aqa reaction pathways
|
OCR A Level Chemistry A
Synthetic routes (A Level) - reaction pathways Aliphatic compounds NaCN(aq)/ H+(aq) H2/Ni carboxylic Synthetic routes (A Level) - reaction pathways |
|
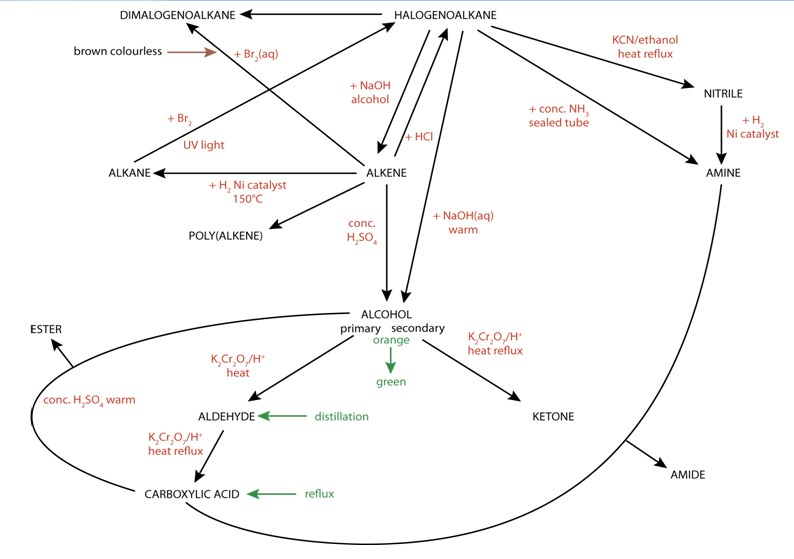
SUMMARY OF REACTION PATHWAYS IN ORGANIC CHEMISTRY
You need to know the mechanism for some of them 1 Alkane → chloroalkane reagents: Cl2 conditions: UV light mechanism: free radical substitution equation |
|
314-revision-guide-organic-synthesis-aqapdf
Synthetic Routes Chemists aim to design processes that do not require a solvent and Reaction: aldehydes only are oxidised by Fehling's Solution into a |
|
Mechanism Summary for A-level AQA Chemistry :
The amine formed in the first reaction has a lone pair of electrons on the nitrogen and will react further with the haloalkane |
Reaction Conditions The environmental conditions, such as temperature, pressure, catalysts & solvent, under which a reaction progresses optimally.
Catalysts are substances that accelerate the rate ( velocity ) of a chemical reaction without themselves being consumed or appearing as part of the reaction product.
How do you memorize synthetic routes?
How do you remember synthetic routes? The easiest way to map synthetic routes on your own is to remember the two synthetic route overview diagrams given in this article (one for aliphatic compounds and the other from aromatic compounds), and construct the route for any given starting and target compound from there.
What are the 4 main types of organic reactions?
The four main reaction classes are additions, eliminations, substitutions, and rearrangements.
In an addition reaction the number of σ-bonds in the substrate molecule increases, usually at the expense of one or more π-bonds.
What are the reaction pathways?
Reaction mechanism, or reaction pathway, describes the successive steps at the molecular level that take place in a chemical reaction.
In each step, molecular bonds are either created or broken.
