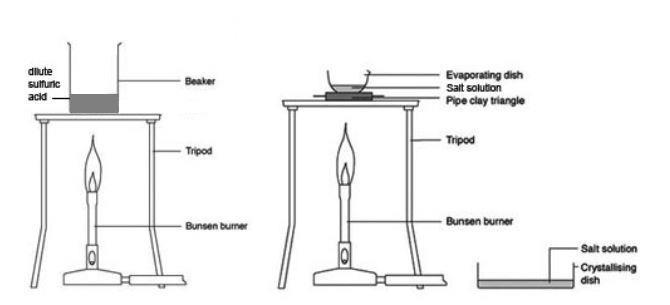
To standardise hydrochloric acid - Creative Chemistry
|
Volumetric Analysis 2 To standardise hydrochloric acid
This process is known as standardising the hydrochloric acid The reaction between sodium carbonate and hydrochloric acid takes place in two stages: |
|
Titration calculations for pleasure and profit
of a solution of hydrochloric acid Calculate the concentration of the acid 8 23 15cm 3 of 0 125M NaOH is needed to titrate 25 00cm 3 of a solution of |
|
Volumetric Analysis – Acids & Bases HL
To standardise a solution of HCl using a standard solution of Na2CO3 Acid: HCl – strong acid Base: Na2CO3 – weak base Indicator: Methyl Orange (SAWBMO) |
|
Standardisation of a hydrochloric acid solution using a standard
In this experiment a standard solution of sodium carbonate is used to determine the exact concentration of a hydrochloric acid solution The neutralisation |
How do you standardize 0.1 N HCl?
Hydrochloric acid (HCl) is not standard substance; therefore it must be standardized by using standard sodium carbonate (Na2CO3).
Na2CO3 is a solid substance.
Dissolve 1.325 g Na2CO3 in 250 ml of distilled water to obtain 0.
1) N Na2CO3.
HCl is a liquid substance.How do you standardize hydrochloric acid using sodium carbonate solution?
Acid-base titration involves reacting a known volume of a solution (HCL) with a standard solution (Na2CO3) sodium carbonate is used as the primary standard in volumetric analysis.
Concentration of hydrochloric acid will be obtained by reacting the standard sodium carbonate solution with hydrochlloric acid solution.How do you standardize hydrochloric acid?
Hydrochloric Acid Solution Standardization
Weigh accurately about 1.5 g of anhydrous sodium carbonate, previously heated at about 270°C for 1 hour.
Dissolve it in 100 ml of water and add 0.1 ml of methyl red solution.
Add the acid slowly from a burette, with constant stirring, until the solution becomes faintly pink.Primary standard sodium carbonate ( N a 2 C O 3 ) was used to standardize a solution of hydrochloric acid (HCl) by acid-base titration.
|
Volumetric Analysis 2 To standardise hydrochloric acid
In the last practical you prepared a standard solution of sodium carbonate. Today you will use it to find the concentration of dilute hydrochloric acid by |
|
Year 12 ATAR Chemistry - Unit 3: Acid Base Titrations
Calculate the concentration of the sodium hydroxide in the original cleaning solution. 2. In a titration 0.105 mol L-1 HCl is used to standardise KOH solution |
|
Volumetric Analysis 3 To determine the relative molecular mass of a
In Volumetric Analysis 1 & 2 you prepared a standard solution of sodium carbonate and hydrochloric acid according to the overall equation below:. |
|
The Role of Virtual and Non- Standard Laboratory Experiences in
skills and creative abilities. performance modeling |
|
Safe Work
Referring to the segregation chart and their SDS John discovers the following: Hydrochloric acid: This chemical is a corrosive acid. It will react with the |
|
EN Horizon Europe Work Programme 2021-2022 8. Climate Energy
10 mai 2022 HORIZON-CL5-2021-D1-01-06: Supporting and standardising climate services ... HORIZON-CL5-2022-D3-02-03: Innovative renewable energy carrier ... |
|
BTEC Assignment Brief
chemical plant Chemcalequip. hydrochloric acid (HCl) by titration with your primary ... https://www.creative-chemistry.org.uk/alevel/practical.htm. |
|
Green Chemistry
9 juin 2020 compared with conventional hydrochloric acid leaching ... This article is licensed under a Creative Commons ... internal standard. |
|
The optimization of standard test method for chemical composition of
Reagents: lithium metaborate mixed flux(lithium hydroxide and boric acid |
| Corrosion Inhibition of Mild Steel in Hydrochloric Acid Environment |
|
To standardise hydrochloric acid - Creative Chemistry
This process is known as standardising the hydrochloric acid Two indicators are needed to cover both stages: in stage 1, phenolphthalein is most suitable, and will respond to the pH change associated with the formation of sodium hydrogencarbonate, NaHCO3 Titrate with the hydrochloric acid |
|
To Standardise Hydrochloric Acid Creative Chemistry
Hydrochloric Acid an overview ScienceDirect Topics Acids 2012 Book Archive PREPARATION OF A STANDARD SODIUM HYDROXIDE SOLUTION Preparation |
|
BTEC Assignment Brief
chemical plant, Chemcalequip As part of your hydrochloric acid (HCl) by titration with your primary standard unknown concentration with the standardised HCl • You must use https://www creative-chemistry uk/alevel/ practical htm |
|
Stardization Of Hcl Acid With Stard Naoh Solution - Blue Bus
Hydrochloric Acid Titration ChemistryTo standardise hydrochloric acid - Creative ChemistryStandardisation of a hydrochloric acid Hydrochloric Acid is a highly corrosive and hazardous chemical and should be handled with extreme care |
|
Acid base titrations - ATAR Chemistry
released under Creative Commons CC BY NC Before re-purposing any third party substances are not a primary standard: NaOH (deliquescence) and Na2CO3 The equivalence point occurs when the number of moles of hydrochloric acid |
|
Sodium Carbonate And Hydrochloric Acid Edexcel
To Standardise Hydrochloric Acid Creative Chemistry Titration Of Sodium Carbonate With Hydrochloric Acid Sodium Bicarbonate Hydrochloric Acid |
|
Sodium Carbonate And Hydrochloric Acid Edexcel
11 mai 2018 · To standardise hydrochloric acid Creative Chemistry January 2016 IAL QP Unit 3 Edexcel Chemistry A level Mole Ratios and Reaction 1 / 8 |
|
RSC resources for A level Chemistry - Pearson qualifications
Royal Society of Chemistry resources for A level Chemistry practicals use volumetric flask, including accurate technique for making up a standard solution 6 use acid-base indicators in titrations of weak/strong acids with weak/strong alkalis 7 purify: • a solid product Rate of Reaction – Magnesium and Hydrochloric acid |