why is alcohol used in excess in esterification
When primary alcohol is treated with a carboxylic acid in the presence of sulphuric acid a compound is formed.
This compound has a sweet smell.
The compound obtained is called ester.
The chemical reaction occurring in the formation of the ester is known as an esterification reaction.
Why is alcohol the limiting reagent in Fischer esterification?
The Fischer esterification uses sulphuric acid as a catalyst.
It protonates the carboxylic acid carbonyl group and not the feature of hydroxyl.
The resulting cation is a stable resonance.
Because the reaction is a 1:1 reaction (carboxylic acid: alcohol), the limiting reagents are the carboxylic acid.
Why is excess reagent used in esterification?
Because there is typically very little difference in stability between both esters, the equilibrium constant of this reaction is usually near one.
Using a large excess of the reactant alcohol or removing the alcohol side-product can push the reaction equilibrium towards the products by Le Chatelier's principal.
Why was excess acetic acid used in the reaction?
A large amount of excess acetic acid is used in the preparation of isopentyl acetate in order to shift the equilibrium towards the products and obtaining high yields of isopentyl acetate.
Also, acetic acid is cheaper as compared to isopentyl alcohol.
|
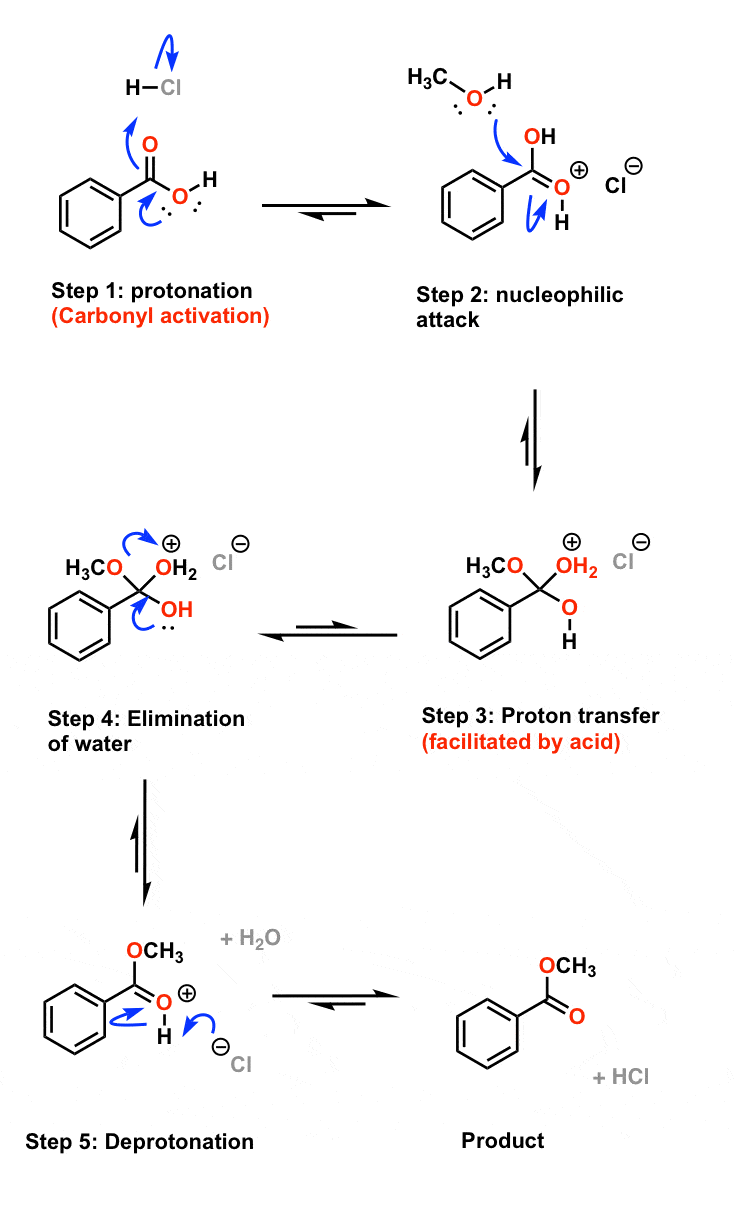
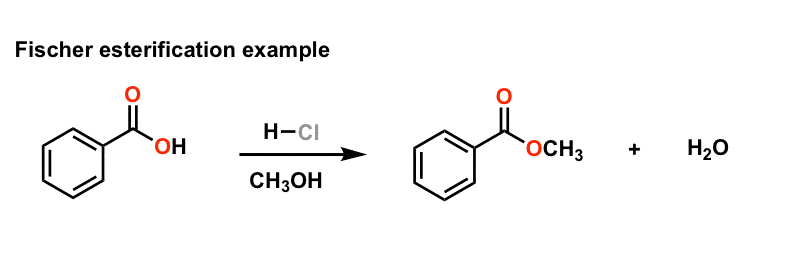
Fischer Esterification
To drive the equilibrium to make more ester excess alcohol is added ethanol used in the reaction mix |
|
Lab 12: Synthesis of an Ester
Esters can be prepared by this method in the presence of an acid catalyst. must be used in excess. R. O. O. R'. R. O. HO R'. OH carboxylic acid alcohol. |
|
The ester is synthesised via a Fischer esterification – the reaction of
Synthesis of an Ester: Fischer Esterification Using butyric acid? ... Namely excess alcohol (assuming it is water soluble) and/or excess carboxylic ... |
|
Simplified Method to Optimize Enzymatic Esters Syntheses in
12 nov. 2021 and kinetics aspects of enzymatic esterification in solvent-free ... of utilized masses of alcohol (even using an excess of alcohol) and ... |
|
Esters. An Introduction.
For esters in which the alcohol and carboxylic acid are sterically unhindered the equilibrium towards product |
|
Esterification of Levulinic Acid to Levulinate Esters in the Presence
of LA with alkyl alcohol produces levulinate ester which can be applied as fragrance excess ethanol from the esterification of LA as well as to. |
