fischer esterification percent yield
|
Chemistry 209 Expt 5
catalyzed by acid but unlike Fischer esterification it is very fast and irreversible 2The term "percent yield” is really short for “actual yield as a |
|
Fischer Esterification
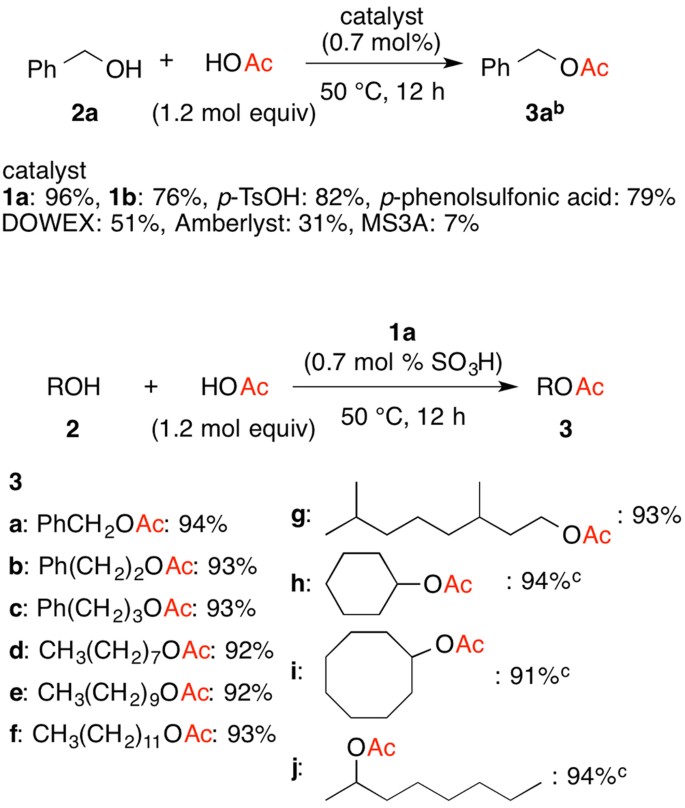
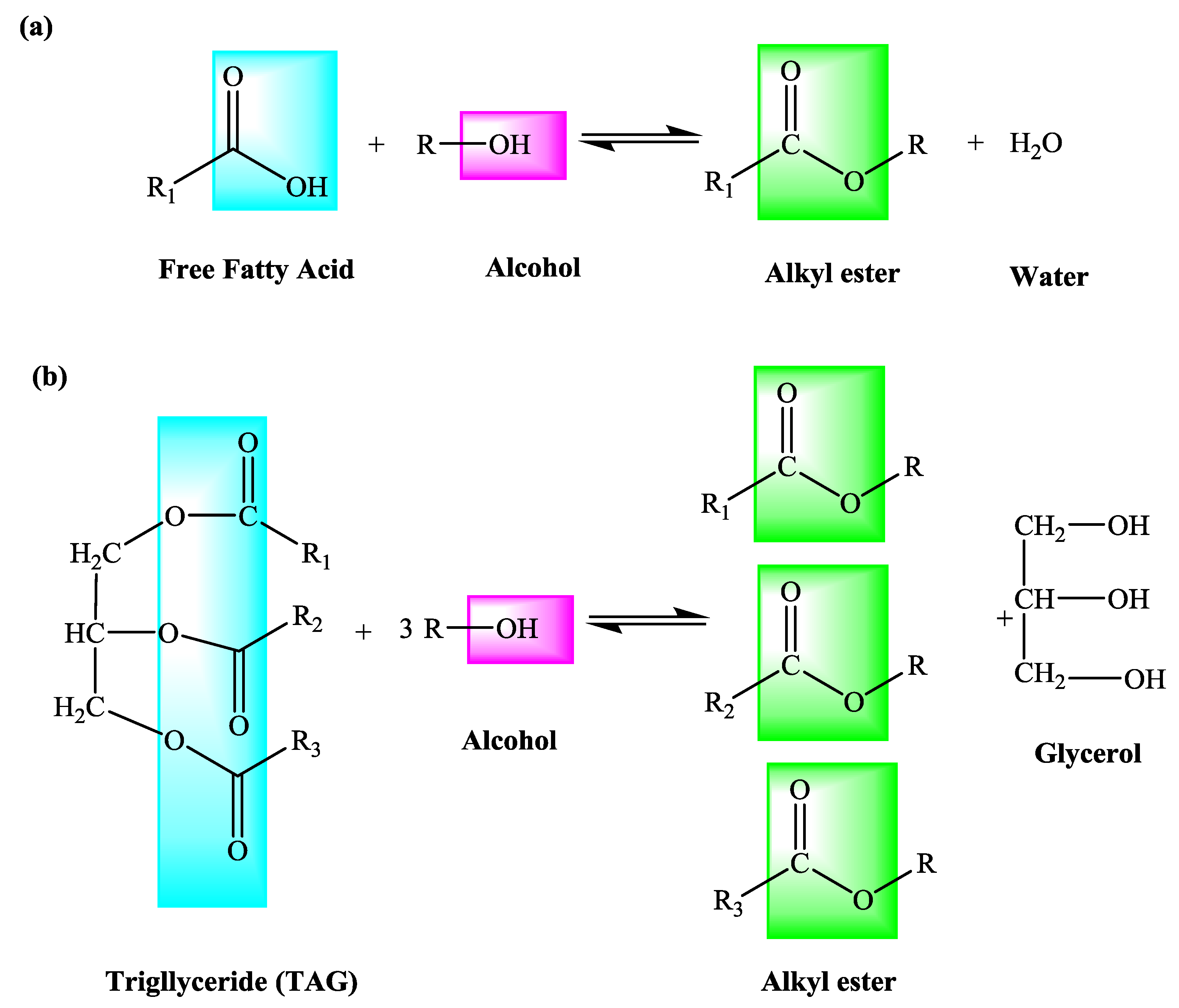
A typical procedure to synthesize esters is the Fischer esterification wherein a Calculate the percent yield 2 Analyze the IR spectrum and compare your |
Typical reaction times vary from 1–10 hours at temperatures of 60-110 °C.
