base catalysed hydrolysis of aspirin
|
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin Esters are susceptible to catalytic hydrolysis by both aqueous acids and bases The possible |
What is the base hydrolysis of aspirin?
Aspirin is a pain reliever and fever reducer, but if it's allowed to react with water then it can undergo hydrolysis, forming salicylic acid and acetic acid, which is no longer effective.
This reaction can occur under acidic or basic conditions.20 déc. 2017How does hydrolysis work with aspirin?
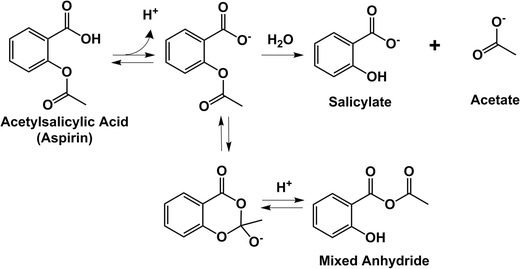
Aspirin (2-ethanoyloxybenzoic acid or acetylsalicylic acid) hydrolyses to produce 2-hydroxybenzoic acid and ethanoic acid.
Here is the equation for the reaction: The rate at which this reaction happens is important for two reasons.
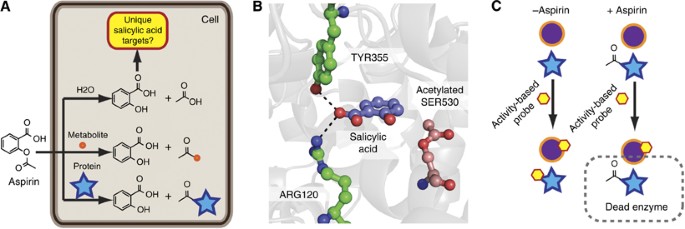
When administered, aspirin hydrolyses in the body.In this experience it is observed that the acetylsalicylic acid hydrolysis reaction is faster at acid pH and at 60 ° C.
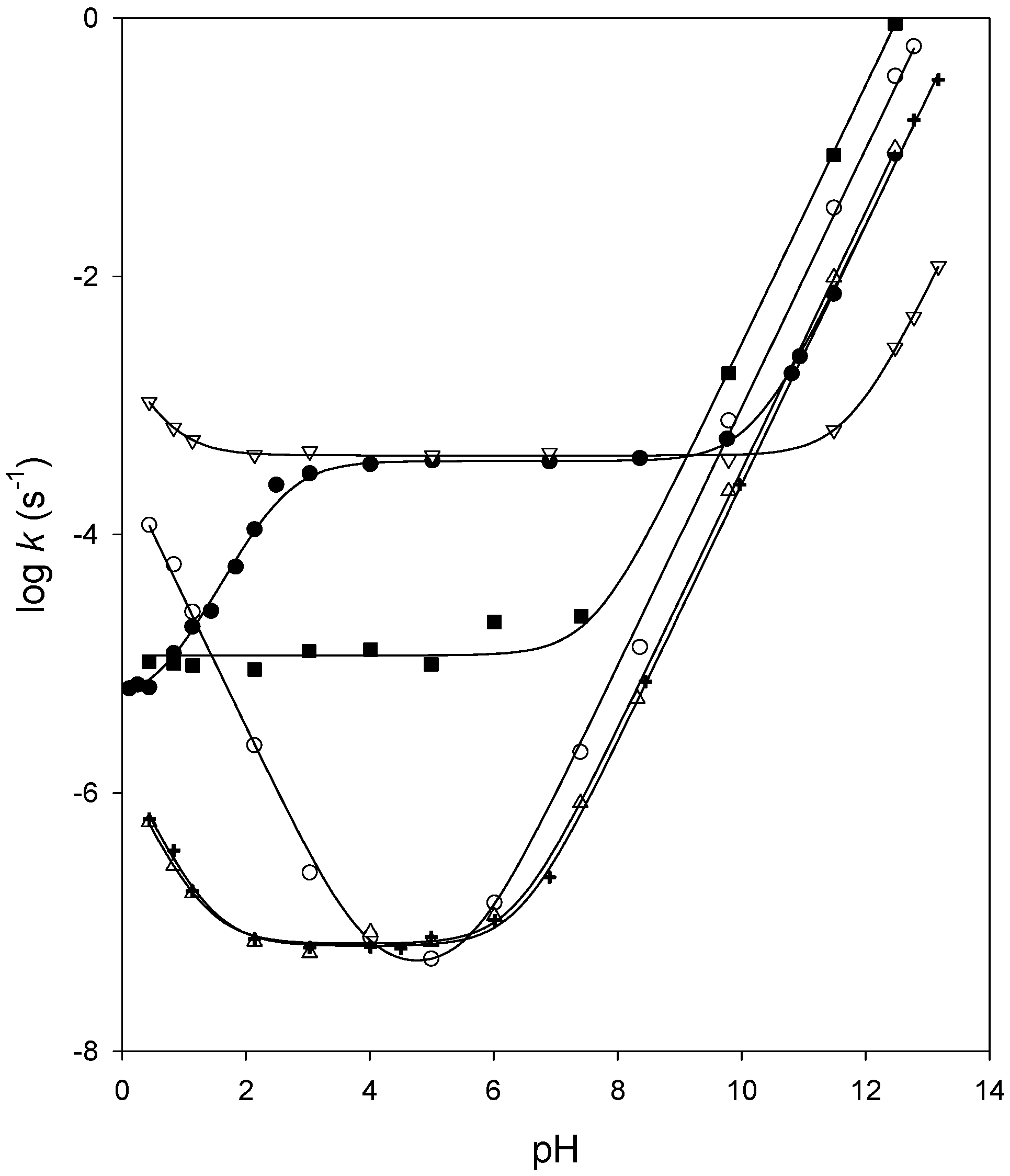
Since Edwards has shown that the rate of hydrolysis of aspirin is independent of pH between pH 4 and 8, the reaction has been the subject of a number of studies, particularly by Garrett [1,3-5].
|
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin
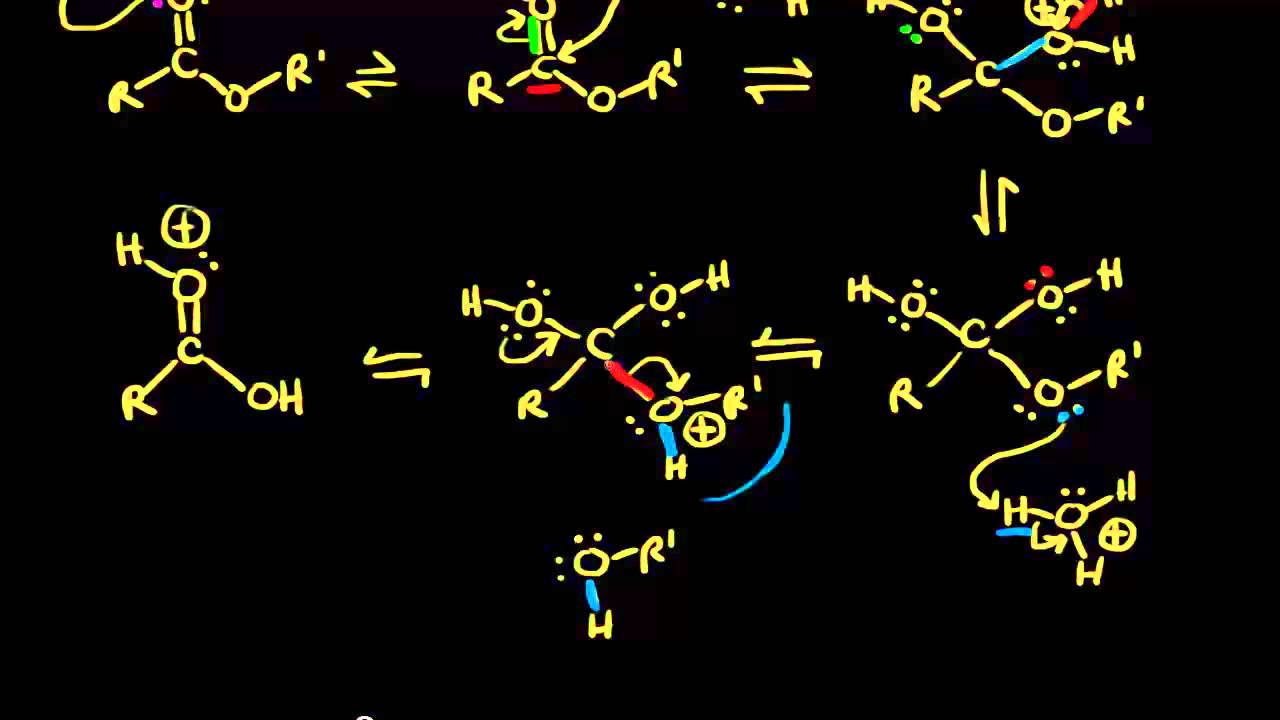
Analogous to the general acid catalysis the equation for the observed rate constant for general base catalysis includes a term for each conjugate base in the |
|
Hydrolysis of aspirin. Intramolecular general base catalysis of ester
corresponding reaction with phenyl acetate which is known to involve general base catalysis |
|
191 STUDY OF HYDROLYSIS OF ACETYLSALICYLIC ACID Aspirin
In this experience it is observed that the acetylsalicylic acid hydrolysis reaction is faster at acid pH and at 60 ° C. Key words: Acetylsalycilic acid |
|
Rate of hydrolysis of aspirin
