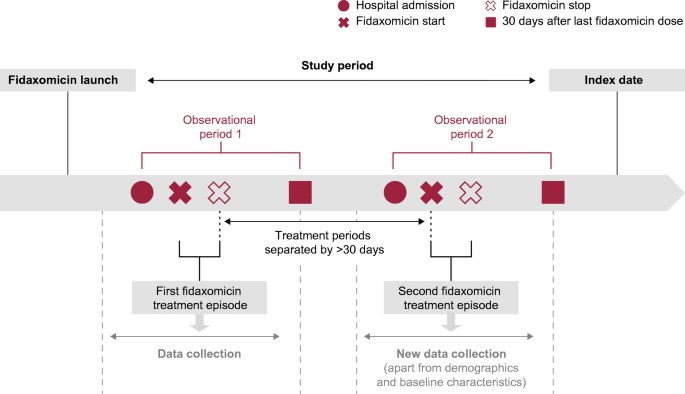
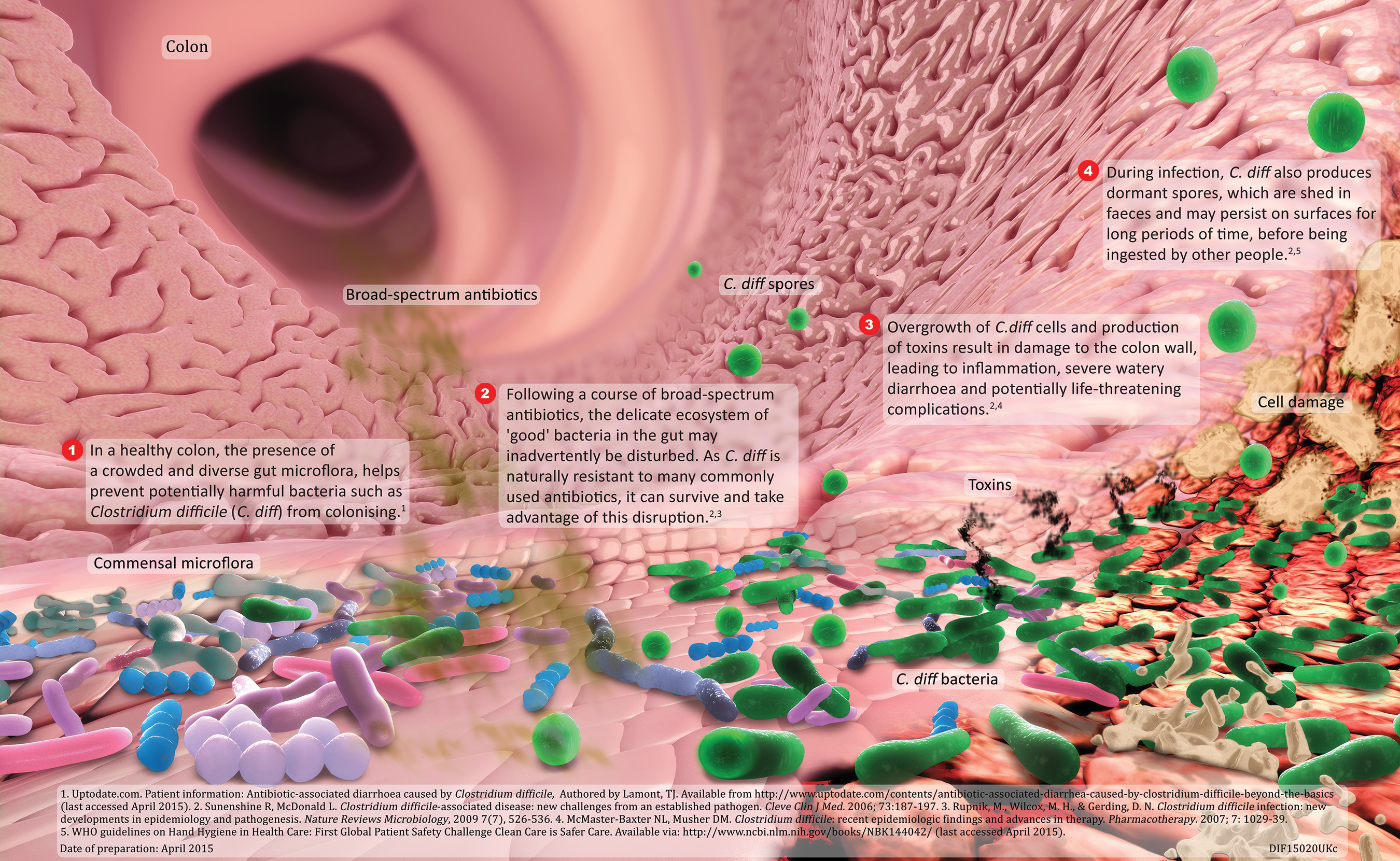
Dificlir, INN-fidaxomicin - Europa EU
|
Dificlir; INN: fidaxomicin
22-Sept-2011 E-mail info@ema.europa.eu Website www.ema.europa.eu ... Dificlir (fidaxomicin) is indicated for the treatment of Clostridium difficile ... |
|
Dificlir INN-fidaxomicin
05-Dec-2011 Detailed information on this medicinal product is available on the website of the European Medicines. Agency http://www.ema.europa.eu. Page 10 ... |
|
Dificlir INN-fidaxomicin
European Medicines Agency 2020. Reproduction is authorised provided the source is acknowledged. EMA/31758/2020. EMEA/H/C/2087. Dificlir (fidaxomicin). |
|
Dificlir INN-fidaxomicin
12-Dec-2019 Dificlir. International non-proprietary name: fidaxomicin. Procedure No. EMEA/H/C/002087/X/0034/G. Note. Assessment report as adopted by the ... |
|
Dificlir INN-fidaxomicin
Hypersensitivity reactions including severe angioedema have been reported (see section 4.8). If a severe allergic reaction occurs during treatment with |
|
Dificlir INN-fidaxomicin
05-Dec-2011 La información detallada de este medicamento está disponible en la página web de la Agencia. Europea de Medicamentos http://www.ema.europa.eu. |
|
Dificlir INN-fidaxomicin
European Medicines Agency 2020. Reproduction is authorised provided the source is acknowledged. EMA/31758/2020. EMEA/H/C/2087. Dificlir (fidassomi?in). |
|
Dificlir INN-fidaxomicin
Kull pillola miksija b'rita fiha 200 mg ta' fidaxomicin. DIFICLIR pilloli miksija b'rita hu indikat g?all-kura ta' infezzjonijiet minn Clostridioides ... |
|
Dificlir INN-fidaxomicin
