base hydrolysis of ester reversible
Hydrolysis using dilute alkali
There are two big advantages of doing this rather than using a dilute acid.
The reactions are one-way rather than reversible, and the products are easier to separate.
What is the reverse of ester hydrolysis?
Acidic hydrolysis is simply the reverse of esterification.
The ester is heated with a large excess of water containing a strong-acid catalyst.
Like esterification, the reaction is reversible and does not go to completion.
As a specific example, butyl acetate and water react to form acetic acid and 1-butanol.31 jan. 2022
Is hydrolysis is reversible?
Hydrolysis is a reversible reaction, meaning that the alcohol and acid groups can react with each other to produce a polyester bond and water as a by-product.
Are ester reactions reversible?
Esters are made in esterification reactions, which are reversible reactions between an alcohol and a carboxylic acid, using a strong acid as a catalyst.
Esters can be hydrolysed using an acid or a base.
|
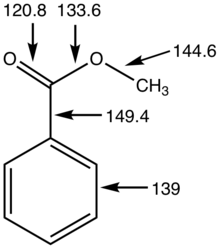
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
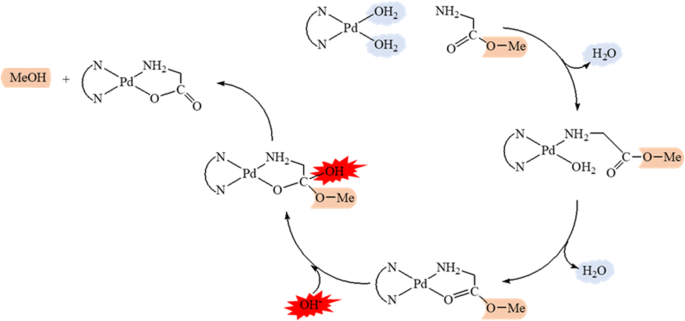
base is not a catalyst because it is consumed by the reaction in Eq. 21.9b. Finally acid-cat- alyzed ester hydrolysis is reversible |
|
Covalent Capture: Merging Covalent and Noncovalent Synthesis
13 feb 2009 Covalent capture by the irreversible or reversible formation of ... basic hydrolysis of an ester moiety (Figure 18b). Functional. |
|
Covalent Capture: Merging Covalent and Noncovalent Synthesis
13 feb 2009 Covalent capture by the irreversible or reversible formation of ... basic hydrolysis of an ester moiety (Figure 18b). Functional. |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides - Objectives
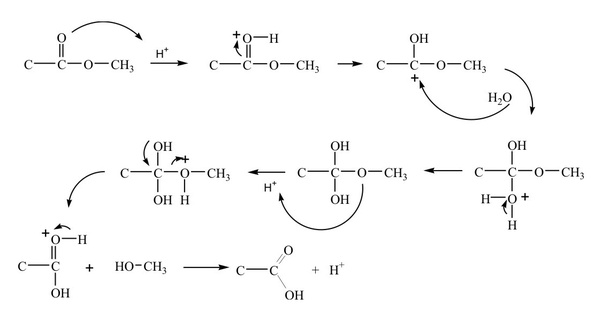
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions Base-Mediated Hydrolysis of Methyl Esters ... This step is reversible. |
|
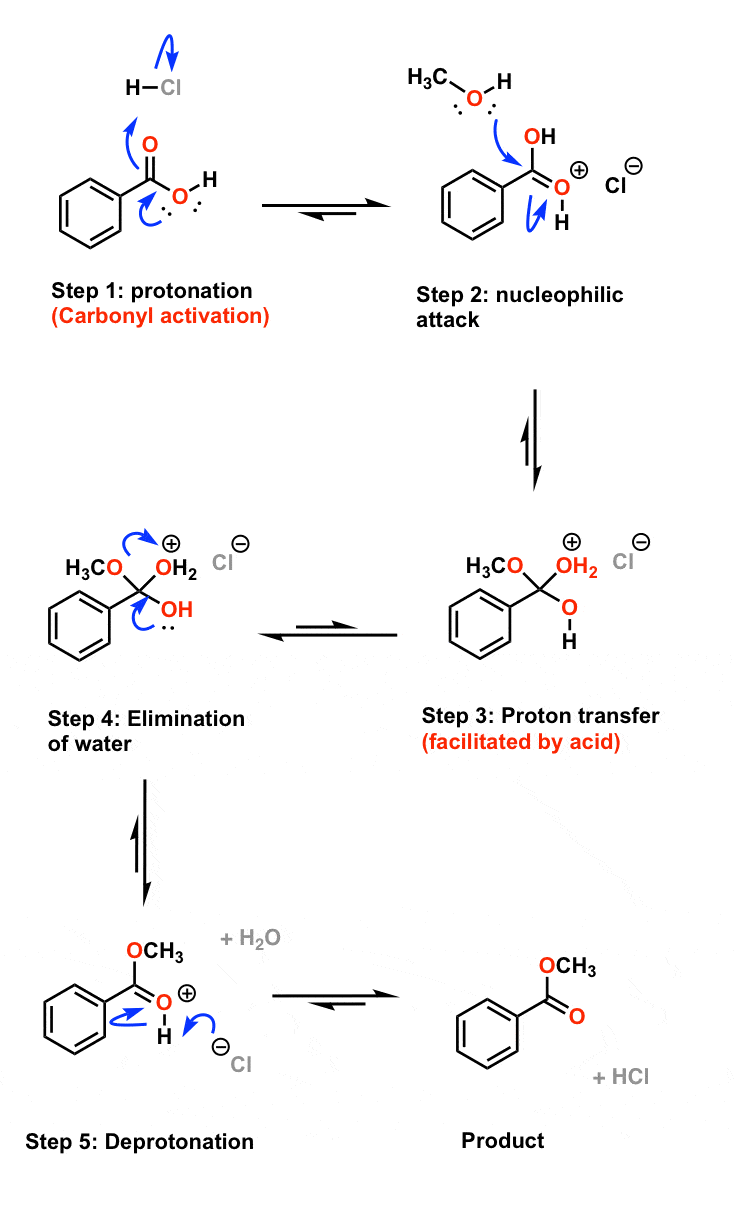
Mechanisms of Acid Hydrolysis of Carboxylic Acid Esters and Amides
tic acid made slightly basic withdilute ammonia |
|
Chapter 5 Carboxylic Acids and Esters
predict the products of ester synthesis and hydrolysis reactions. • Learn some of the important The dissociation of a carboxylic acid is a reversible. |
|
Exploiting Neighboring-Group Interactions for the Self-Selection of a
