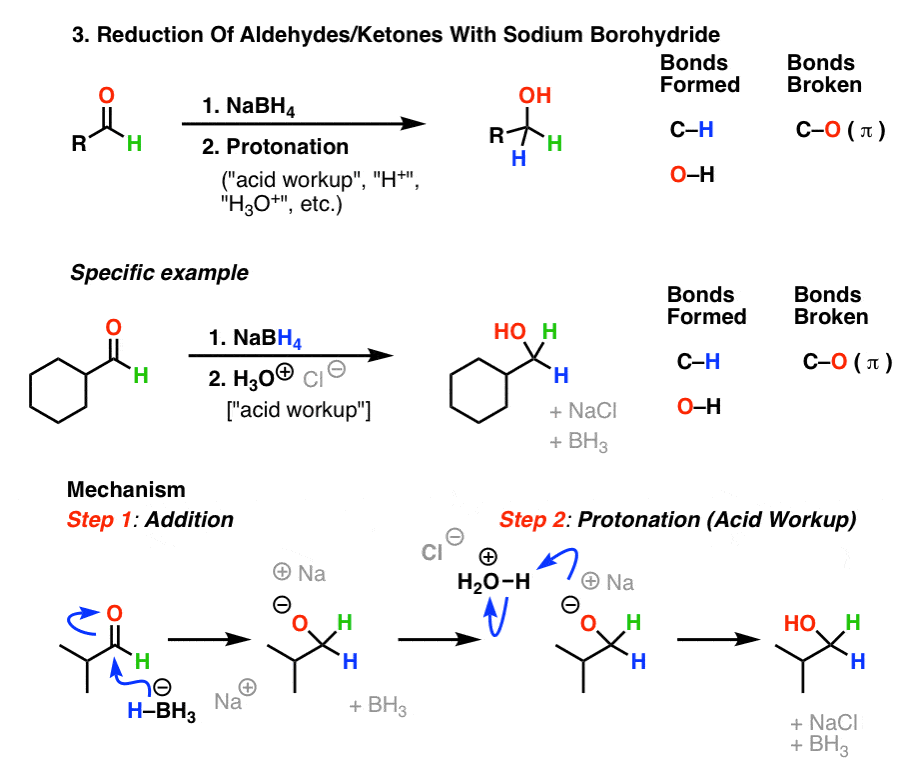
assignment on aldehydes and ketones
What are aldehydes & ketones?
Aldehydes and ketones undergo a variety of reactions that lead to many different products. The most common reactions are nucleophilic addition reactions, which lead to the formation of alcohols, alkenes, diols, cyanohydrins (RCH (OH)C&tbond;N), and imines R 2 C&dbond;NR), to mention a few representative examples.
Why do aldehydes and ketones have higher boiling points?
The polar carbon-to-oxygen double bond causes aldehydes and ketones to have higher boiling points than those of ethers and alkanes of similar molar masses but lower than those of comparable alcohols that engage in intermolecular hydrogen bonding. Aldehydes are readily oxidized to carboxylic acids, whereas ketones resist oxidation. 1.
What happens when aldehydes and ketones react with ammonia?
The reaction of aldehydes and ketones with ammonia or 1º-amines forms imine derivatives, also known as Schiff bases, (compounds having a C=N function). This reaction plays an important role in the synthesis of 2º-amines, as discussed earlier.
Does water add to the carbonyl function of aldehydes and ketones?
It has been demonstrated (above) that water adds rapidly to the carbonyl function of aldehydes and ketones. In most cases the resulting hydrate (a geminal-diol) is unstable relative to the reactants and cannot be isolated. Exceptions to this rule exist, one being formaldehyde (a gas in its pure monomeric state).
Nomenclature of Aldehydes and Ketones
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O. The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents. If at least one of these substituents is hydrogen, the compound is an aldehyde. If neither is hydrogen, the compound is a ketone. The IU
Occurrence of Aldehydes and Ketones
Aldehydes and ketones are widespread in nature, often combined with other functional groups. Example are shown in the following diagram. The compounds in the top row are found chiefly in plants or microorganisms; those in the bottom row have animal origins. With the exception of the first three compounds (top row) these molecular structures are all
Synthetic Preparation of Aldehydes and Ketones
Aldehydes and ketones are obtained as products from many reactions discussed in previous sections of this text. The following diagram summarizes the most important of these. To review the previous discussion of any of these reaction classes simply click on the number (1 to 5) or descriptive heading for the group. With the exception of Friedel-Craft
Properties of Aldehydes and Ketones
A comparison of the properties and reactivity of aldehydes and ketones with those of the alkenes is warranted, since both have a double bond functional group. Because of the greater electronegativity of oxygen, the carbonyl group is polar, and aldehydes and ketones have larger molecular dipole moments (D) than do alkenes. The resonance structures o
Reversible Addition Reactions
It has been demonstrated (above) that water adds rapidly to the carbonyl function of aldehydes and ketones. In most cases the resulting hydrate (a geminal-diol) is unstable relative to the reactants and cannot be isolated. Exceptions to this rule exist, one being formaldehyde (a gas in its pure monomeric state). Here the weaker pi-component of the
Irreversible Addition Reactions
The distinction between reversible and irreversible carbonyl addition reactions may be clarified by considering the stability of alcohols having the structure shown below in the shaded box. If substituent Y is not a hydrogen, an alkyl group or an aryl group, there is a good chance the compound will be unstable (not isolable), and will decompose in
Other Carbonyl Group Reactions
The metal hydride reductions and organometallic additions to aldehydes and ketones, described above, both decrease the carbonyl carbon's oxidation state, and may be classified as reductions. As noted, they proceed by attack of a strong nucleophilic species at the electrophilic carbon. Other useful reductions of carbonyl compounds, either to alcohol

