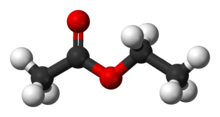
basic hydrolysis of ethyl acetate gives acetate ion and
What are the products of a basic hydrolysis of an ester?
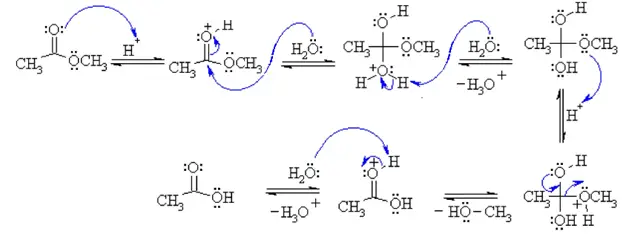
Identify the products of a basic hydrolysis of an ester. Esters are neutral compounds, unlike the acids from which they are formed. In typical reactions, the alkoxy (OR′) group of an ester is replaced by another group. One such reaction is hydrolysis, literally “splitting with water.”
What happens when ethyl acetate and hydroxide ions react?
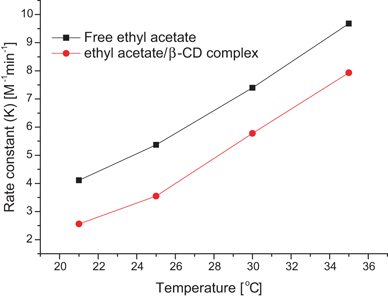
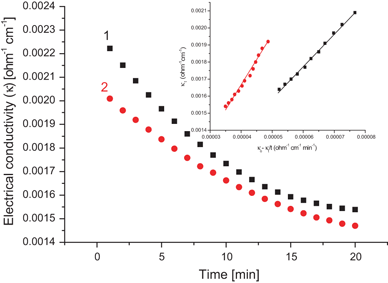
The reaction of ethyl acetate and hydroxide ions yields ethanol and acetate ions, as shown below. The progress of this reaction can be observed by monitoring the conductivity of the reaction mixture. Although the reactants and products each contain an ion, the OH – ion has a higher ionic mobility than the CH 3 COO – ion.
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide) HCl was the presence of a mineral acid gives acetic acid and. |
| The Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate |
|
The Alkaline Hydrolysis of Ethyl Acetate from the Standpoint of Ion
later we have found that atomic oxygen acting on UsOs gives rise to the same crystal modi- fication of U03 (Phase I) as does nitrogen dioxide. As mentioned |
| Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate |
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
Zia-ul-Haq et al define the saponification reaction is the hydrolysis of an ester with basic conditions to produce an alcohol and the sodium salt of carboxylic |
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
suppresses the conductance of sodium acetate. Therefore the conductivity readings were accounted for the unconsumed hydroxide ions as the reaction proceeds and. |
|
XXIII. The Alkaline Hydrolysis of Ethyl Acetate in Dimethyl
organic solvent. In contrast to this the rate of alkaline hydrolysis of ethyl acetate in dimethyl sulphoxide-water mixtures increases with. |
|
Determination of the p K a of Ethyl Acetate: Brønsted Correlation for
ester in aqueous solution because these esters undergo compet- ing hydrolysis induced by hydroxide ion to give the correspond- ing carboxylic acids (khyd |
|
Nonlinear Structure-Reactivity Correlations. The Imidazole
for base catalysis of imidazole-catalyzed hydrolysis fact that ethyl acetate does not undergo a base-cata- ... lower temperatures to give an. |
|
General Basic Catalysis of Ester Hydrolysis and Its Relationship to
ion on the hydrolysis of ethyl acetate.9 However General catalysis by acetic acid ... tions from Skellysolve B gave an almost colorless product |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
The hydrolysis of an ester such as ethyl acetate in the presence of a mineral acid gives acetic acid and ethyl alcohol |
|
Hydrolysis of Ethyl Acetate
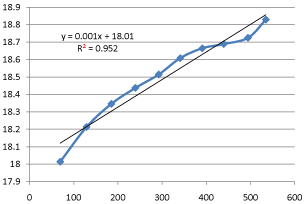
Ethyl acetate (CH3 2H5) hydrolyzes in alkaline solution according to the reaction Since the conductivities of the hydroxyl and acetate ions are very different, the hydrolysis proceeds gives a value for R, from which Lo = 1/Ro Second |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
outside concentration: mainly temperature, ionic strength, surface area of the gives a straight line, which slope and intercept can be used to determine thr Ea the rate constant and the activation energy of the alkaline hydrolysis of ethyl |
|
IV SEMMESTER
KINETICS OF ACID HYDROLYSIS OF AN ESTER AIM: To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE : |
|
Kinetics of Aquation
The Kinetics of the Saponification of Ethylacetate1,2 Purpose: reaction of ethylacetate with base using conductance measurements Prelab: Make sure These steps are followed by a fast H+ ion transfer to give the acetate ion: H3C OH O |
|
Effect of Ion Exchange Resin Catalyst on Hydrolysis of Ethyl Acetate
alternative heterogeneous catalyst for hydrolysis reaction of ethyl acetate The resin had to be washed using sulphuric acid to give an H+ charge on the catalyst |