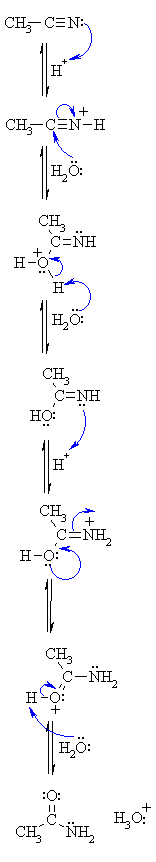
basic hydrolysis of nitriles mechanism
Can nitrile be hydrolyzed to carboxylic acids?
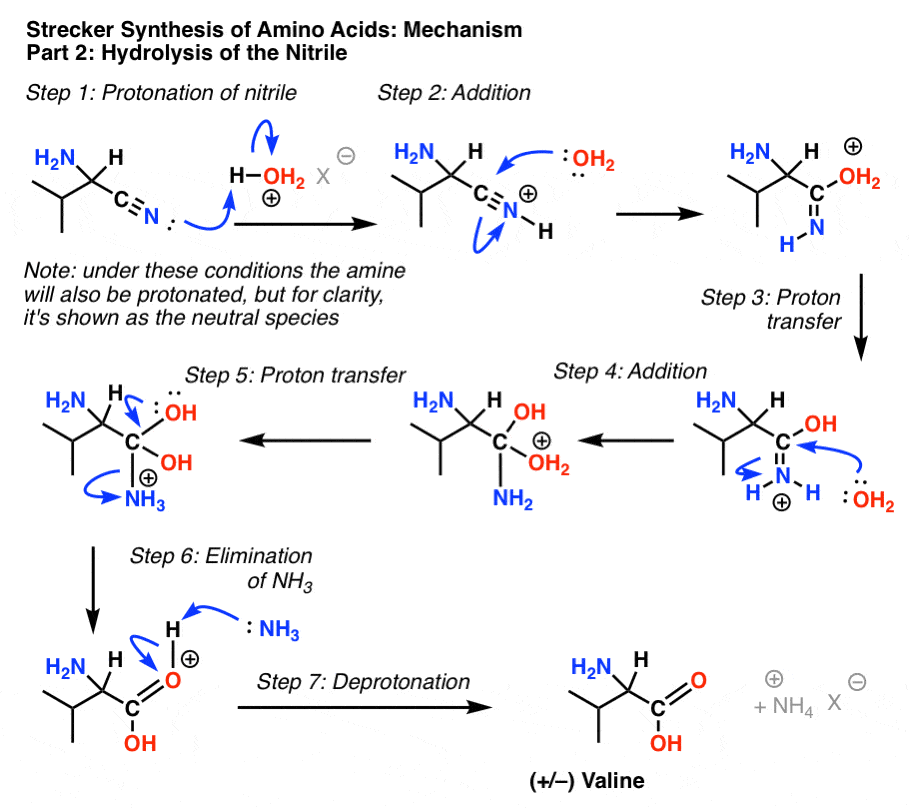
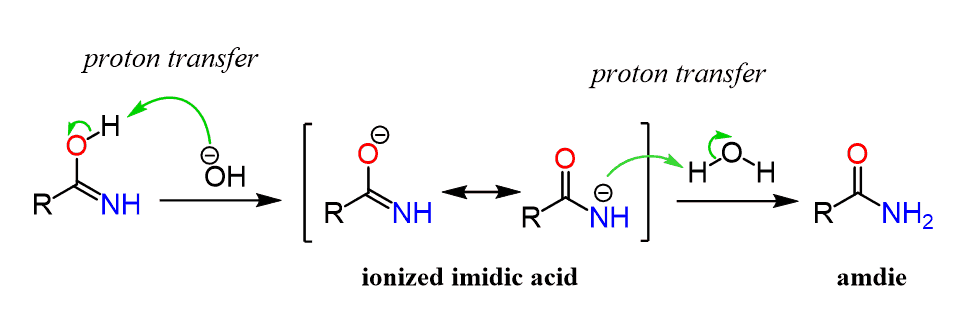
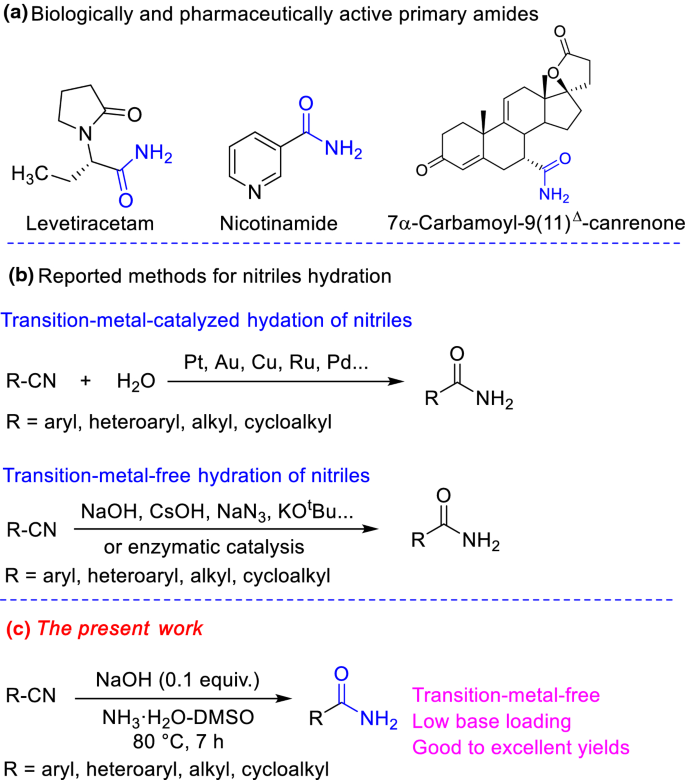
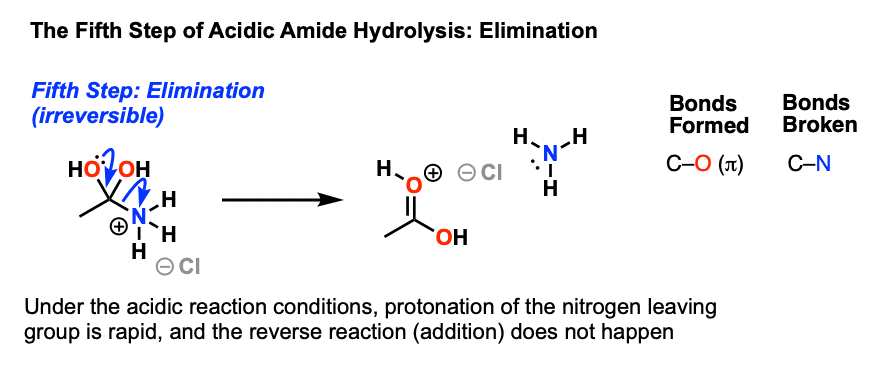
Nitriles can be hydrolyzed to carboxylic acids in acidic aqueous solutions, and to carboxylate salts with base-catalyzed hydrolysis: In both cases, the transformation consists of two main parts; conversion of the nitrile to an amide and hydrolysis of the amide to the corresponding carboxylic acid.
What happens when nitriles are hydrolysed?
This page looks at the hydrolysis of nitriles under either acidic or alkaline conditions to make carboxylic acids or their salts. When nitriles are hydrolysed you can think of them reacting with water in two stages - first to produce an amide, and then the ammonium salt of a carboxylic acid.

Hydrolysis of Nitriles

Hydrolysis of Nitriles

Synthesis of Carboxylic Acids: Hydrolysis of Nitriles
|
20_18_20.html.ppt [Read-Only]
Hydrolysis of nitriles resembles the hydrolysis In basic solution the carboxylic acid product ... The mechanism of amide formation is analogous. |
|
Chem 360 Jasperse Ch. 20 21 Notes + Answers. Carboxylic Acids
via Formation and Hydrolysis of Nitriles (Reaction 6) mechanism required for acid chloride to ester conversion: addition-elimination- deprotonation. |
