apes chapter 15 air pollution and stratospheric ozone depletion
|
APES CHAPTER 15 NOTES (MRS BAUCK): AIR POLLUTION AND
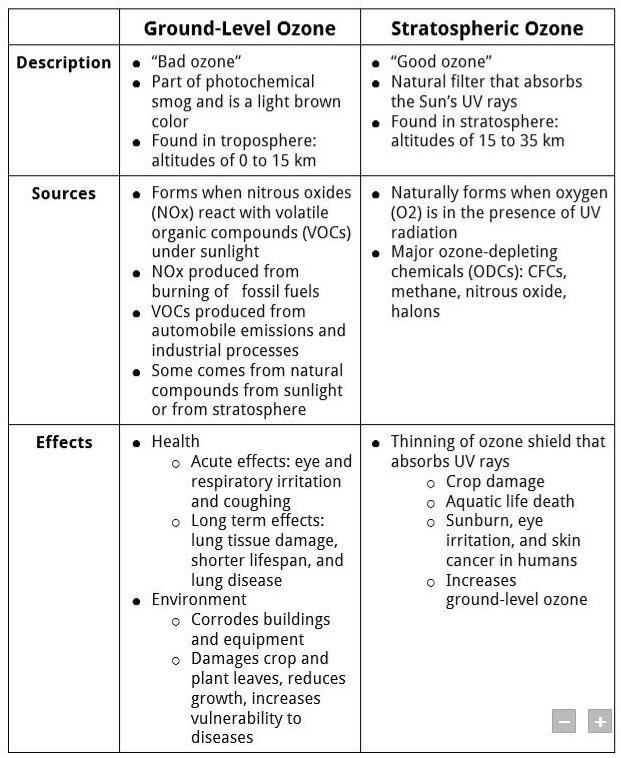
1) “good ozone” = stratospheric ozone 2) “bad ozone” = tropospheric (ground-level) ozone B sources 1) chemicals from cars power plants industrial boilers refineries chemical plants etc reacting with sunlight 2) O 3 pollution is 1) |
|
Chapter 15 Air Pollution and Stratospheric Ozone Depletion
Chapter 15—Air Pollution and Stratospheric Ozone Depletion Cleaning Up in Chattanooga (p 517-8) Module 46—Major Air Pollutants and Their Sources After reading this module you should be able to identify and describe the major air pollutants describe the sources of air pollution Air Pollution is a global system What does that mean? |
|
Chapter 15 Air Pollution and Stratospheric Ozone Depletion
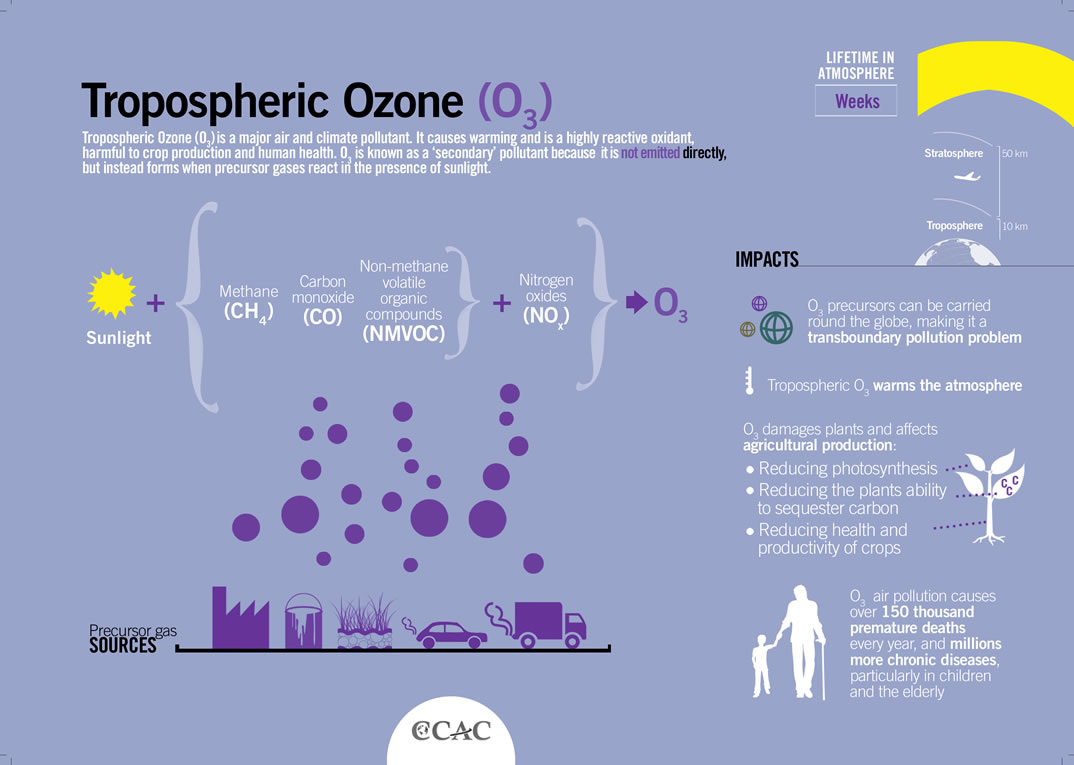
What are the major air pollutants? • Sulfur dioxide nitrogen oxides carbon oxides particulate matter lead VOC mercury and ground-level ozone |
|
Chapter 15—Air Pollution and Stratospheric Ozone Depletion
Chapter 15—Air Pollution and Stratospheric Ozone Depletion—Page 1 APES Name Chapter 15—Air Pollution and Stratospheric Ozone Depletion |
What are the primary pollutants of stratospheric ozone depletion?
