base hydrolysis of amides mechanism
How does base hydrolysis work?
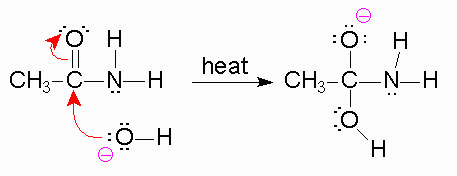
And from there, we learned that basic hydrolysis is when water acts as an acid to break apart a weak base.
For example, water can react with urea, forming a hydroxide anion, which can then release ammonia from the urea, which is critical for the growth of plants.Key Takeaway.
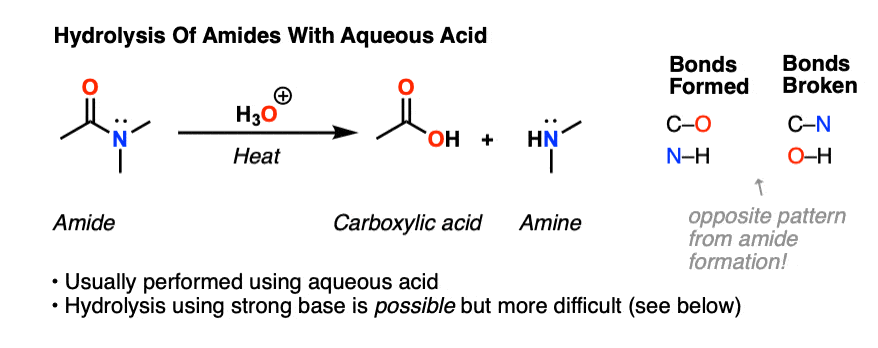
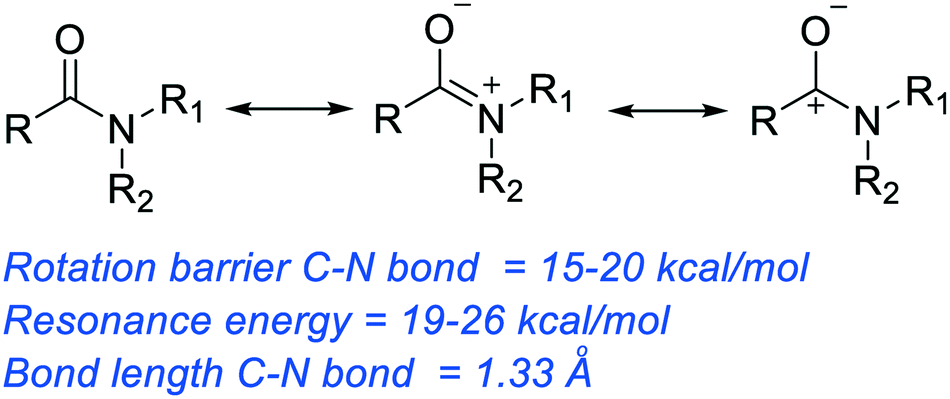
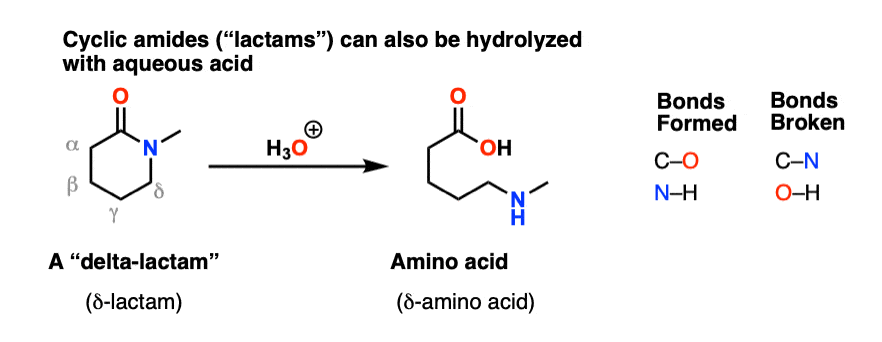
The hydrolysis of an amide produces a carboxylic acid and ammonia or an amine.
What is the mechanism of the amide reaction?
