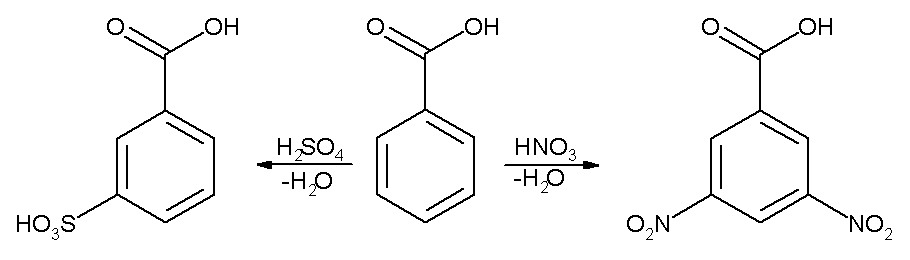
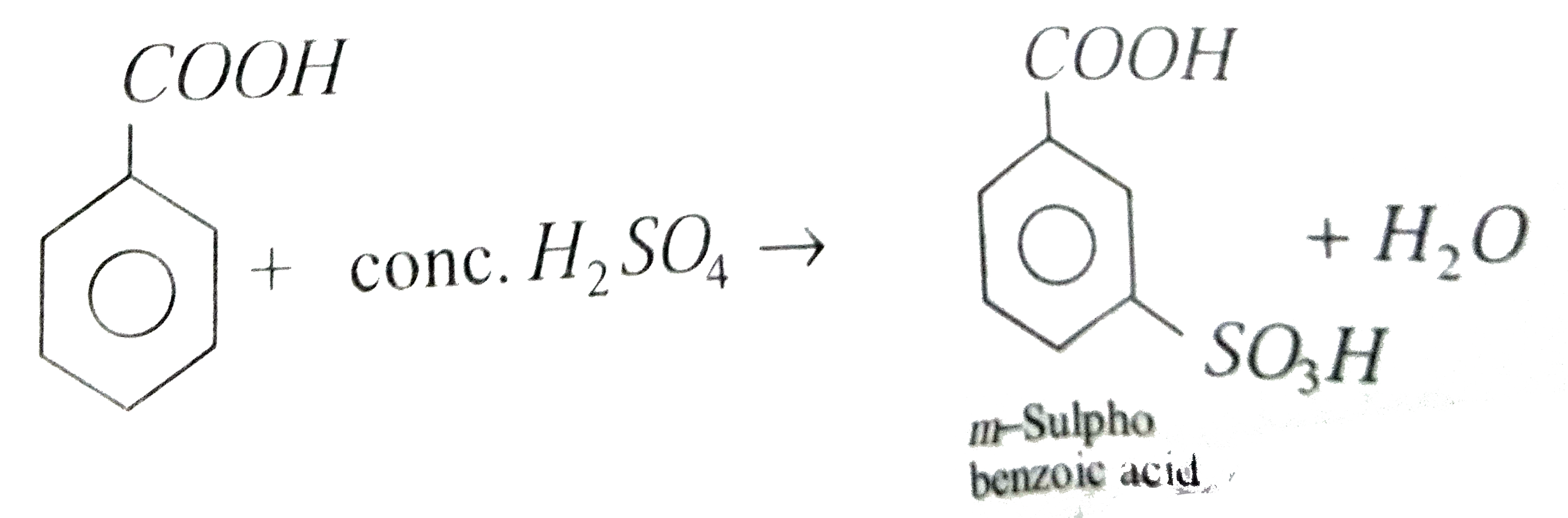
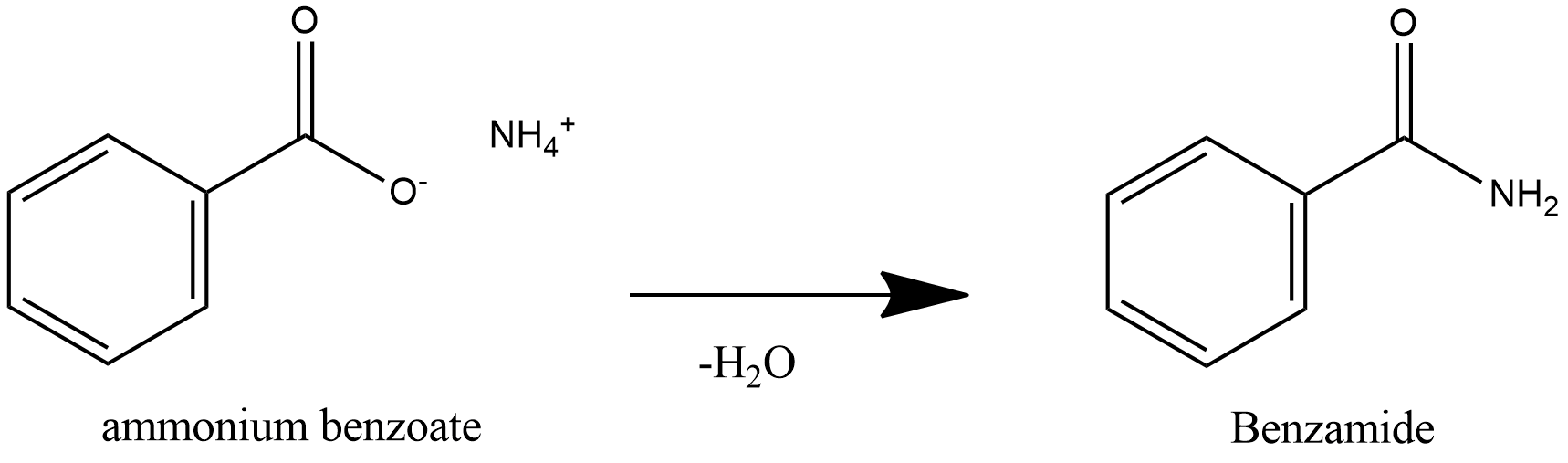
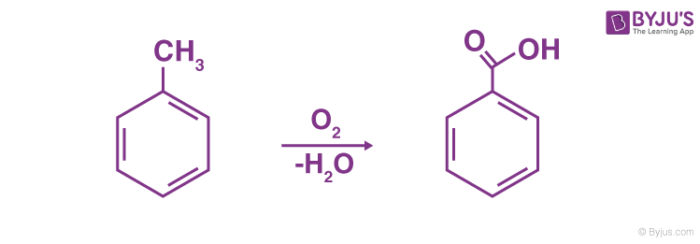
benzoic acid react with nh3
What compound is produced by the reaction of benzoic acid with ammonia?
Ammonium benzoate, a white powder-like substance, is the ammonium salt of benzoic acid.
This compound is prepared by the reaction of benzoic acid and ammonia.Ammonia is stable but very reactive.
Stable to heat and light.
Reacts violently and explosively with oxidizing gases such as chlorine, bromine, and other halogens.
Reacts explosively with hypochlorites such as bleach.
Can NH3 react with acid?
Ammonia is a weak basic in nature and sulphuric acid is a strong acid.
So, ammonia reacts with sulphuric acid leading to the formation of a salt of weak base and strong acid.
Ammonia reacts with Sulphuric acid ( H 2 SO 4 ) to give Ammonium sulphate ( ( NH 4 ) 2 SO 4 ) as a product.
What is the action of NH3 on carboxylic acid?
When carboxylic acids react with ammonia, they produce an ammonium salt.
For example, the reaction between ethanoic acid and ammonia produces a colourless solution of ammonium ethanoate with no other products.

