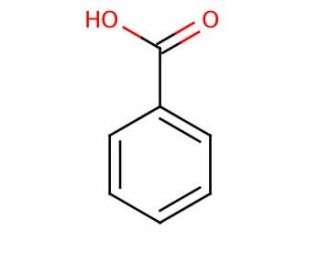
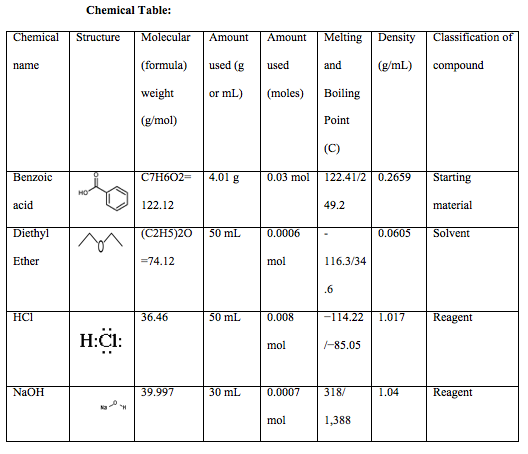
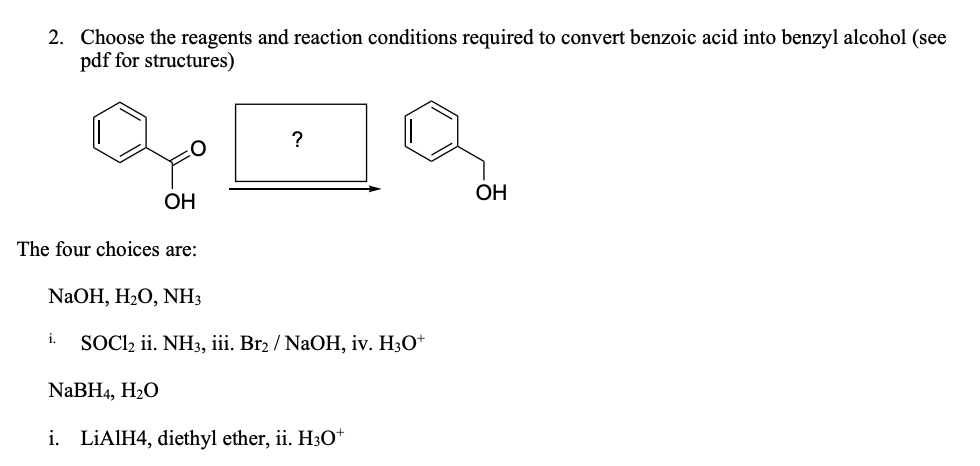
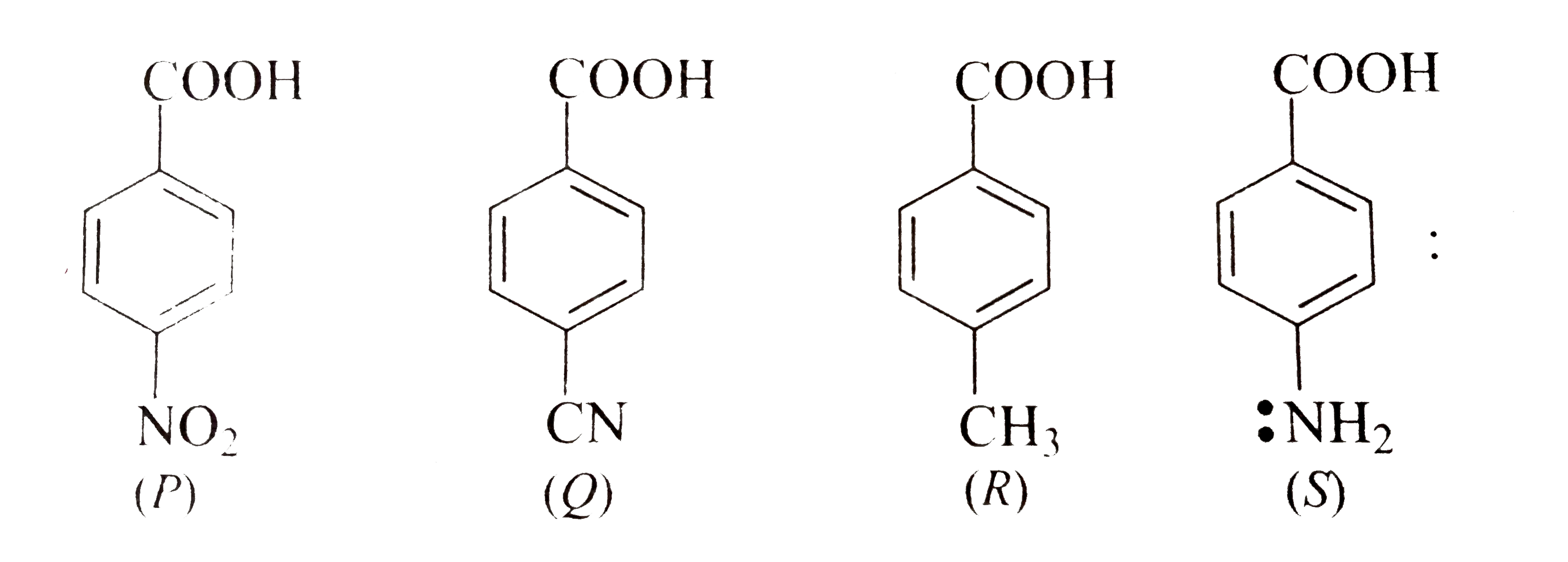
benzoic acid plus nh3
|
Reactions of Benzene & Its Derivatives
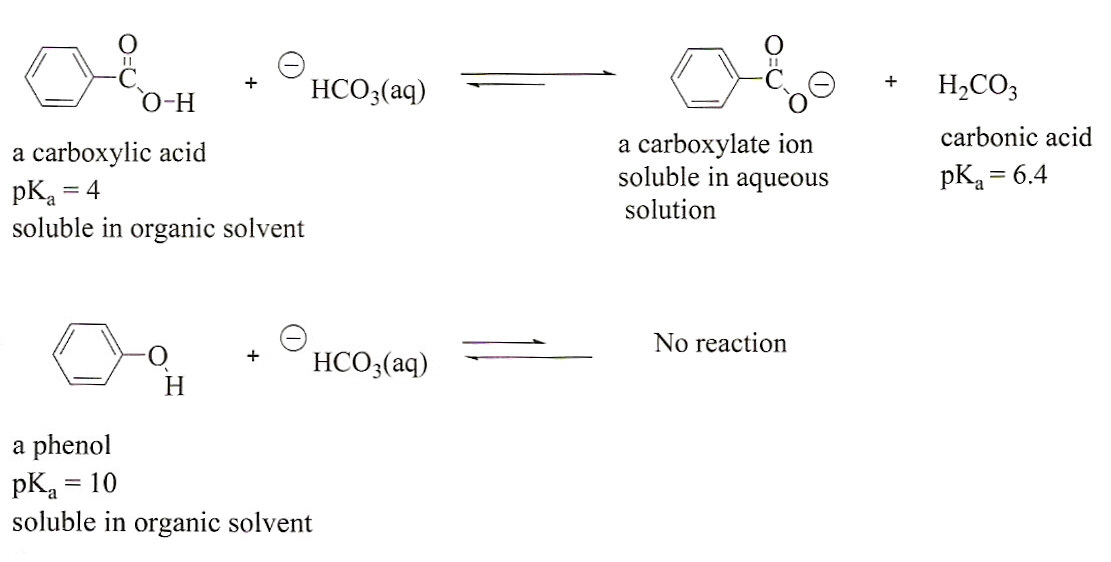
Reactions of Benzene & Its Derivatives Chapter 22 1 Reactions of Benzene The most characteristic reaction of aromatic compounds is substitution at a ring carbon: Halogenation: H + Cl2 FeCl3 Cl + HCl Chlorobenzene Nitration: H2 SO4 H + HNO3 NO2 + H2 O Nitrobenzene Reactions of Benzene Sulfonation: SO3 H2 SO4 SO3 H Benzenesulfonic acid |
|
Sample Exercise 17.1 Calculating the pH When a Common Ion is
Calculate the pH of a buffer composed of 0.12 M benzoic acid and 0.20 M sodium How many moles of NH4Cl must be added to 2.0 L of 0.10 M NH3 to form a ... |
|
Chapter 5 Carboxylic Acids and Esters
after the parent compound benzoic acid. Carboxylic acids react with strong bases such as ... carboxylic acid with an alcohol or phenol (plus an. |
|
DETERMINATION OF URINE PH AND AMMONIA EMISSION AFTER
of benzoic acid and the fiber in diets on the urinary pH ammonia of the The data were subjected to one-way ANOVA using Statgraphic Plus package ( ... |
|
WORKSHEET: SOLUTION EQUILIBRIUM (Weak acids and bases