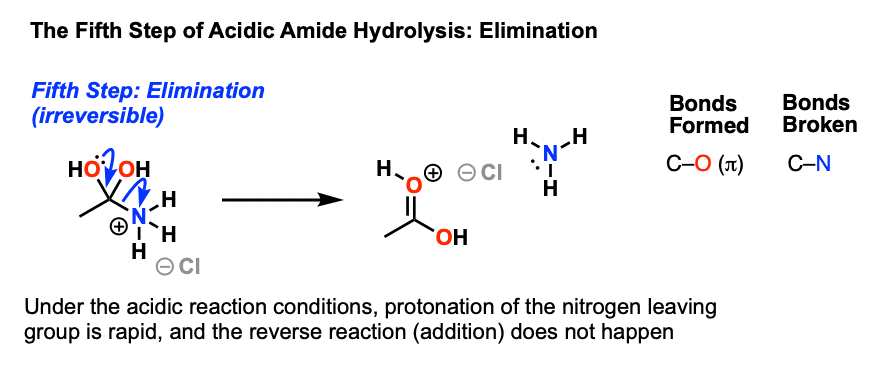
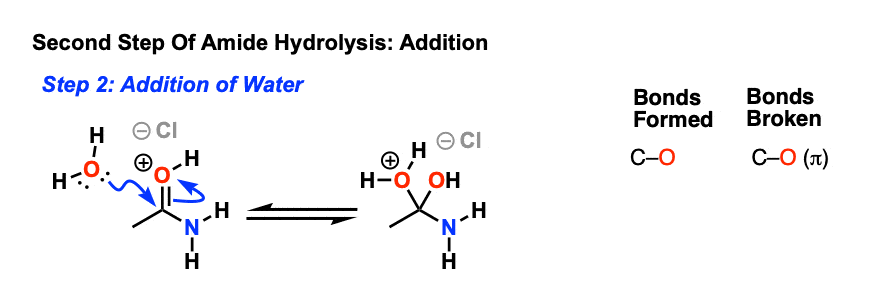
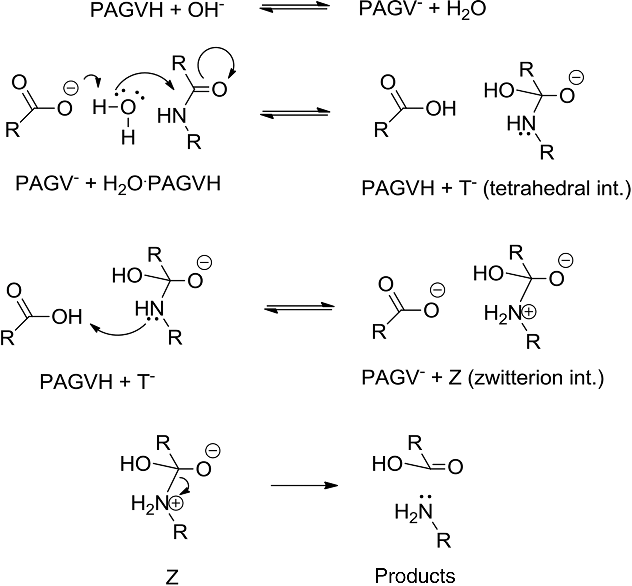
The mechanisms starts with the nucleophilic acyl substitution by water while breaking the amide C-N bond.
This produces a protein fragment with an amine and another with a carboxylic acid.
