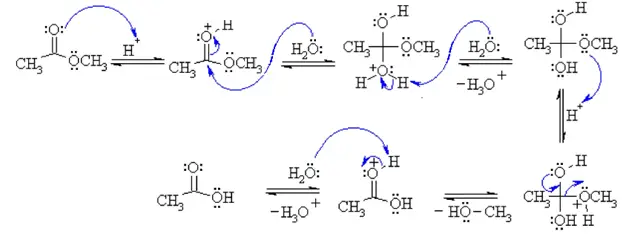
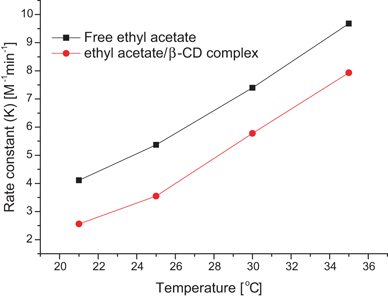
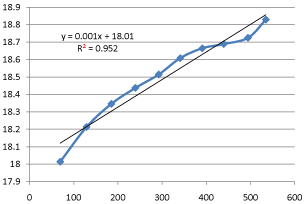
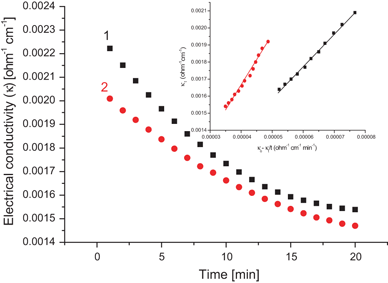
basic hydrolysis of ethyl acetate
How do ethyl acetate and NaOH react?
As a specific example, ethyl acetate and NaOH react to form sodium acetate and ethanol: Write an equation for the hydrolysis of methyl benzoate in a potassium hydroxide solution. In basic hydrolysis, the molecule of the base splits the ester linkage. The acid portion of the ester ends up as the salt of the acid (in this case, the potassium salt).
What is hydrolysis of ethyl acetate?
Hydrolysis is a most important reaction of esters. Acidic hydrolysis of an ester gives a carboxylic acid and an alcohol. Basic hydrolysis of an ester gives a carboxylate salt and an alcohol. Write an equation for the acid-catalyzed hydrolysis of ethyl acetate. Write an equation for the base-catalyzed hydrolysis of ethyl acetate.
What is hydrolysis of ethyl propanoate in a sodium hydroxide solution?
Write the equation for the hydrolysis of ethyl propanoate in a sodium hydroxide solution. Hydrolysis is a most important reaction of esters. Acidic hydrolysis of an ester gives a carboxylic acid and an alcohol. Basic hydrolysis of an ester gives a carboxylate salt and an alcohol.
How do you write an equation for acidic hydrolysis of ethyl butyrate?
Write an equation for the acidic hydrolysis of ethyl butyrate (CH 3 CH 2 CH 2 COOCH 2 CH 3) and name the products. Remember that in acidic hydrolysis, water (HOH) splits the ester bond. The H of HOH joins to the oxygen atom in the OR part of the original ester, and the OH of HOH joins to the carbonyl carbon atom:
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
ABSTRACT: Alkaline hydrolysis of ethyl acetate is essentially an irreversible and second order reaction. Industrial importance of the reaction product |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
ABSTRACT. Hydrolysis is a chemical decomposition involving breaking of a bond and the addition of elements of water. In this hydrolysis of ester (ethyl |
|
Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate
The reaction rate of the alkaline hydrolysis of ethyl acetate was studied by means of a con- tinuous measurement of the electric conductivity change. The second |
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
Zia-ul-Haq et al define the saponification reaction is the hydrolysis of an ester with basic conditions to produce an alcohol and the sodium salt of carboxylic |
|
The Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate
The Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate. By Hiroo TSUJIKAWA and Hakuai. INOUE. Department of Chemical Engineering The University of |
|
The Alkaline Hydrolysis of Ethyl Acetate from the Standpoint of Ion
The alkaline hydrolysis of ethyl acetate was chosen for the purpose of further investigating the ion—dipolar molecule type of reaction. The veloc-. |
|
Hydrolysis of Ethyl Acetate
Hydrolysis of Ethyl Acetate. Ethyl acetate (CH3COOC2H5) hydrolyzes in alkaline solution according to the reaction. EtAc+ OH = EtOH + Ac¯. |
|
Solvent Effects and Ester Interchange in Basic Hydrolysis of Esters
The saponification of ethyl acetate and methyl acetate has been measured at 30° in dioxane-water mixtures containing additional solvents found earlier to |
|
Effect of Pressure on the Alkaline Hydrolysis of Ethyl Acetate in
The effect of pressures up to 3 kbar on the rate of the base-catalyzed hydrolysis of ethyl acetate in acetone-water mixtures has been measured. |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Chemical reactions reaction rate. Chemical kinetics is the part of physical chemistry that studies reaction rates. The reaction. |
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
Hydrolysis of carboxylic ester under alkaline conditions, also known as saponification reaction, produces soap and an alcohol Though sodium acetate, basically a |
|
Hydrolysis of Ethyl Acetate
Hydrolysis of Ethyl Acetate Ethyl acetate (CH3 2H5) hydrolyzes in alkaline solution according to the reaction EtAc + OH = EtOH + Ac- (26-1) Even at room |
|
Hydrolysis of Ethyl Acetate
This experiment is the first in a series of kinetic studies Ethyl acetate hydrolyzes in alkaline solution to give ethanol and acetate The production of acetate, at the |
|
KINETICS OF HYDROLYSIS OF ETHYL ACETATE
follow the progress of a reaction in solution The reaction studied is the hydrolysis of ethyl acetate which can be represented by the chemical equation: |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Determine the rate constant and the activation energy of the alkaline hydrolysis of ethyl acetate using sodium hydroxide This experiment illustrates a |
|
IV SEMMESTER
KINETICS OF ACID HYDROLYSIS OF AN ESTER AIM: To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE : |





![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](https://i1.rgstatic.net/publication/11483798_Hydroxide_as_General_Base_in_the_Saponification_of_Ethyl_Acetate/links/5c742d1a458515831f6fd7c5/largepreview.png)