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification, wherein a carboxylic acid is treated with an alcohol in the presence of a mineral inorganic acid catalyst These are long, straight chain carboxylic acids (C12-C40) found as ester derivatives in oils, fats, and waxes |
|
Synthesis of an Ester: Fischer Esterification The ester is synthesised
The ester is synthesised via a Fischer esterification – the reaction of a carboxylic acid with an alcohol in the presence of a mineral acid catalyst Using butyric |
|
Fischer Esterification - MIT OpenCourseWare
Organic Synthesis: Synthesizing an ester from an acid catalyzed reaction of a carboxylic for the fisher acid catalyzed esterification reaction is shown in (5 3) |
|
Fischer Esterification - MIT OpenCourseWare
Organic Synthesis: Synthesizing an ester from an acid catalyzed reaction of a carboxylic for the fisher acid catalyzed esterification reaction is shown in (5 3) |
|
19_13_15htmlppt [Read-Only]
Reaction with Thionyl Chloride (Section 12 7) Reactions But first we revisit acid-catalyzed esterification to examine its Mechanism of Fischer Esterification |
|
215-216 HH W12-notes-Ch 15
5 oct 2012 · Mechanism for the hydrolysis of an ester under acidic conditions is virtually identical with that for the esterification from an acid, but to the reverse |
|
Exp 22 Fischer Esterification_2017
reaction is catalyzed by addition of an acid such as sulfuric acid or phosphoric acid Figure 4 shows the mechanism for the acid catalyzed Fischer esterification |
|
Lab9 methyl benzoate handout1
Example: esterification of benzoic acid to methyl benzoate MECHANISM ( Fischer esterification) The overall K, for the reaction-must remain constant at a |
|
Synthesis of Isopentyl Acetate
To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer esterification reaction Introduction Esters are derivatives of carboxylic acids |
|
Anleel J Shabdin, Michael D Drake* - California State University
Given that it is safe, cheap, and versatile, we will use Fischer Esterification to synthesize the esters Fischer Esterification Reaction Mechanism 1 |
























![Fischer Esterification - [PDF Document] Fischer Esterification - [PDF Document]](https://imgv2-2-f.scribdassets.com/img/document/150540581/original/e004e504eb/1612802334?v\u003d1)