basic ester hydrolysis mechanism
What is the mechanism of base-induced ester hydrolysis (saponification)?
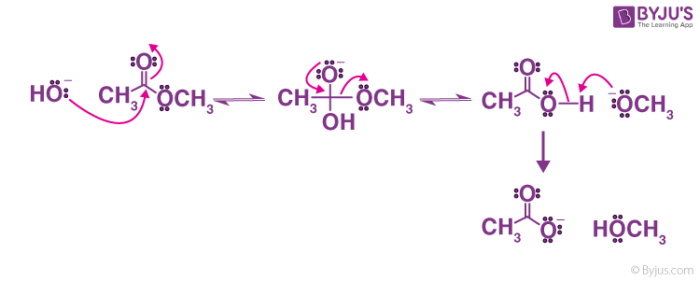
Mechanism of base-induced ester hydrolysis (saponification). The mechanism shown in Figure 21.8 is supported by isotope-labeling studies. When ethyl propanoate labeled with 18 O in the ether-like oxygen is hydrolyzed in aqueous NaOH, the 18 O label shows up exclusively in the ethanol product.
What is the mechanism of ester hydrolysis?
This organic chemistry video tutorial provides the mechanism of the ester hydrolysis reaction catalyzed by an acid or promoted under basic conditions. Under acidic conditions, the ester will be converted into a carboxylic acid and an alcohol. Under alkaline conditions, it will chang
What is a base used to hydrolyze an ester?
When a base (such as sodium hydroxide [NaOH] or potassium hydroxide [KOH]) is used to hydrolyze an ester, the products are a carboxylate salt and an alcohol. Because soaps are prepared by the alkaline hydrolysis of fats and oils, alkaline hydrolysis of esters is called saponification (Latin sapon, meaning “soap,” and facere, meaning “to make”).
Does water hydrolyze esters?
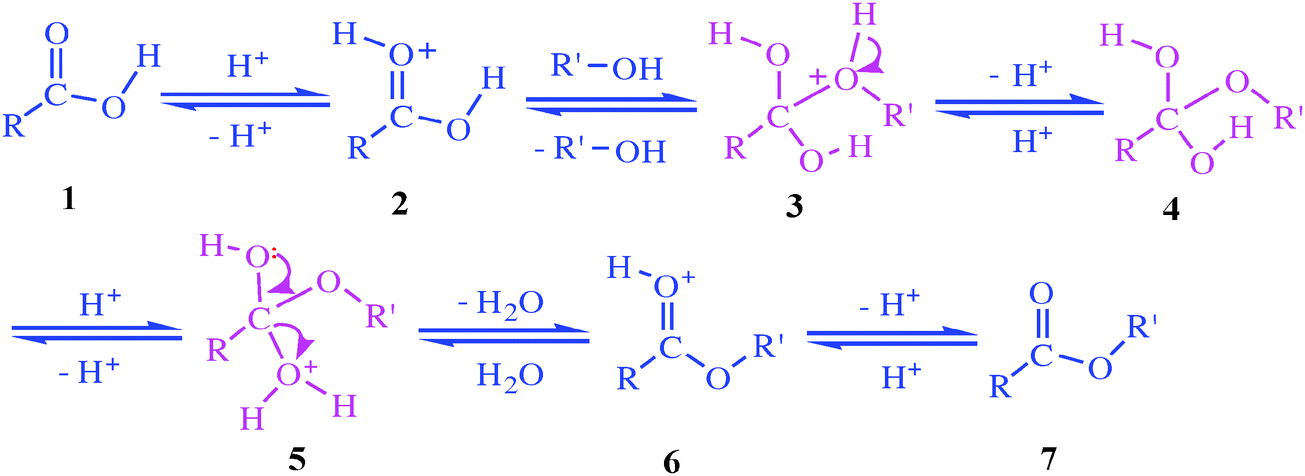
Ester hydrolysis is usually catalyzed by acid or bases. The hydrolysis of ester may involve acyl-oxygen bond breaking (more common) or alkyl-oxygen bond breaking (less common). Since, -OR’ (alkoxy) is a much poorer leaving group than halide or -OCOR (carboxylate) group, water alone does not hydrolyze much esters.