Aldehydes and Ketones

Nomenclature of aldehydes and ketones Aldehydes and ketones Organic chemistry Khan Academy

An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
The carbonyl carbon of an aldehyde or ketone is sp2-hybridized. Carbonyl groups in aldehydes and ketones undergo addition reactions. |
|
Assignment Chapter 12: Aldehydes Ketones and Carboxylic Acids 1
Assignment. Chapter 12: Aldehydes Ketones and Carboxylic Acids b) Acetaldehyde |
|
Aldehydes and Ketones
this chapter the reactions of aldehydes and ketones are also cleverly 20.1 Assign a systematic (IUPAC) name to each of the following compounds:. |
|
SCH 206
(a) Assignments covering Aldehydes Ketones and Carboxylic. Acids |
|
INFRARED SPECTROSCOPY (IR)
IR SPECTRUM OF ALDEHYDES AND KETONES. Carbonyl compounds are those that contain the C=O functional group. In aldehydes this group is at the end of a carbon |
|
Ch18 Ketones and Aldehydes (landscape)
The simplest carbonyl compounds are aldehydes and ketones. A ketone has two alkyl (or aryl) groups bonded to the carbonyl carbon. |
|
Alkynes Synthesis of Aldehydes and Ketones
Draw a stepwise mechanism for the intramolecular. Friedel–Crafts acylation of compound A to form B. Assignment 7. |
|
How to Read and Interpret FTIR Spectroscope of Organic Material
Functional group/assignment. Wavenumber (cm-1) 1725–1700. Ketone. 1725–1705. Aldehyde. 1740–1725/(2800–2700)3) ... aldehydes ketones |
|
Aldehydes and Ketones are simple organic compounds containing a
Jun 13 2020 This reagent does not oxidize aldehydes to carboxylic acids. Dr. S.Hasan Mehdi. 6/13/2020. Aldehyde. 1. 0. |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
In aldehydes the carbonyl group is bonded to a carbon and hydrogen while in the ketones |
|
Assignment Aldehydes, Ketones and Carboxylic acids (To be done
Assignment Aldehydes, Ketones and Carboxylic acids (To be done in class register) Q1 Explain the following name reactions with suitable example: (i) |
|
Assignment Chapter 12: Aldehydes, Ketones and - Sanskriti School
Assignment Chapter 12: Aldehydes, Ketones and Carboxylic Acids 1 Write IUPAC names for the following : CH3 (a) = O (b) CH2=CHCH2CHO (c) (CH3) 2C= |
|
XII CHEMISTRY ASSIGNMENT Unit – 12 Aldehydes, Ketones and
CHEMISTRY ASSIGNMENT Unit – 12 Aldehydes, Ketones and Carboxylic Acids 1 What product is obtained when ethyl benzene is oxidized with alkaline |
|
BRAIN INTERNATIONAL SCHOOL Chemistry Assignment CLASS
Aldehyde ,ketone and carboxylic acids 1 Carbonyl compounds mainly show nucleophilic addition reactions Why? 2 Why it is necessary to control the pH |
|
Aldehydes , Ketones
Grignard reagent on reaction with acetone forms (a) Tertiary alcohol (b) Secondary alcohol (c) Acetic acid (d) Acetaldehyde ALDEHYDES , KETONES AND |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Aldehydes and ketones are not hydrogen bond donors (they can't donate a proton); therefore, they have lower boiling points than alcohols of similar molecular |
|
Fundamentals of Organic Chemistry CHAPTER 5 ALDEHYDES
27 jan 2019 · o Aldehydes and ketones are characterized by the presence of the carbonyl group -CH=O group is assigned the number 1 position |
|
Chap-13 Aldehydes Ketones and Carboxylic - SelfStudys
Solutions of Assignment (Level-II) Aldehydes, Ketones and Carboxylic Acids Aakash Educational Services Limited - Regd Office : Aakash Tower, 8, Pusa Road |