base hydrolysis of ester procedure
What is the basic ester hydrolysis procedure?
The procedure for this reaction is starting with the ester, adding the water in an acid or basic solution (depending on the catalyst used), and then heating the mixture.
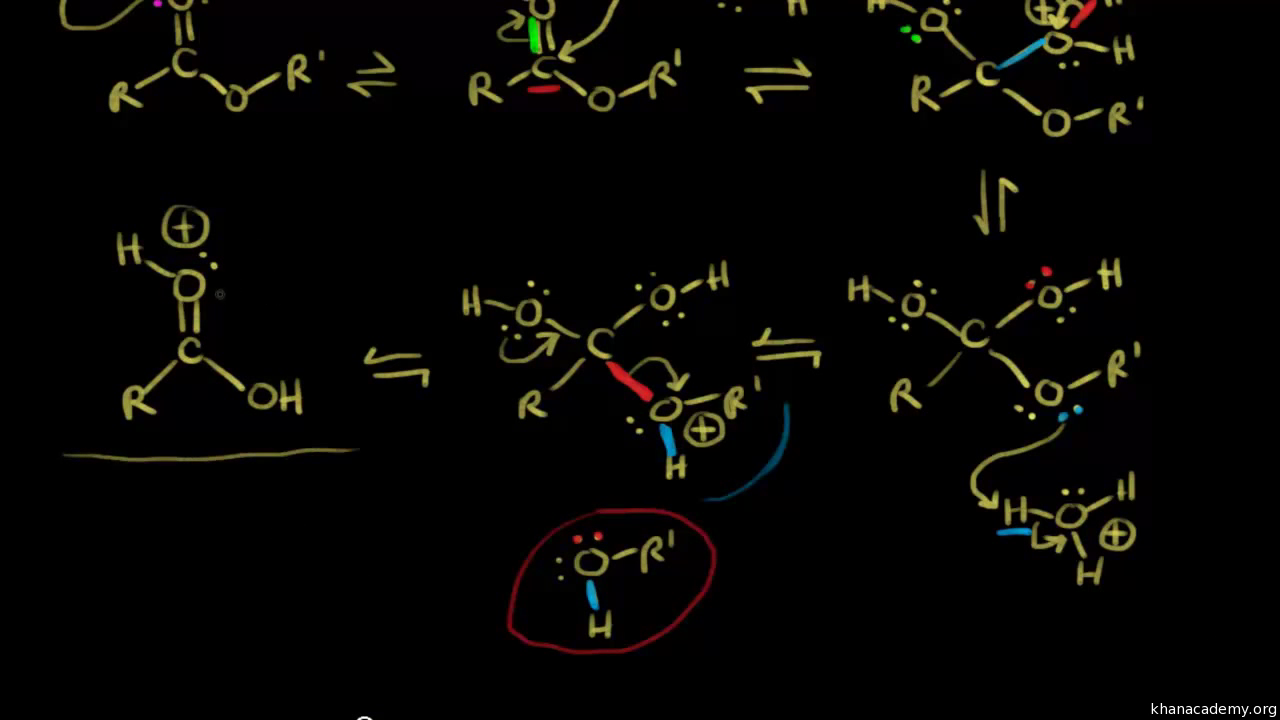
The acid protonates the oxygen, allowing the carbon in the carbonyl to be open to weak nucleophilic attacks.What is the mechanism of base hydrolysis of an ester?
Mechanism of ester hydrolysis with NaOH
In the first step, the hydroxide ion attacks the electron-deficient carbon centre of the ester.
It breaks the C=O. π bond of ester and forms a tetrahedral intermediate structure.
In the second step, an internal attack of O– ion takes place, reforming the C=O.Base-catalyzed hydrolysis of ester occurs by SN2 pathway and is irreversible, because the end product of base-catalyzed hydrolysis of ester produces alcohol and carboxilate ion (not carboxylic acid), which being resonance stabilized shows very little tendency to react with alcohol.
How do you perform the hydrolysis of an ester experiment?
