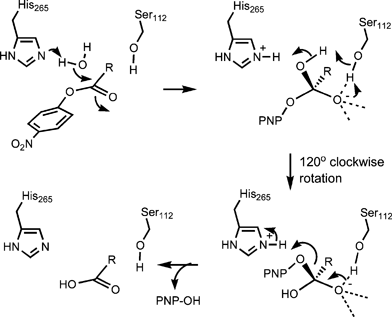
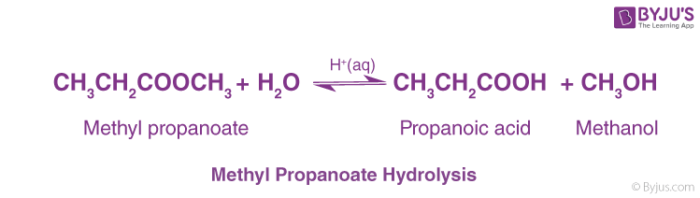
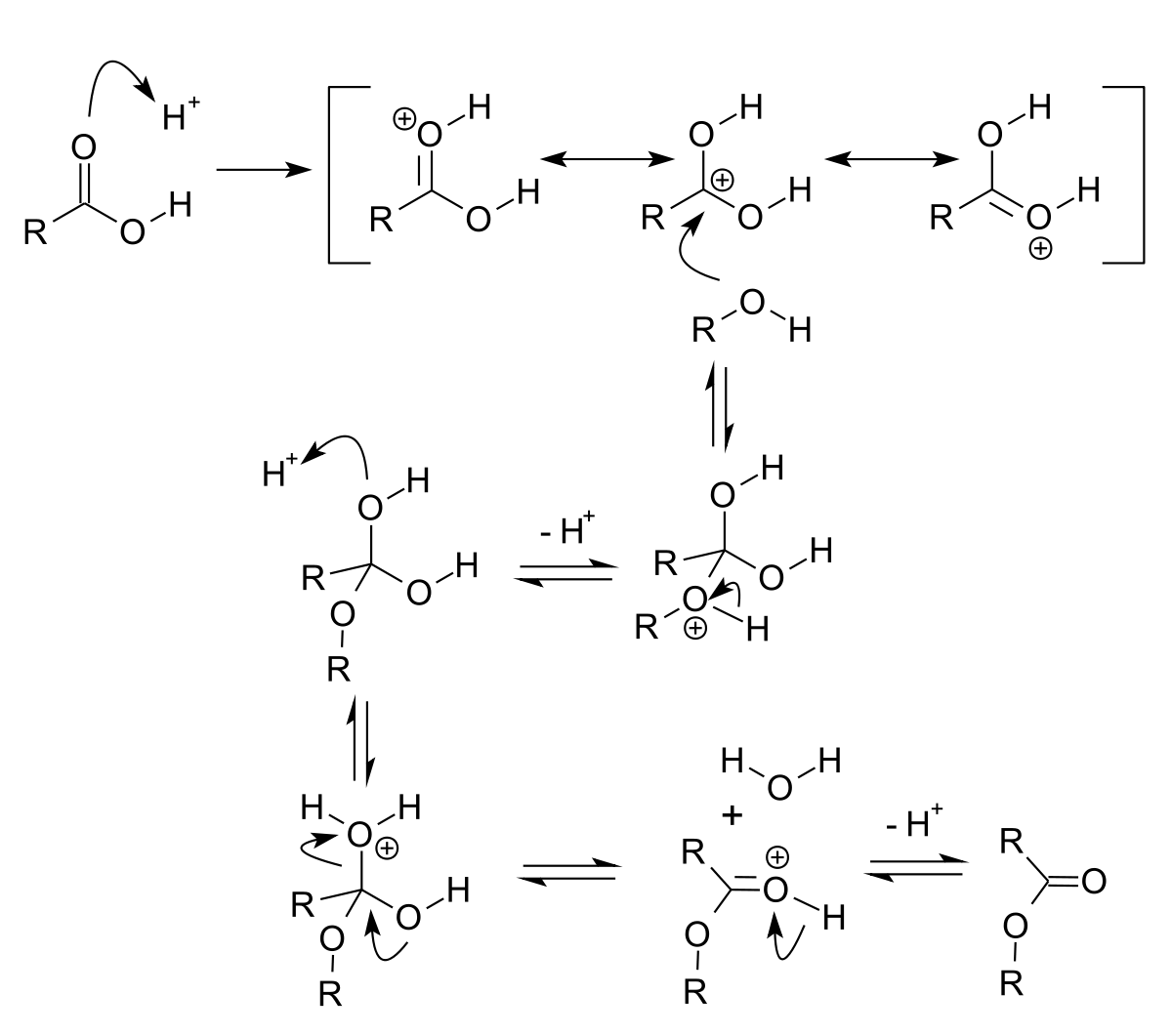
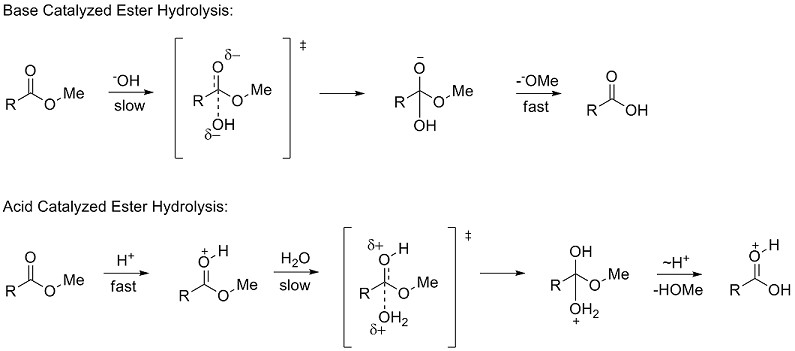
base catalysed hydrolysis of ester
One of the advantage of base catalysed hydrolysis is that it is unidirectional .