Ester Hydrolysis Reaction Mechanism

Acid-catalyzed ester hydrolysis Organic chemistry Khan Academy

s=
|
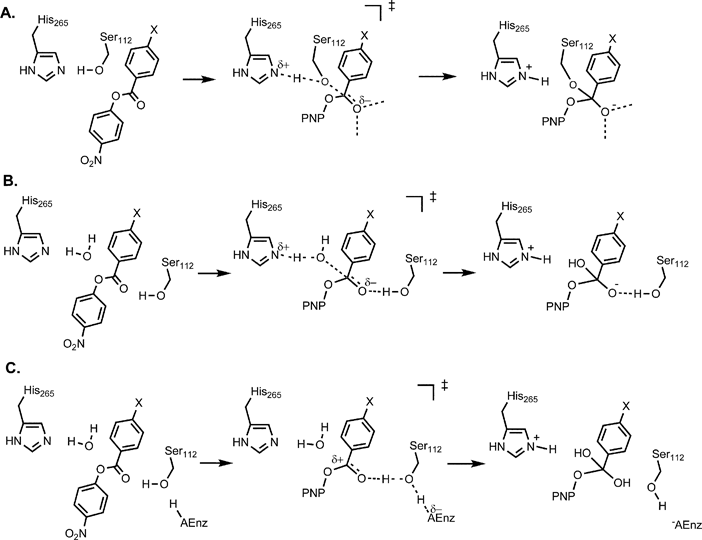
General base catalysis of ester hydrolysis
Both nucleophilic and general base mechanisms of catalysis by acetate anions are observed for the hydrolysis of substituted phenyl formates with leaving |
|
Mechanisms of carbonyl participation in phosphate ester hydrolysis
termined the mechanism of the alkaline hydrolysis of methyl- acetoin diethyl phosphate and related compounds using a com- bination of kinetic and isotope |
|
Mechanisms of carbonyl participation in phosphate ester hydrolysis
into phosphate ester products was determined by 31P NMR analysis. Studies of proton-exchange reactions show that the conjugate base. |
|
Studies on the BAL2 mechanism for ester hydrolysis
JUDY E. DOUGLAS GRANT CAMPBELL |
|
Revisiting the mechanisms of low-temperature base-catalysed ester
Abstract: Ester interchange reactions such as the interesterification of triglycerides and their transes- terification with methanol (methanolysis) to |
|
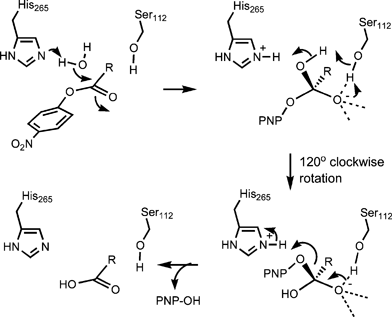
Carbanion mechanism of ester hydrolysis. II. o-Nitrophenyl .alpha
The second-order rate constant for alkaline hydrolysis of p-nitrophenyl acetate is within reactions of ester types la and lb with both lyate species. |
|
The mechanism of hydrolysis of a cobalt(III)-bound phosphate ester
The intermediate hydroxo(phosphoramido)tetraammine complex is hydrolyzed slowly by base to liberate phosphoramidate anion by the same mechanism. The ester |
|
A Facile Base-catalyzed Ester Hydrolysis Involving Alkyl-Oxygen
(4) base catalysis. Reaction 1 is of course the ordinary mechanism by which most esters undergo base-catalyzed hydrolysis.4 Anchimeric catalysis (2)has been. |
|
Carbonion (ElcB)mechanism of ester hydrolysis. I. Hydrolysis of
modes of HO~ attack on malonate esters are conceivable a priori (1 and 2a and 2b). In eq 1 the mechanism is that for normal alkaline hydrolysis of. |
|