Aspirin (2-ethanoyloxybenzoic acid or acetylsalicylic acid) hydrolyses to produce 2-hydroxybenzoic acid and ethanoic acid. Here is the equation for the reaction |
|
HYDROLYSIS 2016.pdf
Base catalysed elimination (E2 or E1cB) becomes important relative to neutral hydrolysis (SN2 SN1) as the degree of chlorination increases. |
|
Micellar Catalysis of Organic Reactions. VIII Kinetic Studies of the
chymotrypsin-catalysed hydrolysis) reactions catalysed by normal micelles do not General base catalysis of the hydrolysis of the ionized aspirin. |
|
Prediction of stability of drugs and pharmaceutical preparations
independent hydrolysis of the aspirin anion (50) rather than general acid-base catalysis by water sirice variations in acetic acid-acetate buffer. |
|
8-Synthesis-of-Aspirin.pdf
Aspirin can be made by reacting salicylic acid with acetic acid in the presence set up a ring stand and set a hot plate/magnetic stirrer on the base of. |
|
Hydrolysis as a Function of pH A. INTRODUCTION PURPOSE
12?/05?/1981 Hydrolysis refers to a reaction of a chemical RX with water ... in which case it is referred to as specific acid or specific base catalysis ... |
|
Experiment 5 - Synthesis of Aspirin
In the presence of moisture aspirin may decompose (hydrolysis) into salicylic acid and acetic acid. This reaction is the reverse of the synthesis reaction. |
|
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin
Analogous to the general acid catalysis, the equation for the observed rate constant for general base catalysis includes a term for each conjugate base in the reaction Aspirin I, acetylsalicylic acid, is an ester The equation for its hydrolysis to salicylic and acetic acids may be written very simply |
|
191 STUDY OF HYDROLYSIS OF ACETYLSALICYLIC ACID Aspirin