Benzoic acid i) NH3 ---

Reactions of Benzoic Acid With Mechanism BP 301T L~22

Type of Reaction for NH3 + HCl = NH4Cl
|
Exam Paper Topics for CHEM1002 November 2012
2 nov. 2012 Write the equation for the reaction that occurs when benzoic acid reacts with ammonia? Marks. 5. C6H5COOH(aq) + NH3(aq) ? C6H5COO–(aq) + ... |
|
Reactions of molecular crystals with gases. I. Reactions of solid
Abstract: Single crystals of carboxylic acids such as benzoic and 4-chlorobenzoic acid have been found to react quantitatively with ammonia gas to give |
|
The Pyrolysis of Amides
The carboxylic acid in turn |
|
CHEM1102 2014-J-4 June 2014 Calculate the pH of a 0.010 M
4 juin 2014 Write the equation for the reaction that occurs when benzoic acid reacts with ammonia? Marks. 5. C6H5COOH(aq) + NH3(aq) ? C6H5COO–(aq) + ... |
|
Sample Exercise 17.1 Calculating the pH When a Common Ion is
Calculate the pH of a buffer composed of 0.12 M benzoic acid and 0.20 M sodium The acid HCl and base NH3 react transferring a proton from HCl to NH3 |
|
Reaction of ammonia gas with crystalline benzoic and related acids
found that benzoic acid and a number of related car- boxylic acids react either as powders or as single crystals |
|
Some Mechanistic Aspects of the Reaction of Anhydrides with
zoic acid a product of the reaction |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
be benzoic acid and compound (A) should therefore |
|
Gold and Silver-Catalyzed Reductive Amination of Aromatic
10 juin 2021 ABSTRACT: The reductive amination of benzoic acid and its derivatives would ... reacted with ammonia to form benzylamine.23 This method is. |
|
WCH15/01
(a) The bonding within the complex [Cu(NH3)4(H2O)2]2+ is 6 Methylbenzene reacts with a mixture of concentrated nitric acid and concentrated. |
|
Exam Paper Topics for CHEM1002 November 2012
2 nov 2012 · Write the equation for the reaction that occurs when benzoic acid reacts with ammonia? Marks 5 C6H5COOH(aq) + NH3(aq) → C6H5COO–(aq) |
|
Direct preparation of primary amides from carboxylic acids and urea
a mixture of benzoic acid and imidazole also confirmed the above result In addition, the effect of different ammonia sources on the reaction progress was |
|
Montmorillonite Clay Catalysis: Conversion of - ScienceDirect
The reaction of methyl benzoate [1] with NH3 was stud- ied over Montmorillonite C Benzonitrile [2], benzoic acid [3], benzamide [4], N-methylbenzamide [5] |
|
Carboxylic acid part 2 - Shia College, Lucknow
(iii) Formation of acid amides/ Reaction with ammonia Acids + NH3 + COONH4 heat OH2 CONH2 Benzoic acid Ammonium benzoate Benzamide |
|
8 Carboxylic Acids and their Derivatives
+ 2H20 -~ RCOOH+NH3 Most reactions can be carried out in acidic or alkaline conditions, but acid hydrolysis of an ester will give an equilibrium mixture 135 |





![What is the action of ammonia [NH3] on benzoic acid? Write equation What is the action of ammonia [NH3] on benzoic acid? Write equation](https://0.academia-photos.com/attachment_thumbnails/50166800/mini_magick20190129-21943-kluls9.png?1548811486)