|
Sustainable Biocatalytic Procedure for Obtaining New Branched
13 nov. 2021 established: 70 ?C with a 10% excess of alcohol which requires a longer ... Esters are widely used compounds with a great diversity of ... |
|
Microwave-Assisted Esterification: A Discovery-Based Microscale
17 juil. 2014 esterification have capitalized on using ester fragrances to enhance learning. ... procedures A and B used either excess alcohol or excess. |
|
Synthesis of Isopentyl Acetate.pdf
Esterification generally refers to the formation of esters from alcohol and equilibrium reaction might be increased either by using an excess of one of ... |
|
Synthesis of ??-unsaturated esters of perfluoropolyalkylethers
a fast and eco-friendly process; using UV-light at room recent procedures used only a slight excess of the carboxylic acid or the alcohol with a high. |
|
Esters An Introduction
For esters in which the alcohol and carboxylic acid are sterically unhindered, a 1: 1 the equilibrium towards product, whereas using an excess of one of the |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
In this experiment you will react a carboxylic acid and an alcohol under acidic Esters can be prepared by this method in the presence of an acid catalyst to the right (in favor of the ester), one of the starting materials must be used in excess |
|
Synthesis of an Ester: Fischer Esterification The ester is synthesised
In order to shift the equilibrium position to the right, favouring the ester product, a two-fold excess of either the carboxylic acid or alcohol is used Page 3 Reaction |
|
Kinetic Study of Esterification Reaction - CORE
Keywords: Esterification, ethyl acetate, acetic acid, kinetics, non-ideal systems 1 Introduction therefore the use of excess ethanol (M=30 and 50) decreases |
|
Esters - NJgov
excessive loss of alcohol by overheating -0 3 Use of a primary alcohol rather than a secondary if possible For example, the change from secondary butyl |
|
Study of the Influence of Alcohols in the Synthesis of Short - at NTNU
synthesis of short chain esters by enzymatic catalysis in n-hexane First, the effect of the esterification extent To impel reaction yield, excess alcohol was used |


















![Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry](https://upload.wikimedia.org/wikipedia/commons/thumb/d/d7/Fischer_esterification_mechanism.svg/518px-Fischer_esterification_mechanism.svg.png)