In this experience it is observed that the acetylsalicylic acid hydrolysis reaction is faster at acid pH and demonstration, showing that the hydrolysis reaction is not catalyzed by acetate ion, a In-tramolecular General Base Catalysis of Ester |
|
Rate of hydrolysis of aspirin
Aspirin (2-ethanoyloxybenzoic acid or acetylsalicylic acid) hydrolyses to produce 2-hydroxybenzoic acid and ethanoic acid Here is the equation for the reaction: |
|
Hydrolysis as a Function of pH - OECD
12 mai 1981 · Hydrolysis refers to a reaction of a chemical RX with water, with the net exchange of Aspirin – Diazinon These substances need not be employed in all cases when investigating a new possibility of acid or base catalysis |
|
REACTIONS OF SOME CARBOXYLIC ACID - Leicester Figshare
Base Catalysed Hydrolysis of Hydroxy-esters 182 214 227 Rearrangement The hydrolysis of acetylsalicylic acid was shown to be eighty times greater than |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
acid and base catalysis, general acid and base catalysis, nucleophilic catalysis, metal oxide surface catalysis and metal ion catalysis (1,2) Hydrolysis (reaction |
![PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin](https://file.scirp.org/Html/1-1230015/1576602b-d193-41de-bfda-c9ef70fa379e.jpg)


![PDF] Class Projects in Physical Organic Chemistry: The Hydrolysis PDF] Class Projects in Physical Organic Chemistry: The Hydrolysis](https://imgv2-2-f.scribdassets.com/img/document/434142396/298x396/37135d3fb8/1573305016?v\u003d1)


















![PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/attachment/b4afe119-dd2d-4434-b749-548bf9a123dc/gr1_lrg.jpg)



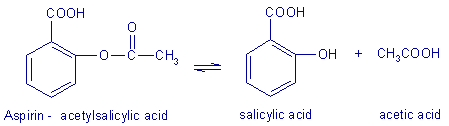



![PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/7a183eaf-8791-4fb7-b58d-fc2e2c02b441/mfor001.jpg)








![POSTLAB] Experiment 3 - Kinetics of Aspirin Hydrolysis - Google POSTLAB] Experiment 3 - Kinetics of Aspirin Hydrolysis - Google](https://file.scirp.org/Html/1-1230015/a5d7fbd9-768e-4753-bd6b-c815198a6192.jpg)