Další informace o p?ípravku Dificlir jsou k dispozici na internetových stránkách agentury na adrese ema.europa.eu/medicines/human/EPAR/dificlir. Tento p?ehled |
|
Dificlir INN-fidaxomicin
05-Dec-2011 Ausführliche Informationen zu diesem Arzneimittel sind auf den Internetseiten der Europäischen. Arzneimittel-Agentur http://www.ema.europa.eu ... |
|
Dificlir, INN-fidaxomicin - europaeu
DIFICLIR 200 mg, comprimé pelliculé 2 COMPOSITION QUALITATIVE ET QUANTITATIVE Chaque comprimé pelliculé contient 200 mg de fidaxomicine |
|
Dificlir, INN-fidaxomicin - European Medicines Agency - europaeu
Dificlir contient la substance active fidaxomicine Comment Dificlir est-il utilisé? Dificlir est disponible sous forme de comprimés (200 mg) ou de granules pour |
|
Dificlir; INN: fidaxomicin
Dificlir fidaxomicin This is a summary of the European public assessment report Dificlir is used to treat adults with infections of the gut caused by bacteria |
|
Dificlir, INN-fidaxomicin - Astellas Pharma
13 fév 2020 · DIFICLIR filmomhulde tabletten is geïndiceerd voor de behandeling van Clostridioides difficile- DIFICLIR 40 mg/ml granulaat voor orale suspensie kan worden Geneesmiddelenbureau (EMA) http://www ema europa eu |
|
Mise à jour du guide dusage optimal pour le - INESSS: WWW
infections Despite the fact that fidaxomicin has been shown to be equivalent to vancomycin in et en Europe depuis le début des années 2000 [ASPC, 2014a] https://www ahrq gov/downloads/pub/evidence/ pdf /probiotics/probiotics pdf |
|
Fidaxomcin publisher finalpub - Welcome to the St Georges Hospital
Fidaxomicin was approved in the EU in December 2011 for the The cost of Dificlir will be £135 per day, making the cost of a 10 day course £1,350 compared |