|
Aqa-mechanisms-a-level-summary.pdf
Mechanism Summary for A-level AQA Chemistry The amine formed in the first reaction has a lone pair of electrons on the ... Aromatic synthetic routes. |
|
SUMMARY OF REACTION PATHWAYS IN ORGANIC CHEMISTRY
You need to know the equations conditions |
|
Question paper (A-level) : Paper 2 Organic and physical chemistry
12 Jun 2018 This question is about the reactions of alkanes. 0 1 . 1 Alkanes can be used as fuels. ... A reaction pathway is shown. CH2(OH)CH(CH3)CH2Br. |
|
Detailed Notes - Topic 3.14. Organic Synthesis - AQA Chemistry A
3 Mar 2014 Synthesis pathways are needed to convert starting materials into a ... will only react with one enantiomer the synthetic pathway should be. |
|
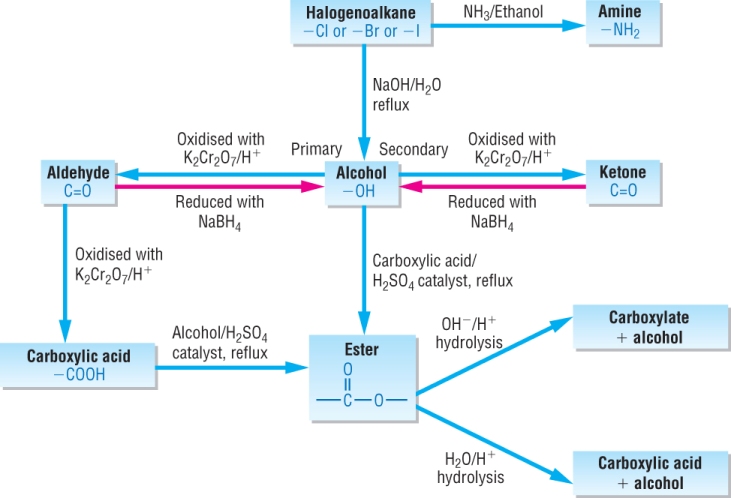
Synthetic routes (A Level) - reaction pathways Aliphatic compounds
OCR 2017. Version 1. Haloalkane. Alcohol. 1°. 2°. Nitrile. Amine. Alkene. Aldehyde. Ketone. Carboxylic acid. Acyl chloride. 1o Amide. 2o Amide. |
|
GCSE Combined Science: Trilogy Specification Specification for first
So you can be confident that our GCSE Combined Science: Trilogy is interesting and relevant pathway for the reaction that has a lower activation energy. |
|
Detailed Notes - Topic 3.14. Organic Synthesis - AQA Chemistry A
3 Mar 2014 Synthesis pathways are needed to convert starting materials into a ... will only react with one enantiomer the synthetic pathway should be. |
|
Paper 1 The human body and movement - June 2018 - Mark scheme
MARK SCHEME – GCSE PHYSICAL EDUCATION – 8582/1 – JUNE 2018. 3. Level of response marking instructions. Level of response mark schemes are broken down into |
|
Gcse chemistry 8462/2h
2 Jun 2018 expanded on the basis of students' reactions to a particular paper. ... 4.6.2.4. 4.6.2.7. 09.5 provides different reaction pathway. |
|
Practical handbook: GCSE Biology
measurement of rate of reaction by a variety of methods including an uptake of may be possible using micro-habitats such as trees pathways |
|
Mechanism Summary for A-level AQA Chemistry : - chemrevise
Mechanism Summary for A-level AQA Chemistry The amine formed in the first reaction has a lone pair of electrons on the nitrogen Aromatic synthetic routes |
|
SUMMARY OF REACTION PATHWAYS IN ORGANIC CHEMISTRY
You need to know the equations, conditions, reagents and type of reaction for all these reactions You need to know the mechanism for some of them 1 |
|
THE SCIENCE DEPARTMENT Year 10 GCSE - Emmanuel College
calculate the atom economy of a reaction to form a desired product from the balanced equation (HT only) explain why a particular reaction pathway is chosen to |
|
Organic synthesis reaction pathways - OCR
Synthetic routes (A Level) - reaction pathways Aliphatic compounds NaCN(aq)/ H+(aq) H2/Ni carboxylic acid/ conc H2SO4 OR acid anhydride K2Cr2O7/ |
|
AQA Chemistry (Triple) Specification Checklists - Isaac Newton
write formulae and balanced chemical equations for the reactions in this (HT only) explain why a particular reaction pathway is chosen to produce a specified |
|
AQA GCSE The rate and extent of chemical change Rate of reaction
Catalysts provide a different reaction pathway where reactants do not require as much energy to react when they collide If a catalyst is used in a reaction, it is not |
|
Personalised Learning Checklists AQA Chemistry - Devizes School
AQA Chemistry (8462) from 2016 Topics C4 1 Atomic structure and the periodic Chem HT ONLY: Explain why a particular reaction pathway is chosen to |

![Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry](https://d3i71xaburhd42.cloudfront.net/31568eb203993b891059af015fe265f6e0eeadc3/4-Figure2-1.png)