fischer esterification lab report conclusion
|
Fischer Esterification
In this experiment lauric acid (dodecanoic acid) is converted to ethyl laureate Lauric acid is representative of a class of molecules called fatty acids |
AIM: Investigate the formation of an Ester through the reaction of an alcohol with a carboxylic acid, where the ester formation is promoted by the presence of concentrated sulphuric acid acting as a catalyst.
What are the results of esterification?
Esterification is the process of combining an organic acid (RCOOH) with an alcohol (ROH) to form an ester (RCOOR) and water; or a chemical reaction resulting in the formation of at least one ester product.
Ester is obtained by an esterification reaction of an alcohol and a carboxylic acid.
What are the results of Fischer esterification?
Esters are formed from an esterification reaction, with simple esters being formed through Fisher esterification.
This reaction converts a carboxylic acid and alcohol into an ester with water as a by-product.
Fisher esterification is a reversible reaction that proceeds very slowly.
What is the conclusion for esterification lab report?
Conclusion. -The hypothesis made was correct; this experiment proved that esters can indeed be created by combining carboxylic acids and alcohols. Another source of error was that the ratio of carboxylic acid and alcohol might have slightly varied from each sample due to inaccurate measuring.31 jan. 2019
|
Experiment 14A: Isopentyl Acetate
14-Oct-2020 product's strong banana-like odor it was concluded that the product ... This experiment focused on the synthesis of an ester by a Fischer ... |
|
Fischer Esterification
In this experiment lauric acid (dodecanoic acid) is converted to ethyl laureate. Lauric acid is representative of a class of molecules called fatty acids. |
|
Microwave-Assisted Esterifications: An Unknowns Experiment
09-Sept-2015 B.A Simple Combinatorial Experiment Based on Fischer. Esterification. An Experiment Suitable for the First-Semester. Organic Chemistry Lab J. |
|
Esterification.pdf
21-Jul-2006 Pre- Lab Discussion: Esters are responsible for the smell of many fruits and perfumes. In this experiment a microscale technique is used to ... |
|
5.310 F17 Experiment 5: Fischer Esterification
EXPERIMENT #5. Fischer Esterification: The Organic Synthesis Isolation |
|
Ester Synthesis Lab (Student Handout)
Results. 6. Mechanism (In class). 7. Conclusion. Introduction Esters are produced by the reaction between alcohols and carboxylic acids. For example. |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