How many moles of NaOH must be added to a 1.00 liter of 0.230 M benzoic acid HC7H502 |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
be benzoic acid and compound (A) should therefore |
|
CHEM1102 2014-J-4 June 2014 Calculate the pH of a 0.010 M
04-Jun-2014 Write the equation for the reaction that occurs when benzoic acid reacts with ammonia? Marks. 5. C6H5COOH(aq) + NH3(aq) ? C6H5COO–(aq) + ... |
|
Chapter 3: Stoichiometry
the carbon reacts with oxygen to form CO2 Example: The compound para-aminobenzoic acid (you ... If 1.50 g of NH3 reacts with 2.75 g O2 |
|
24-DIHYDROXYBENZOYL-ORTHO-BENZOIC ACID AND SOME OF
form an addition product which then reacts with a second molecule of Meyer and Conzetti3 prepared 2 |
|
Acids and Basesx
A conjugate acid – base pair differ by H+. For example NH4. + is the conjugate acid of NH3 and NH3 is the conjugate base of NH4. +. Write the formula of the. |
|
Dr. Stewards CHM152 Exam #2 Review Spring 2014 (Ch. 16. 17
Explain how NH3 is both a Bronsted/Lowry base and a Lewis base. (assume the benzoic acid was added to the water with no other ions initially)? The acid. |
|
Exam Paper Topics for CHEM1002 November 2012
2 nov 2012 · Write the equation for the reaction that occurs when benzoic acid reacts with ammonia? Marks 5 C6H5COOH(aq) + NH3(aq) → C6H5COO–(aq) |
|
MASTER IJAS - CORE
