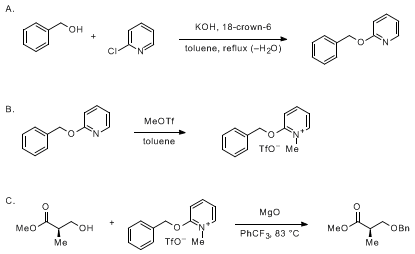
benzyl alcohol vs phenol acidity
Why are phenols called carbolic acid and phenolic acid?
Phenols are also known as carbolic acid and phenolic acid. This is because they are acidic in nature and have −OH − O H group on aromatic rings. Acidity is the measure of the amount of acid a substance contains. Acidity represents the acidic nature of a compound, solvent or solution.
Is P -nitrobenzyl alcohol more acidic than benzyl alcohol?
p -Nitrobenzyl alcohol is more acidic than benzyl alcohol, but p -methoxybenzyl alcohol is less acidic. Explain. As an Amazon Associate we earn from qualifying purchases. This free textbook is an OpenStax resource written to increase student access to high-quality, peer-reviewed learning materials.
Do phenols change the pH of a solution?
In aqueous solutions, phenols are weakly acidic and lower the pH of a solution. Sodium hydroxide can be used to fully deprotonate a phenol. Water soluble alcohols do not change the pH of the solution and are considered neutral.
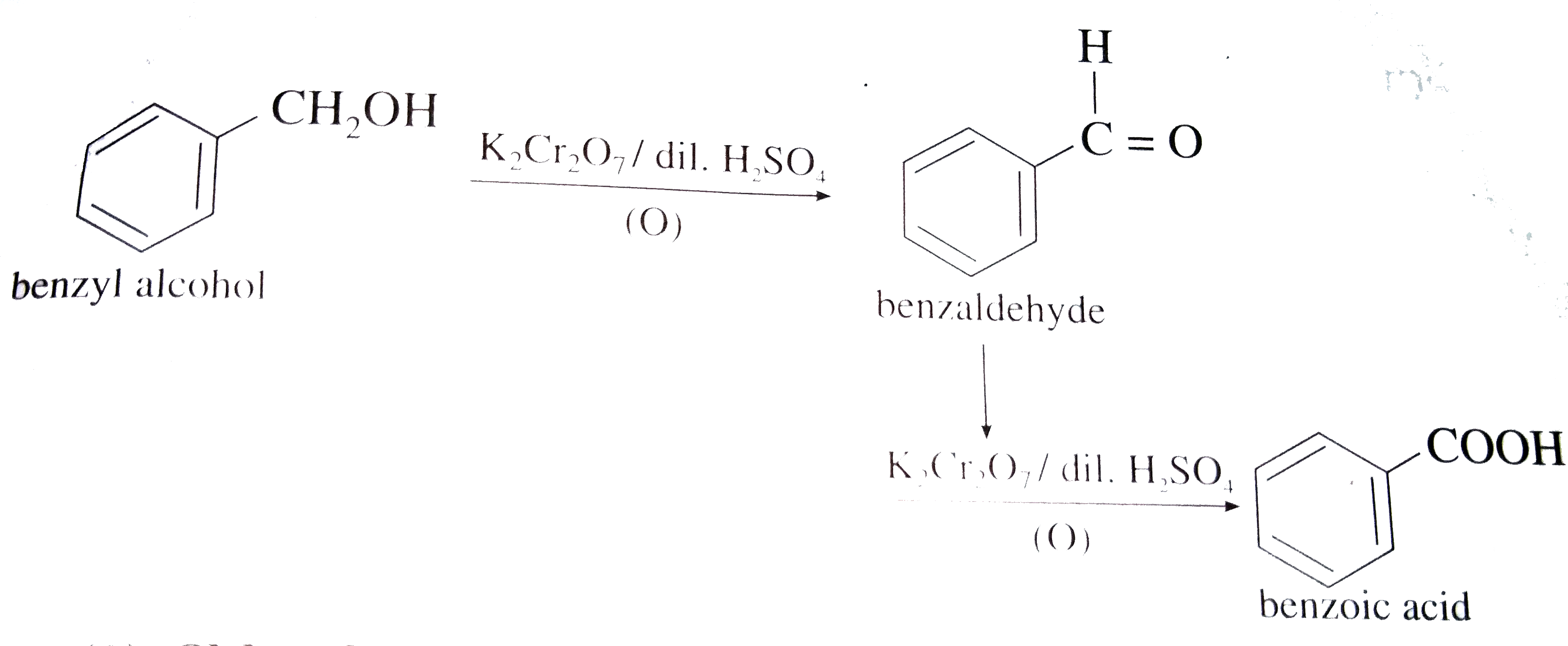
Why is phenol more acidic than benzyl alcohol?