Acidic hydrolysis is simply the reverse of esterification.
The ester is heated with a large excess of water containing a strong-acid catalyst.
Like esterification, the reaction is reversible and does not go to completion.
As a specific example, butyl acetate and water react to form acetic acid and 1-butanol.31 jan. 2022
|
A Mild and Selective Method for the Hydrolysis of Esters with
26 janv. 2005 General procedure: The carboxylic ester (0.01–0.15mmol) was dissolved in 12-dichloroethane and after addition of trimethyltin hydroxide (1–10 ... |
|
Solvent Effects and Ester Interchange in Basic Hydrolysis of Esters
The saponification of ethyl acetate and methyl acetate has been measured at 30° in dioxane-water this procedure shaking could be started within one. |
|
Kinetics of alkaline hydrolysis of the monomethyl ester of
hydrolysis according to the procedure described in paper [11]. to which the alkaline hydrolysis of esters is a bimolecular reaction [14] (mechanism. |
|
General Basic Catalysis of Ester Hydrolysis and Its Relationship to
The action of imidazole as a general basic catalyst offers only a partial ex- planation of the mechanism of enzymatic hydrolysis. Introduction. |
|
Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate
Day and Ingold proposed a sequential reaction mechanism passing through an addition complex. This mechanism of the basic hydrolysis of esters. |
|
The mechanism of hydrolysis of a cobalt(III)-bound phosphate ester
The intermediate hydroxo(phosphoramido)tetraammine complex is hydrolyzed slowly by base to liberate phosphoramidate anion by the same mechanism. The ester |
|
Hydrolysis of a Carboxylic Acid Ester: Neutral and Base Enhanced
where Kw is the autoionization constant for water. Chemistry 331: Laboratory Manual. Environmental Organic Chemistry. Page 3. In this experiment |
|
A Facile Base-catalyzed Ester Hydrolysis Involving Alkyl-Oxygen
(4) base catalysis. Reaction 1 is of course the ordinary mechanism by which most esters undergo base-catalyzed hydrolysis.4 Anchimeric catalysis (2)has been. |
|
The Kinetics of the Base-catalyzed Methanolysis of Ortho Meta and
Although the basic hydrolysis of esters has been the subject of numerous course of each experiment (five or six polarimeter readings. |
|
Efficiency of lithium cations in hydrolysis reactions of esters in
31 mars 2021 6-13) These reports noted that LiOH was an effective base for the hydrolysis of esters but did not explain the mechanism. In addition many of ... |
|
Facile Hydrolysis of Esters with KOH-Methanol at Ambient
27 nov 2003 · Ester hydrolysis has been known for long and is usually catalyzed by acids and bases Both acidic and alkaline hydrolysis are equilibrium |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
Hydrolysis of a Carboxylic Acid Ester: Neutral and Base Enhanced Reaction of p -Nitrophenyl Acetate Background: The investigation of anthropogenic organic |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions form new esters by base- or acid-catalysed transesterification mechanisms; |
|
IV SEMMESTER
EXPERIMENT Page No 1 Kinetics of Acid hydrolysis of an ester 2 2 Estimation of mixture of acids conductometrically 4 3 Estimation of Copper (II) by |
|
Benzoic Acid from Ethyl Benzoate by Base Hydrolysis
This process is called base hydrolysis (or saponification) of an ester and is used in this experiment to first obtain sodium benzoate solution, and then benzoic |
|
Saponification (Base Hydrolysis) of Organic Materials
Saponification (base hydrolysis) is more frequently employed than acid hydrolysis The procedure is commonly applied to total lipid extracts (TLE) to release ester- |




















![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://0.academia-photos.com/attachment_thumbnails/46422824/mini_magick20190209-11202-1cnp8ew.png?1549775948)
![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/10/2-opposite-of-condensation-is-hydrolysis-which-can-be-achieved-through-aqueous-acid-treatment-of-amides-or-concentrated-hcl-to-give-carboxylic-acid-and-amine.gif)