Effects of benzoic acid on nitrogen, phosphorus and energy balance and on ammonia emission from slurries in the heavy pig Gianluca Galassi, Luca Malagutti, |
|
Incidence de lincorporation dacide benzoïque dans lalimentation
acid in the diet on performance and ammonia emission Two levels of incorporation were compared to a control diet, i e 0 5 and 1 of benzoic acid The pigs |
|
7 Transferts de protons Equilibres acide-base - EPFL
L'ammoniac NH3 capte un proton H+ de l'eau pour former NH4+ De même, il réagit Plus un acide sera fort, plus la valeur de Ka sera élevée et plus celle de pKa sera faible De même, plus une Benzoïque C6H5COOH 4 19 Ammoniac |
|
Publication Ineris- DRC-17-163622-06436A - Mis en ligne le 27/11
27 nov 2017 · « Influence of adding 0,5 or 1 of benzoic acid to the feed of growing-finishing pigs on ammonia emission and performance » (Guingand, Broz, & |
|
DETERMINATION OF URINE PH AND AMMONIA EMISSION AFTER
of benzoic acid and the fiber in diets on the urinary pH, ammonia of the slurry and redistribution of nitrogen between faeces and urinary For the pigs there were |











![What is the action of ammonia [NH3] on benzoic acid? Write equation What is the action of ammonia [NH3] on benzoic acid? Write equation](https://www.prepchem.com/uploads/b1/7d/6c7c209fa88d06d55514f2501607.png)