In phenol, there is an electron-withdrawing group present, which helps in increasing the acidity of phenol by stabilizing phenoxide ions. In benzyl alcohol, there is an electron donating or electron releasing group, which reduces the acidity of phenol by destabilizing phenoxide ions. Therefore, phenol is more acidic than benzyl alcohol.

12.2 Properties of Alcohols Organic Chemistry

Alcohols Phenols n Ethers 11 I Properties Of Phenol 2 :Esterification & Reactions of Benzene Ring

Ch#18Lec#5 Acidity of Phenol Comparisonof Acidity of phenol alcohols and Carboxylic acid
|
Chapter 17: Alcohols and Phenols
17 3: Properties of alcohols and phenols: acidity and basicity: Like water alcohols are weak Brønsted bases and weak Brønsted acids |
|
Chapter 24: Phenols Alcohols contain an OH group bonded
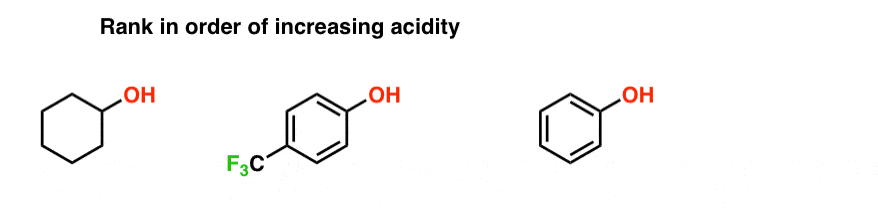
Electron-withdrawing substituents make a phenol more acidic by stabilizing the phenoxide ion through delocalization of the negative charge and through inductive |
|
Chapter_7-alcohols-phenols-145pdf
Both alcohol and phenol show acidic property to a certain degree since the hydrogen in the hydroxy group (-OH) can be removed by a base as a proton ?Alcohols |
|
Alcohols Phenols and Ethers - NCERT
From the above data you will note that phenol is million times more acidic than ethanol Arrange the following compounds in increasing order of their acid |
|
CHEM1902/4 2012-N-13 November 2012 • Benzoic acid H benzyl
13 nov 2012 · Benzoic acid H benzyl alcohol I and phenol J are shown below The pKa values of these three compounds are 15 2 9 9 and 4 2 |
|
Chapter 3 Alcohols Phenols and Ethers - Angelo State University
Chapter Objectives: • Learn to recognize the alcohol phenol and ether functional groups • Learn the IUPAC system for naming alcohols phenols |
|
Principles of Drug Action 1 Spring 2005 Alcohols
Solubility and Other Physicochemical Properties of Alcohols and Phenols benzyl alcohols can undergo elimination reactions particularly in acid |
|
Phenols
that both alcohols and phenols can be converted into ethers and esters In most of benzenesulfonic acid benzoic acid benzyl alcohol phenol |
|
Chapter 17: Alcohols and Phenols