20 feb 2008 a 60-fold rate enhancement in the basic hydrolysis of a neighboring carboxylic ester; that is the selection of a. |
|
Exploiting Neighboring-Group Interactions for the Self-Selection of a
20 feb 2008 a 60-fold rate enhancement in the basic hydrolysis of a neighboring carboxylic ester; that is the selection of a. |
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
reaction is the hydrolysis of an ester with basic saponification of ethyl acetate the value of ... irreversible second order reaction occurs as a. |
|
Imido Ester Formation in Aromatic Nitriles
triles with alcohols; (ii) the faster reversible base- then the hydrolysis to ester andNH4+ commenced and that acid-base but not Cl- titers were. |
|
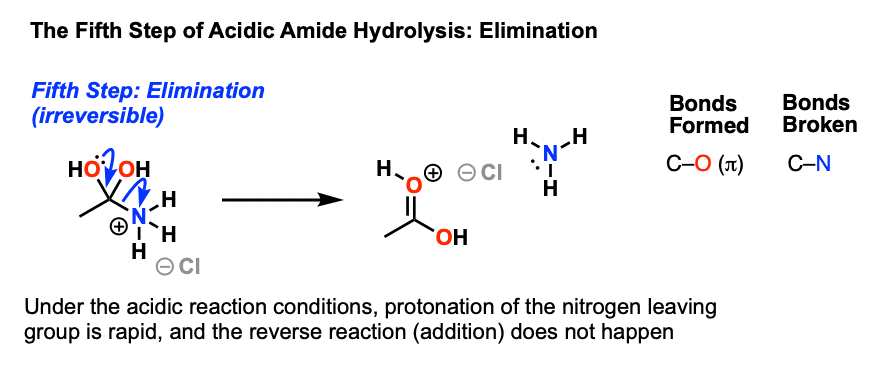
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions; • account for the form new esters by base- or acid-catalysed transesterification mechanisms; • draw the This step is reversible • Collapse of the |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
base is not a catalyst because it is consumed by the reaction in Eq 21 9b Finally, acid-cat- alyzed ester hydrolysis is reversible, but saponification is irreversible, |
|
Ester hydrolysis-lectute note
Acid Catalysed hydrolysis are reversible bert not alkaline hydrolysts – A shorthand notation had been suggested to describe these mechanisms in which A and |
|
Mechanisms of Lactone Hydrolysis in Acidic Conditions
3 jui 2013 · A parallel work addresses the neutral and base-catalyzed hydrolysis of lactones 1 The existing mechanisms of acid-catalyzed ester hydrolysis can be seen as reversible and using very harsh conditions 7,8 In this work |
|
Text Related to Segments 1902 & 1903 ©2002 Claude E Wintner
Commonly known as "saponification," the base-catalyzed hydrolysis of esters occurs by irreversible, since it is driven to the right by the formation of the stable |
|
Ester Chemistry Tutorial
This reaction is reversible and may simply revert back to the Basic conditions enhance the rate of ester hydrolysis by increasing the concentration of attacking |











![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://scielo.conicyt.cl/fbpe/img/jcchems/v58n2/tab13.1.gif)





![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://upload.wikimedia.org/wikipedia/commons/thumb/3/34/Ester-general.svg/1200px-Ester-general.svg.png)