|
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
term saponification can be used to refer to the hydrolysis in base of any carboxylic acid derivative. The mechanism of ester saponification involves the |
|
Chapter 21: Carboxylic Acid Derivatives
mechanism it is possible to transform one acid The reactivity of acid derivatives can be correlated ... Base Catalyzed Hydrolysis of Nitriles. |
|
Chem 360 Jasperse Ch. 20 21 Notes + Answers. Carboxylic Acids
via Formation and Hydrolysis of Nitriles (Reaction 6) mechanism required for acid chloride to ester conversion: addition-elimination- deprotonation. |
|
Reactivity of coordinated nitriles
Abstract: The base hydrolysis of coordinated acrylonitrile in 180 tracer studies indicate the mechanism of hydrolsis by carbonate ion to involve direct ... |
|
Untitled
Mechanism of the Reaction of Nitriles with Alkaline Hydrogen Peroxide. The alkaline hydrolysis: 0.1-0.4 M NaOH 1-2.5 M. MeCN or 0.1 M PhCN. |
|
Alkaline hydrolysis of polyacrylonitrile. On the reaction mechanism
Nov 1 2000 reactive in hydrolysis than nitrile groups. These data enable us to reconsider the mechanism of alkaline PAN hydrolysis. |
|
HYDROLYSIS 2016.pdf
Hydrolysis reactions are generally enhanced by both acids and bases and three independent reaction mechanisms account for neutral acid and base hydrolysis. |
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