Phenols contain an OH group connected to a carbon of a benzene ring 77 O H H O R H O 17 3: Properties of alcohols and phenols: acidity and basicity: |
|
Alcohol and Phenol Tutorial
OH OH Primary Alcohol Primary Alcohol (Benzyl Alcohol) (Ethanol) Phenol Alcohols are considered to be relatively non-acidic and non-basic compounds, |
|
CHEM1902/4 2012-N-13 November 2012 • Benzoic acid H, benzyl
13 nov 2012 · Benzoic acid H, benzyl alcohol I and phenol J are shown below The pKa values of these three compounds are 15 2, 9 9 and 4 2, but not in that order |
|
Alcohols, Phenols and Ethers - NCERT
Phenol is obtained by acidification of sodium phenoxide so produced (Unit 10, Class XII) 2 From benzenesulphonic acid Benzene is sulphonated with oleum |
|
LABORATORY MANUAL FOR ORGANIC CHEMISTRY I
22 Experiment 3: Synthesis of benzoic acid and benzyl alcohol 27 Experiment 4 : Nitration of phenol 33 Experiment 5: Synthesis of triphenylmethanol 37 |
|
Ethanol - Nature
solvent properties, acidity of the reaction process media and temperature on rate BPE acidolysis in ethanol were identified to be phenol (Ph) and benzyl ethyl |
|
NCERT Exemplar Problems Class 12 Chemistry Alcohols, Phenols
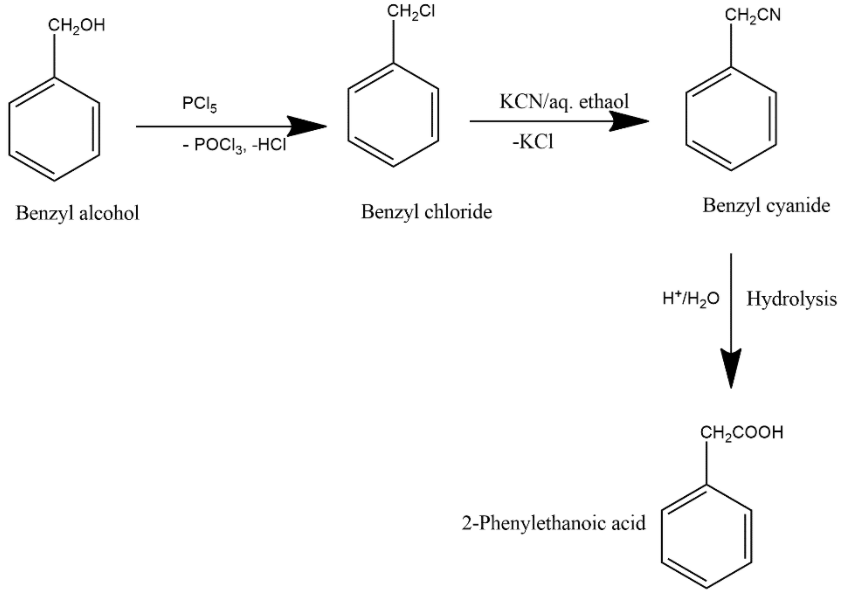
NaOH, benzyl chloride shows nucleophilic substitution reaction to give benzyl alcohol Question 2 Solution: Solution: (d) Alcohols are less acidic than phenol |
|
A Simple and Mild Acylation of Alcohols, Phenols, Amines - CORE
a Yields were expressed from GLC data Table 4 WD-Acylation of phenol and benzyl alcohol with acid anhydridesa Acid anhydride Yield of phenyl ester |
|
Class 12 Chemistry Alcohol Phenols and Ethers - Studymate
(d) Benzyl alcohol 4 The product of acid-catalysed hydration of 2- phenylpropene is (a) 3-phenyl-2-propanol (b) 1-phenyl-2-propanol (c) 2-phenyl -2-propanol |
|
Alcohols and Phenols Chem 145
Acidity of Alcohols and Phenols carbon atom(s) of benzene ring by hydroxyl group O Alcohols and phenols may be viewed as organic derivatives of water |

![PDF] Benzylation of Phenol with Benzyl Alcohol in the Presence of PDF] Benzylation of Phenol with Benzyl Alcohol in the Presence of](https://0.academia-photos.com/attachment_thumbnails/64233996/mini_magick20200820-7934-34a2qr.png?1597986356)