api supplier
|
SUPPLIER QUALIFICATION & MANAGEMENT GUIDELINE
Chapter 1: Supplier Selection Chapter 2: Due Diligence Chapter 3: Quality Assessment Chapter 4: Change Control + Production Assessment Chapter 5: Supply Chain Security Chapter 6: Ongoing Monitoring and Evaluation Appendices INTRODUCTION |
|
Quality Agreement Guideline Template
A Quality Agreement is a major element of an API/intermediate user’s supplier qualification program but of course it is not a substitute for the supplier qualification processes including audits as necessary and for understanding the supplier processes and capabilities |
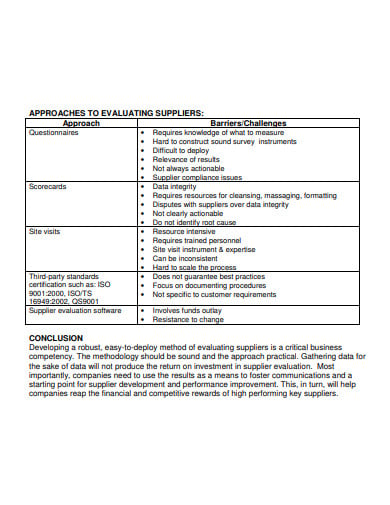
Why is supplier selection important in API manufacturing?
Still the majority firms don’t have their own API manufacturing facility. Since the finished product cost is majorily driven by API, the supplier selection plays prominent role in the generic profitability as well as quality. The supplier screening and selection includes extensive evaluation and comparison of documents, quality and cost.
Are API suppliers a key target for pharmaceutical companies?
API suppliers that can prove the security of their supply chains from raw materials through to ingredient, are likely to see considerable demand from pharmaceutical firms. Growth of the generic sector in major markets, particularly the US and Europe, will cement their position as key targets for API manufacturers.
What is the global API supplier base?
The current global API supplier base consists of hubs in which manufacturers specialize in producing different types of ingredients for separate sections of the pharmaceutical market. China, for example, has a reputation for the production of low cost, high volume ingredients. And it is a major global source for the drug industry.
What should a medicinal product manufacturer look for in an API audit?
As part of this audit, the medicinal product manufacturer should ensure that the API supplier / manufacturer also has supplier qualification procedures in place for their suppliers of critical and non-critical raw materials, API Starting Materials, Registered Intermediates and APIs (in the case of contract manufacturers).
|
API Supplier Change or Addition of Alterate API Supplier in Generic
5 mai 2015 The applicant has to go through regulatory process for supplier changes. API manufacturers facing inspection failures owing to GMP (good. |
|
Guidance on Amendments to an Active Pharmaceutical Ingredient
18 oct. 2019 API manufacturers are encouraged to announce all changes to their recipient FPP manufacturers. For manufacturers of a prequalified API ... |
|
Supplier Data API with Pagination
Only applications approved by the. SAP Ariba APIs administration can execute OAuth authentication. Retrieving supplier certificate attachments and approving |
|
Risk Category Information API for Supplier Risk Exposure
This API includes the following custom field data endpoints: PATCH /suppliers Imports values specified in the query body into the custom fields configured in |
|
INSPECTION OF ACTIVE PHARMACEUTICAL INGREDIENTS
13 jan. 2009 and/or sections of GMP PE 009 (Part II) which are specific to the inspection of. API manufacturers or critical for the quality of APIs. |
|
Purchase Orders Supplier API
This guide is for SAP Ariba suppliers who want to use the Ariba Network Purchase Orders API to extract purchase order information. The Purchase Orders Supplier |
|
Alternate Source of the Active Pharmaceutical Ingredient in Pending
good manufacturing practice (CGMP) inspection for the primary API supplier the application would not be approved until the CGMP issues were resolved. |
|
Supplier Invite API
The Supplier Invite API allows you to create vendor records in Ariba Network. This guide applies to: ? Ariba Network. ? SAP Ariba developer portal. |
|
Additional guidance for manufacturers
27 fév. 2018 Retest/shelf life parameters should be indicated in the API specification of the FPP manufacturer. Control of related substances in APIs and ... |
|
Additional guidance for manufacturers
27 fév. 2018 The Active Pharmaceutical Ingredient (API) supplier's vs the FPP manufacturer's API specifications. Most of the reviewed dossiers had issues ... |
|
API Supplier Change or Addition of Alterate API Supplier in Generic
5 mai 2015 · The applicant has to go through regulatory process for supplier changes API manufacturers facing inspection failures owing to GMP (good |
|
Impact of API (Active Pharmaceutical Ingredient) Source Selection
USP [1] has published the manual for API supplier selection “USP pharmaceutical ingredient supplier qualification program” Generic drug product approval |
|
APIC QUICK GUIDE FOR API SOURCING - APIC (CEFIC)
30 sept 2008 · The document does not address in detail the vendor qualification process as it is taken for granted that APIs are only purchased from suppliers |
|
Global API industry snapshot Pharma demands and evolving - CPhI
The current global API supplier base consists of hubs in which manufacturers specialize in producing different types of ingredients for separate sections |
|
Q 7 Good Manufacturing Practice for Active Pharmaceutical
(GMP) for the manufacturing of active pharmaceutical ingredients (APIs) under an appropriate Approving intermediate and API contract manufacturers; 9 |
|
Annex 4 Guidelines on active pharmaceutical ingredient master file
prequalification of medicinal products and the manufacturers of their APIs this case, the API manufacturer should provide a signed declaration that |
|
Active Pharmaceuticals Ingredients Suppliers role to - Diva Portal
The rise of antibiotic resistance and the difficulty in the procurement of active pharmaceutical ingredients (APIs) used in the manufacturing reduce the availability |
|
Q7A: Questions and Answers - CMC Drug Product Development
Section 7 1 states "Manufacturers of intermediates and/or APIs should have a system for evaluating the suppliers of critical materials " The API Starting Material |




















![Download] EBook~PDF Supplier Relationship Management: Unlocking Download] EBook~PDF Supplier Relationship Management: Unlocking](https://www.formsbirds.com/formimg/supplier-evaluation-form/455/vendor-performance-evaluation-form-d1.png)