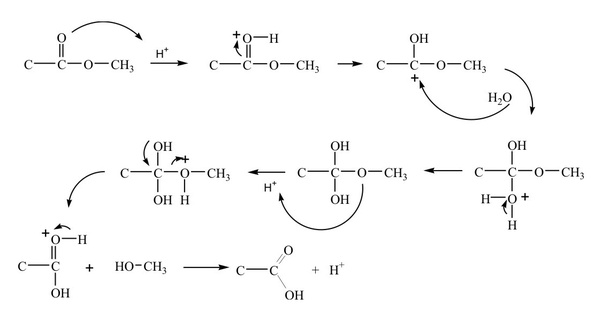
On the other hand acid catalysed hydrolysis of ester is reversible process.
What is the base hydrolysis of an ester?
Hydrolysis is a most important reaction of esters.
Acidic hydrolysis of an ester gives a carboxylic acid and an alcohol.
Basic hydrolysis of an ester gives a carboxylate salt and an alcohol.31 jan. 2022
What is the base hydrolysis?
And from there, we learned that basic hydrolysis is when water acts as an acid to break apart a weak base.
For example, water can react with urea, forming a hydroxide anion, which can then release ammonia from the urea, which is critical for the growth of plants.
What is hydrolysis of ester in basic catalyst?
Ester Hydrolysis with NaOH or base catalysed ester hydrolysis is the reaction of an ester with water under a basic medium.
In it, an ester is heated under reflux with dilute NaOH to yield carboxylate salt and alcohol.
|
The Kinetics of the Base-catalyzed Hydrolysis of the Methyl Esters of
in rate constants for acid-catalyzed esterification of the first andsecond carboxyl groups in the corresponding cyclo- hexanedicarboxylic acids. |
|
HYDROLYSIS 2016.pdf
electronegative than oxygen thiophosphate esters exhibit greater stability toward neutral and base catalyzed hydrolysis than the corresponding phosphate |
|
General Base Catalysis of Ester Hydrolysis1
shown that the hydrolysis of esters with a good leaving group such as the phenyl and thiol acetates |
|
A Facile Base-catalyzed Ester Hydrolysis Involving Alkyl-Oxygen
(4) base catalysis. Reaction 1 is of course the ordinary mechanism by which most esters undergo base-catalyzed hydrolysis.4 Anchimeric catalysis (2)has been. |
|
Acid/base-catalyzed ester hydrolysis in near-critical water
1 janv. 1999 Acid/base-catalyzed ester hydrolysis in near-critical water. Heather P. Lesutis Roger Gläser |
|
Sugar fatty acid ester surfactants: Base-catalyzed hydrolysis
kinetic study of base-catalyzed ester hydrolysis has revealed reasons for the inhibition of bacterial hydrolysis and provided. |
|
Intramolecular general-base-catalyzed hydrolysis and aminolysis of
A de- tailed discussion of the intramolecular reaction is provided. Intramolecular catalysis in the reactions of esters with nucleophiles continues to be |
|
Evidence for General Base Catalysis in an Ester Hydrolysis. II
Items 13 - 31 The protonated salicylate di-ester diethylaminoethyl acetylsalicylate hydrochloride (I) |
|
Concentration effects in the base-catalyzed hydrolysis of oligo
29 août 2016 [810] Bases may be used to adjust the pH of aqueous solutions containing water-soluble methacrylic esters. Amine-containing meth- acrylates ... |
|
Steric Effects. II. Base-Catalyzed Ester Hydrolysis - Marvin Charton
of carboxylic acids or the acid-catalyzed hydrolysis of their esters is of the same magnitude as the steric effect upon the base-catalyzed hydrolysis of the |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions form new esters by base- or acid-catalysed transesterification mechanisms; |
|
HYDROLYSIS
carbon centre, such as with carboxylic acid derivatives including esters, Epoxides undergo hydrolysis by neutral and acid catalyzed mechanisms under |
|
[2002] Intramolecular General Base Catalyzed Ester Hydrolysispdf
Hydrolysis of 2-Aminobenzoate Esters The 2-aminobenzoate esters hydrolyze with similar rate constants in the pH-independent reactions, and these water reactions are ∼2-fold slower in D2O than in H2O The most likely mechanism involves intramolecular general base catalysis by the neighboring amine group |
|
Sugar fatty acid ester surfactants: Base-catalyzed hydrolysis
A kinetic study of base-catalyzed ester hydrolysis has revealed reasons for the inhibition of bacterial hydrolysis and provided information regarding ester stability at |
|
Ester hydrolysis-lectute note
(11) Hydrolysis is bode - on acid-catalyzed li Hydrolysis and esterification may broceed either by a linimolecular of a bimolecular mechanism A Scanned with |
|
Mechanisms of Lactone Hydrolysis in Acidic Conditions
3 jui 2013 · ABSTRACT: The acid-catalyzed hydrolysis of linear esters and lactones was studied using a hybrid supermolecule−polarizable continuum |






![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://imgv2-2-f.scribdassets.com/img/document/15042819/298x396/c6395ef292/1370198898?v\u003d1)









![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://haygot.s3.amazonaws.com/questions/195087_208142_ans.png)