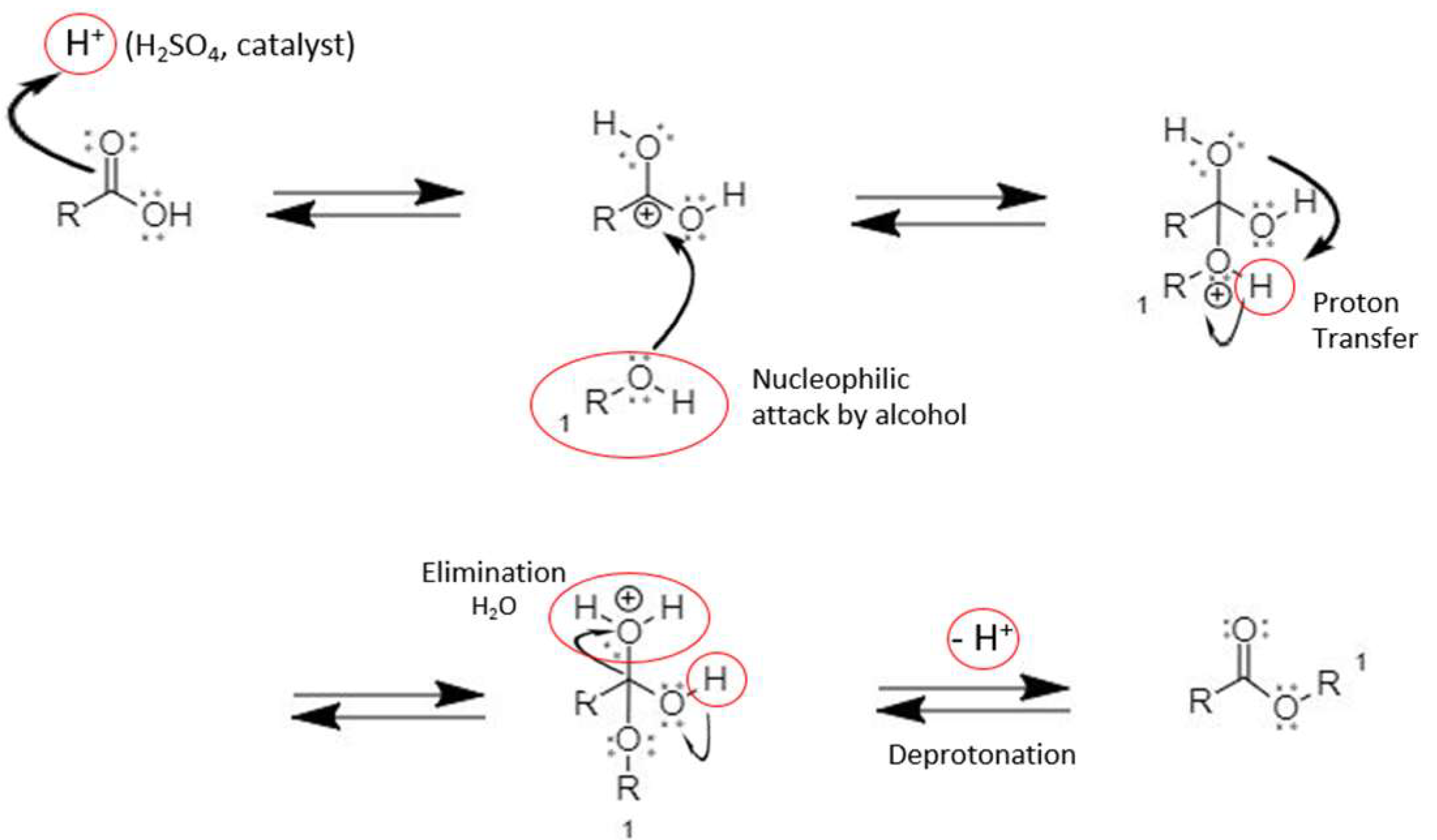
A carboxylic acid and an alcohol react in the presence of an acid catalyst to form an ester and water as shown in equation 1. This reaction termed Fischer |
|
Synthesis and Investigation of Thermal Properties of Highly Pure
30-Jun-2018 Based on an International Energy Agency (IEA) report [1] ... Reaction mechanism of Fischer esterification. ... Results and Discussion. |
|
Transmutation of Scent: An Evaluation of the Synthesis of Methyl
23-Sept-2020 Common fragrant esters synthesized by Fischer esterification. Laboratory Experiment pubs.acs.org/jchemeduc. © 2020 American Chemical Society and. |
|
Microwave-Assisted Esterification: A Discovery-Based Microscale
17-Jul-2014 opportunity for discussion of important chemistry concepts including acid ... In this experiment |
|
5310 F17 Experiment 5: Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and This is an integrated experiment, which combines techniques from Organic, Day 2— “ Introduction to Atmospheric Distillation” Assemble a distillation apparatus |
|
5310 F17 Experiment 5: Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and This is an integrated experiment, which combines techniques from Organic, Day 2— “ Introduction to Atmospheric Distillation” Assemble a distillation apparatus |
|
Lab 1: Synthesis
Results 6 Mechanism (In class) 7 Conclusion Introduction The compounds you Esters are produced by the reaction between alcohols and carboxylic acids |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
Video ~ A Brief Introduction to Fractional Distillation In this experiment you will react a carboxylic acid and an alcohol under acidic conditions to form |
|
Experiment 18 — Synthesis of Esters
The most common method for preparing esters is acid-catalyzed reaction of a carboxylic acid and an alcohol, a process known as Fischer esterification |
|
LABORATORY MANUAL FOR ORGANIC CHEMISTRY I
Lab Manual i TABLE OF CONTENTS Introduction 1 Laboratory safety 4 Laboratory operations 8 Experiment 1: Esterification reaction: Synthesis of n- butyl |
|
EXPERIMENT 5 ORGANIC SYNTHESIS: FISCHER
A carboxylic acid and an alcohol react to form an ester and water as shown in equation 1 below This reaction is reversible but proceeds slowly in either direction |
|
Improved Fischer Esterification of Substituted Benzoic Acid under
INTRODUCTION Microwave-assisted organic disadvantage in Fischer esterification lies in the equilibrium nature of the reaction It is even more difficult 4-Fluoro-3-nitrobenzoic acid was used in this experiment as a precursor to synthesise |