See hydrochlorofluorocarbons, hydrofluorocarbons, perfluorocarbons, ozone depleting substance. (CFCs), and Class II ODS, such as hydrochlorofluorocarbons.
Although ozone depleting substances, they are less potent at destroying stratospheric ozone than chlorofluorocarbons (CFCs).chlorofluorocarbons (CFCs) Organic compounds made up of atoms of carbon, chlorine, and fluorine.
An example is Freon-12 (CCl2F2), used as a refrigerant in refrigerators and air conditioners and in making plastics such as Styrofoam.
What is the stratospheric ozone depletion?
Ozone Depletion.
When chlorine and bromine atoms come into contact with ozone in the stratosphere, they destroy ozone molecules.
One chlorine atom can destroy over 100,000 ozone molecules before it is removed from the stratosphere.
Ozone can be destroyed more quickly than it is naturally created.
What are the causes of ozone depletion apes?
Ozone Depletion with Chlorofluorocarbons (CFCs)
One molecule of Chlorofluorocarbon can continuously bond with and destroy ozone .
A chlorofluorocarbon is hit by electromagnetic radiation causing it to lose a chlorine atom.
|
Chapter 15 Air Pollution and Stratospheric Ozone Depletion
Chapter 15 Air Pollution and Stratospheric Ozone Depletion Friedland and Relyea Environmental Science for AP®, second edition © 2015 W H Freeman and |
|
Chapter 15—Air Pollution and Stratospheric Ozone Depletion
Chapter 15—Air Pollution and Stratospheric Ozone Depletion Cleaning Up in Chattanooga (p 517-8) Module 46—Major Air Pollutants and Their Sources |
|
APES Chapter 15 - misdorg
Air Pollution and Stratospheric Ozone Depletion Chapter 15 The main objectives of this chapter is to understand the major indoor and outdoor air pollutants |
|
APES Study Guide Chapter 15 Air Pollution and Stratospheric
APES Study Guide Chapter 15 Air Pollution and Stratospheric Ozone Depletion Vocab: • air pollution • volatile organic compounds (VOCs) • secondary |
|
AIR POLLUTION AND STRATOSPHERIC OZONE DEPLETION
APES CHAPTER 15 NOTES (MRS BAUCK): AIR POLLUTION AND STRATOSPHERIC OZONE DEPLETION [NOTE: The abbreviations NOx and SOx are often |
|
Lecture Outline
This chapter will help you: Describe the composition Identify major pollutants, outline the scope of outdoor air pollution, and assess potential solutions Explain stratospheric ozone depletion and identify steps taken to address it Define acidic |
|
POLLUTION UNIT - CHAPTER 16: AIR POLLUTION
AP ENVIRONMENTAL SCIENCE POLLUTION UNIT - CHAPTER 16: AIR POLLUTION Stratospheric ozone is a protective shield against UV radiation Give examples of harmful effects due to ozone depletion: UV radiation can damage |
|
Ch 21 Notes: Atmospheric Pollution 211 Notes I Air Pollution
2) air pollutants—gases, aerosols, and particulates with harmful effects 3) atmospheric the Clean Air Act: ground-level ozone, particulate matter, carbon monoxide, sulfur dioxide and nitrogen dioxide c) depleting soil nutrients d) damaging |
|
Chapter 18: Air Pollution - South Miami Senior High School
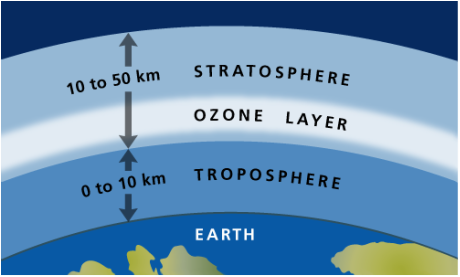
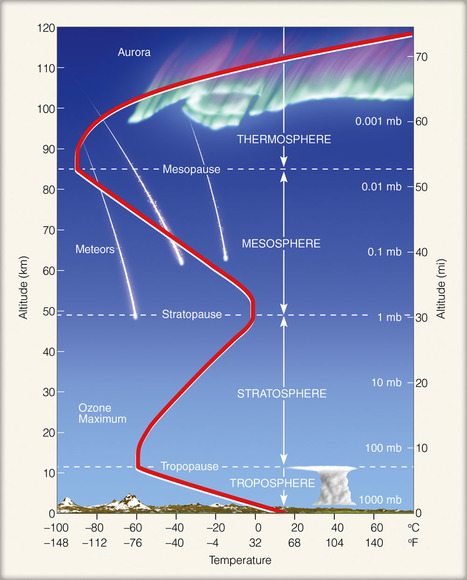
Concept 18-1 The two innermost layers of the atmosphere are the troposphere, which supports life, and the stratosphere, which contains the protective ozone layer |
|
Chapter 21 AIR POLLUTION - AP Environmental Science
Chapter 21 AIR Air pollutants are either from mobile or stationary sources Ozone (O3) and other photochemical oxidants result from atmospheric stratosphere, where it forms a protective layer that blocks much of the UV radiation |