Steric Effects in the Hydrolysis of N-Acylimidazoles and Esters of p
for reactions exemplifying both nucleophilic and classical general base mechanisms. Nucleophilic catal- ysis of ester hydrolysis is strongly retarded bysub-. |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
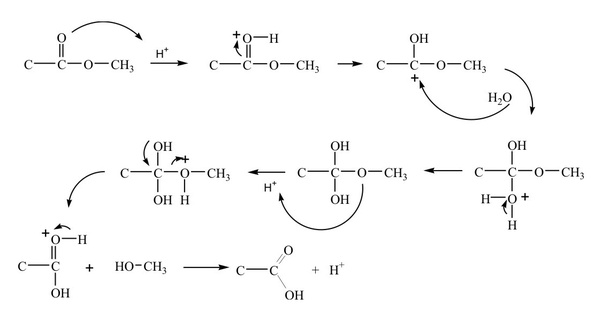
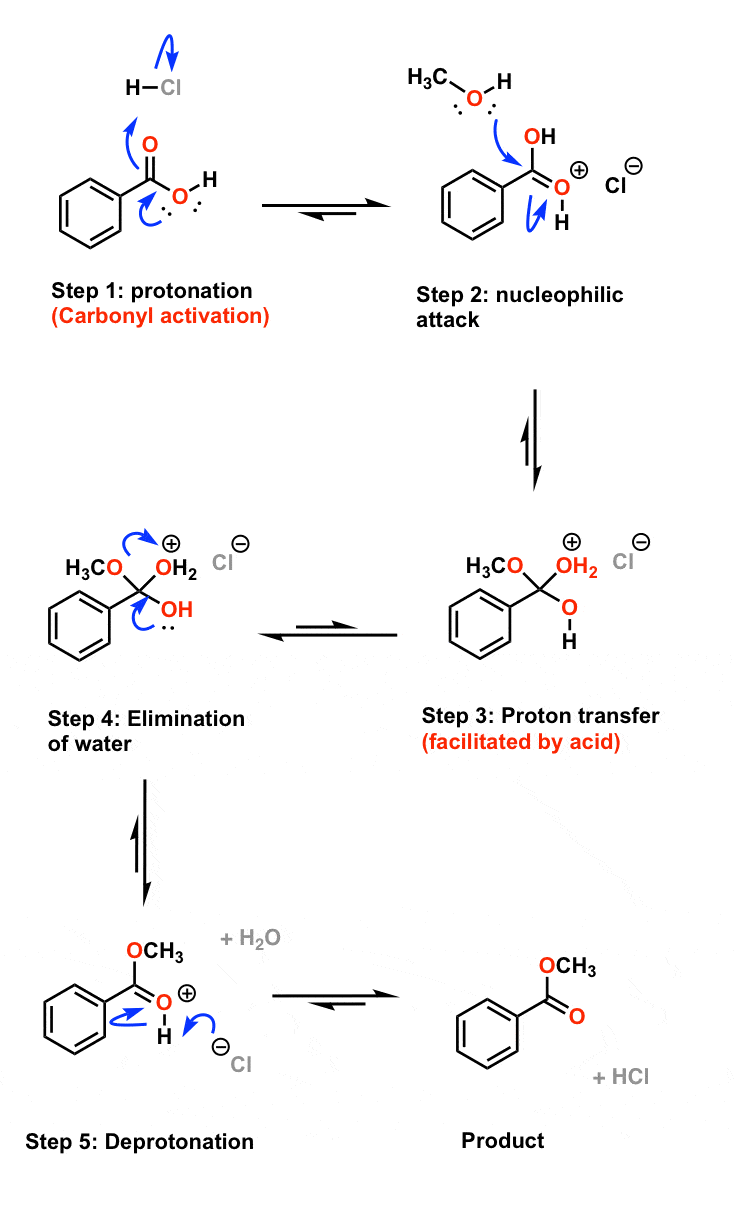
Simple alkyl esters, such as methyl and ethyl esters, are hydrolysed under acidic conditions in a reaction mechanism that is described as an AAC2 reaction: the reaction is acid-catalysed, the acyl−oxygen bond is cleaved and there are two molecules involved in the rate-determining step, i e it is a bimolecular process |
|
HYDROLYSIS
carbon centre, such as with carboxylic acid derivatives including esters, anhydrides, amides, mechanisms account for neutral, acid and base hydrolysis |
|
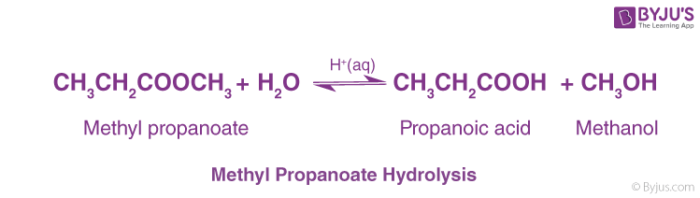
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
The hydrolysis of a carboxylic acid ester may proceed by a number of different mechanisms, depending on the substrate structure, the pH and the presence of catalyzing species (3-6) Under neutral conditions, the reaction generally proceeds via addition to the carbonyl carbon to produce a tetrahedral intermediate |
|
Facile Hydrolysis of Esters with KOH-Methanol at Ambient
27 nov 2003 · Both acidic and alkaline hydrolysis are equilibrium reactions [1] However for preparative purposes, ester hydrolysis is invariably performed in |
|
Ester hydrolysis-lectute note
Thus BAC2 hydrolysis indicates a bimolecular basic hydrolysis of an ester which proceeds through acyl- oxygen bond cleavage Type of mechanism e |
|
GENERAL BASE AND NUCLEOPHILIC CATALYSIS OF ESTER
In addition to general base-catalyzed hydrolysis reactions, general base- catalyzed aminolysis reactions are common The reaction of glycine with N,O- |
|
Mechanisms of Lactone Hydrolysis in Acidic Conditions
3 jui 2013 · 1 INTRODUCTION As is also the case with neutral and base-catalyzed mechanisms, the acid-catalyzed hydrolysis of esters has seldom been |