base hydrolysis of ethyl ethanoate
What is the base hydrolysis of an ester?
Hydrolysis is a most important reaction of esters.
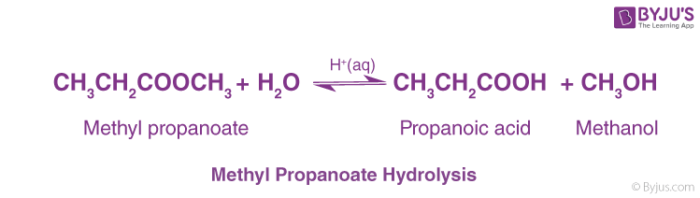
Acidic hydrolysis of an ester gives a carboxylic acid and an alcohol.
Basic hydrolysis of an ester gives a carboxylate salt and an alcohol.What is produced by hydrolysis of ethyl ethanoate?
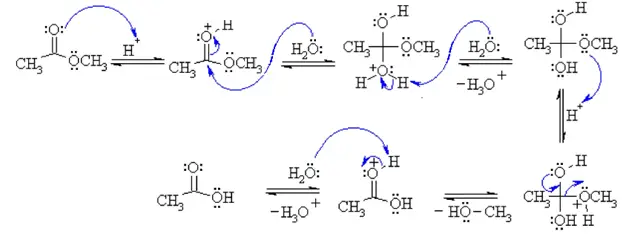
The mechanism for the hydrolysis of ethyl ethanoate
When ethyl ethanoate is heated under reflux with a dilute acid such as dilute hydrochloric acid or dilute sulfuric acid, the ester reacts with the water present to produce ethanoic acid and ethanol.22 jan. 2023What is the mechanism of base hydrolysis of ethyl acetate?
The reaction of ethyl acetate and hydroxide ions yields ethanol and acetate ions, as shown below.
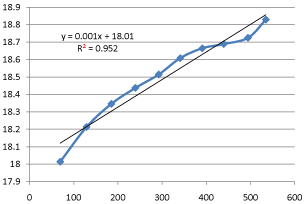
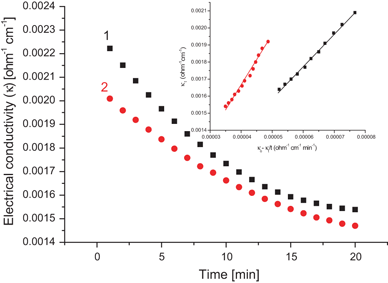
The progress of this reaction can be observed by monitoring the conductivity of the reaction mixture.
Although the reactants and products each contain an ion, the OH– ion has a higher ionic mobility than the CH3COO– ion.
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
ABSTRACT: Alkaline hydrolysis of ethyl acetate is essentially an irreversible and second order reaction. Industrial importance of the reaction product |
|
The Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate
The Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate. By Hiroo TSUJIKAWA and Hakuai. INOUE. Department of Chemical Engineering The University of |
|
Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate
The reaction rate of the alkaline hydrolysis of ethyl acetate was studied by means of a con- tinuous measurement of the electric conductivity change. The second |
| Determination of the Expression Rate of Ethyl Acetate Hydrolysis |
|
XXIII. The Alkaline Hydrolysis of Ethyl Acetate in Dimethyl
organic solvent. In contrast to this the rate of alkaline hydrolysis of ethyl acetate in dimethyl sulphoxide-water mixtures increases with. |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Chemical reactions reaction rate. Chemical kinetics is the part of physical chemistry that studies reaction rates. The reaction. |
|
The Alkaline Hydrolysis of Ethyl Acetate in Acetone—Water Mixtures
Alkaline Hydrolysis of Ethyl Acetate in Acetone-Water. 675 in all subsequent analyses of samples. The equation given above does not require an extrapolation |
|
The Alkaline Hydrolysis of Ethyl Acetate from the Standpoint of Ion
The alkaline hydrolysis of ethyl acetate was chosen for the purpose of further investigating the ion—dipolar molecule type of reaction. The veloc-. |
|
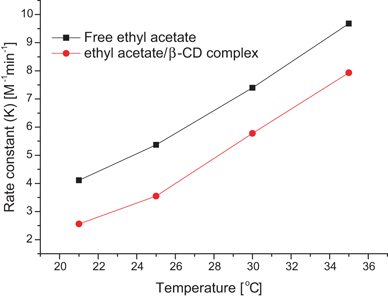
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide) 500ml reactor vessels which contains 0.05N of NaOH and thoroughly mixed. |
|
Solvent Effects and Ester Interchange in Basic Hydrolysis of Esters
The saponification of ethyl acetate and methyl acetate has been measured at 30° in dioxane-water mixtures containing additional solvents found earlier to |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
i The hydrolysis of an ester such as ethyl acetate in the presence of a mineral acid gives acetic acid and ethyl alcohol Therefore, the kinetics of the reaction can |
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
ABSTRACT: Alkaline hydrolysis of ethyl acetate is essentially an irreversible and second order reaction Industrial importance of the reaction product, sodium |
|
Hydrolysis of Ethyl Acetate
This experiment is the first in a series of kinetic studies Ethyl acetate hydrolyzes in alkaline solution to give ethanol and acetate The production of acetate, at the |
|
Hydrolysis of Ethyl Acetate
Hydrolysis of Ethyl Acetate Ethyl acetate (CH3 2H5) hydrolyzes in alkaline solution according to the reaction EtAc + OH = EtOH + Ac- (26-1) Even at room |
|
KINETICS OF HYDROLYSIS OF ETHYL ACETATE
follow the progress of a reaction in solution The reaction studied is the hydrolysis of ethyl acetate which can be represented by the chemical equation: |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
When the endpoint of titration has been reached, read the used volume of NaOH from the burette (VNaOH) Write it down to the Table 1 7 Repeat the step 5 and 6 |