Direct acylations of alcohols with carboxylic acids is preferred over acylations with anhydrides (poor atom economy) or acid chlorides (moisture sensitive).
What is the percent yield for the Fischer product?
Based on the experiment that was conducted the synthesis of isopentyl acetate from a carboxylic acid and an alcohol could be done by a Fisher Esterification reaction, and the percent yield of the product is about 61.9%.16 août 2017
What is the percent yield of an ester?
With a 1:1 mixture of carboxylic acid and alcohol, the esterification reaction reaches equilibrium with about 70% yield of the ester.26 mar. 2020
What is the yield of Fischer esterification?
This esterification reaction is reversible.
With a 1 to 1 mixture of the carboxylic acid and the alcohol, it tends to reach equilibrium with about a 70% yield of the ester at best.
|
Chemistry 209 Expt 5 - Esterification
This reaction termed Fischer esterification in honor of its discoverer |
|
Improved Fischer Esterification of Substituted Benzoic Acid under
alcohols produced the highest yield of the ester product compared to secondary alcohols and tertiary alcohol resulted in the lowest. |
|
Experiment 14A: Isopentyl Acetate
14 oct. 2020 Theoretical Yield: 8.85 mmol. isopentyl acetate may be produced. ... focused on the synthesis of an ester by a Fischer esterification. |
|
Fischer Esterification of 3-ntrobenzoic acid 2017
Fischer Esterification of 3-nitrobenzoic acid to Produce Methyl 3- To do so you can use the percent yields for each step to compute the overall. |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
This reaction termed Fischer esterification in honor of its discoverer |
|
Fischer Esterification
Loss of water yields a carbocation stabilized by resonance which need only lose a proton to give the desired ester |
|
Synthesis and Investigation of Thermal Properties of Highly Pure
30 juin 2018 Carboxylic esters can be obtained via Fischer esterification of carboxylic ... decreasing and ester yield is increased for increased heating ... |
|
EXPERIMENT 5 ORGANIC SYNTHESIS: FISCHER
In this experiment you will synthesize the ester n-butyl acetate (bp 126.5°C) |
|
Investigating Esters
It is difficult to achieve 100% conversion and the yield of the ester Alcohols Organic Acids |
|
Multistep Synthesis Ester step
Step 3: Fischer Esterification of m-Nitrobenzoic Acid to Produce Methyl m- To do so you can use the percent yields for each step to compute the overall. |
|
Chemistry 209 Expt 5 - Esterification - Winona State University
This reaction, termed Fischer esterification in honor of its discoverer, can be used to product obtained (i e the yield) will be measured and the percent yield |
|
Synthesis of an Ester: Fischer Esterification The ester is synthesised
The carboxylic acid is the limiting reagent Using acetic acid? ⇒ The alcohol is the limiting reagent Weigh compounds directly into the reaction flask Be careful |
|
Esters An Introduction
For esters in which the alcohol and carboxylic acid are sterically unhindered, a 1:1 mixture of alcohol and carboxylic acid will yield an equilibrium mixture that is about 70 ester This means that if ester were isolated from this mixture, at best a 70 yield would be obtained |
|
Fischer Esterification - MIT OpenCourseWare
EXPERIMENT #5 Fischer Esterification Fischer Esterification: The Organic Synthesis, Isolation, Purification, and and percent yield of product formed 6 |
|
Fischer Esterification - MIT OpenCourseWare
EXPERIMENT #5 Fischer Esterification Fischer Esterification: The Organic Synthesis, Isolation, Purification, and and percent yield of product formed 6 |
|
Fischer Esterification
Fischer Esterification Introduction: A typical procedure to synthesize esters is the Fischer esterification, wherein a carboxylic Calculate the percent yield 2 |
|
1 a The first reaction is an esterification leading to a methyl ester
Sulfuric acid is used as catalyst in the Fischer esterification It protonates In order to calculate the yield, first the limiting reagent has to be identified nA= 2 45 |
|
Theoretical Yield - Chemistry
Example 2 Consider the acid-catalyzed esterification of isoamyl alcohol to produce isoamyl acetate If you begin with 10 g of isoamyl alcohol, 5 mL of acetic acid, |
|
Experiment 18 — Synthesis of Esters
acid and an alcohol, a process known as Fischer esterification the quantities of reagents needed to make a theoretical yield of 2 0 g of ester To drive the |