The amine fragment will deprotonate the carboxylic acid forming the carboxylate and ammonium fragments.24 sept. 2022
|
Hydrolysis of amides. Electronic effects on kinetics and mechanism
Hydrolysis of Amides. Electronic Effects on Kinetics and Mechanism of the Basic Hydrolysis of N-Substituted Benzoylpyrroles. |
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
The mechanism of the alkaline amide hydrolysis has been intensively investigated. 14-21. It is similar to that of the esters with the exception that the |
|
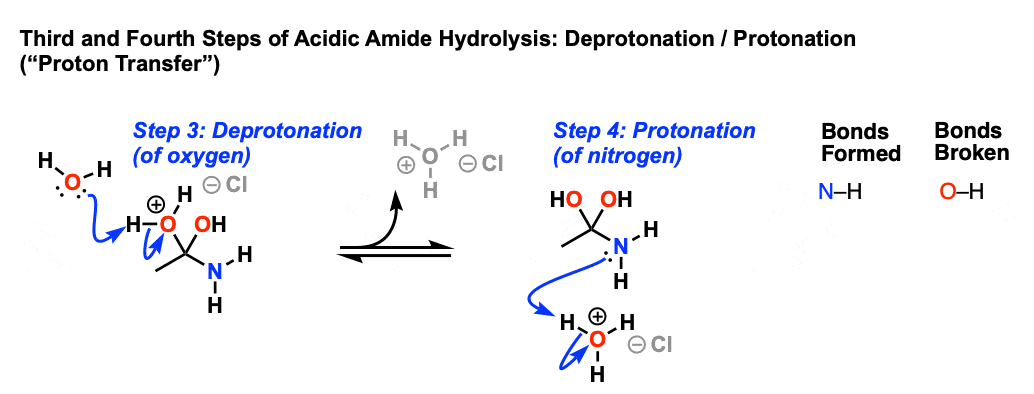
Acidic and Basic Amide Hydrolysis //
The mechanism of basic amide hydrolysis is straightforward and recent hydrolysis of amides in dilute acid solutions involves attack by a water molecule. |
|
On the hydrolysis mechanisms of amides and peptides
20 avr. 2018 amide formamide |
|
Theoretical Study of Base-Catalyzed Amide Hydrolysis: Gas-and
mechanism which has been suggested for the hydrolysis of simple aliphatic amides benzamides |
|
Theoretical Studies of Hydrolysis and Stability of Polyacrylamide
Both acid and base can catalyze the hydrolysis reaction by lowering activation Mechanisms of the water-promoted amide hydrolysis by a second water. |
|
Predominant oxygen-18 exchange accompanying the base
by —14 pAT units a unified mechanism for base-promoted hydrolysis is presented. hydrolysis of amides in base.1"* The current scheme that accounts. |
|
Predominant oxygen-18 exchange accompanying the base
by —14 pAT units a unified mechanism for base-promoted hydrolysis is presented. hydrolysis of amides in base.1"* The current scheme that accounts. |
|
Hydrolysis of Amides to Carboxylic Acids Catalyzed by Nb2O5
25 déc. 2020 KEYWORDS: Nb2O5 Catalyst Amide hydrolysis |
|
Base-promoted hydrolysis of amides at ambient temperatures
However base hydrolysis of amides is fre- quently an inefficient process. (7) More recent mechanistic support for our suggested6 mechanism has. |
|
On the hydrolysis mechanisms of amides and peptides
15 mai 2018 · amide, formamide, hydrolysis rate law, mechanism, peptide, square root which kineticists apply a rate law with base-, water-, and acid- |
|
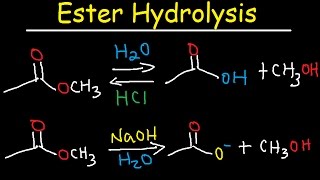
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions; • account for the irreversibility of the hydrolysis reaction under basic |
|
HYDROLYSIS
mechanisms account for neutral, acid and base hydrolysis Amides are inherently less reactive than carboxylic acid esters with half-lives typically > 103 yrs |
|
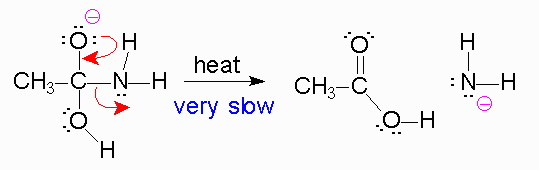
Hydrolysis of Amides
amides The reaction that occurs is proton-transfer (acid-base) RCOH O 2) dissociation of tetrahedral intermediate Mechanism of Amide Hydrolysis in Base |








![PDF] On the hydrolysis mechanisms of amides and peptides PDF] On the hydrolysis mechanisms of amides and peptides](https://image.slidesharecdn.com/21-2-part2reactionsofcarboxylicacidderivatives-wade7th-140409033918-phpapp02/95/212-part-2-reactions-of-carboxylic-acid-derivatives-wade-7th-20-638.jpg?cb\u003d1397015929)

![A New Mechanism for Amide Hydrolysis - [PDF Document] A New Mechanism for Amide Hydrolysis - [PDF Document]](https://imgv2-2-f.scribdassets.com/img/document/15042819/298x396/c6395ef292/1370198898?v\u003d1)