Keywords: Alkaline hydrolysis amides |
|
Hydrolysis of Nitriles
Hydrolysis of nitriles resembles the hydrolysis of amides The reaction is In basic solution the carboxylic acid product Mechanism of Hydrolysis of Nitriles |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
D, a so-called orthoester, can either be isolated as such or hydrolyzed to give a normal carboxylic acid ester B under mild conditions (mechanism: Figure 9 13) In |
|
88 Chapter 20: Carboxylic Acids and Nitriles 201 Naming
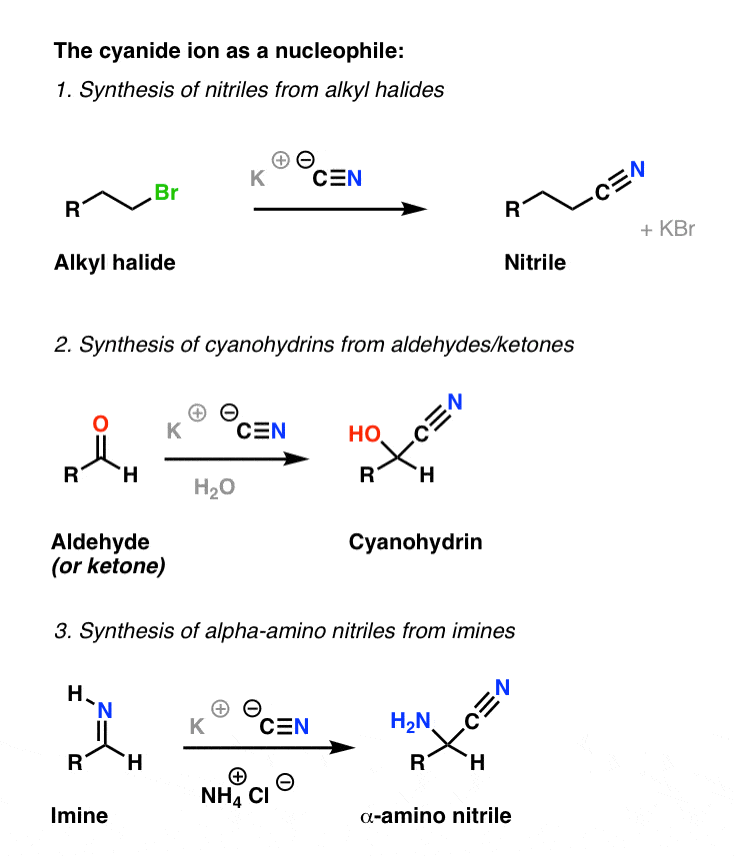
Acid or basic hydrolysis of a nitrile (mechanism, Fig 20 4) cyanide ion is an excellent nucleophile and will react with 1° and 2° alkyl halides and tosylates to give nitriles This reaction add one carbon |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
Notice that nitriles behave mechanistically much like carbonyl compounds Compare, for example, the mechanism of acid-promoted nitrile hydrolysis in Eqs 21 20 |
|
General Reaction of Anhydrides
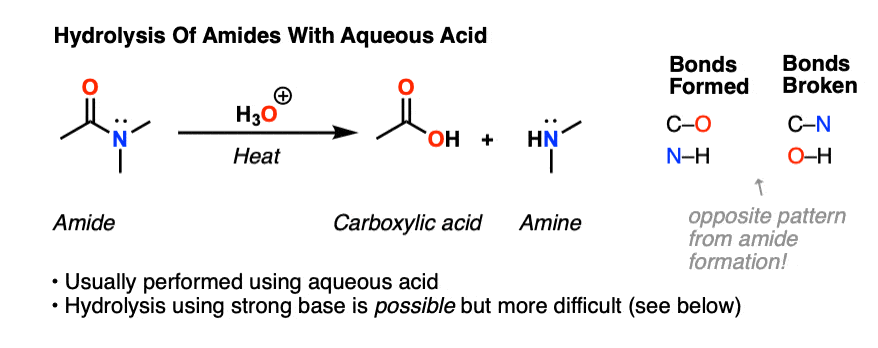
The mechanism is similar to acid-catalyzed esterification/hydrolysis • Esters can Amides are hydrolyzed in acid or base to form carboxylic acids or carboxylate |
|
PDF (Chapter 24) - Caltech Authors
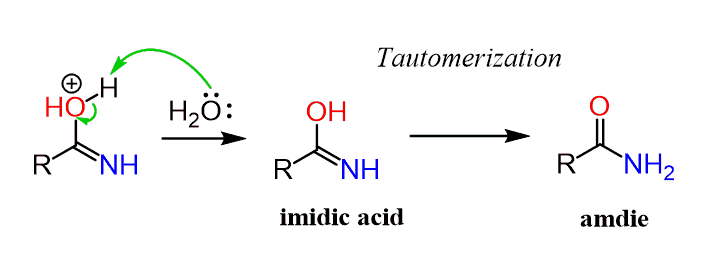
24-4 HYDROLYSIS OF AMIDES Generally, amides can be hydrolyzed in either acidic or basic solution The mechanisms are much like those of ester hydrolysis |