ich q10 pharmaceutical quality system
|
ICH guideline Q10 on pharmaceutical quality system - Step 5
ICH Q10 describes one comprehensive model for an effective pharmaceutical quality system that is based on International Standards Organisation (ISO) quality |
|
Guidance for Industry Q10 Pharmaceutical Quality System
Regional GMP requirements the ICH guidance “Q7 Good Manufacturing Practice Guidance for. Active Pharmaceutical Ingredients |
|
ICH Q10 Pharmaceutical Quality System (PQS)
ICH Q9 Quality Risk Management. ? ICH Q10 Pharmaceutical Quality System. ? Vision. ? Move from regulatory guidance to scientific guidance. |
|
Pharmaceutical Quality System Q10
4 jui. 2008 This Guideline has been developed by the appropriate ICH Expert Working Group and has been subject to consultation by the regulatory parties ... |
|
ICH Q10 - Pharmaceutical Quality System
Q10 Pharmaceutical Quality System. (PQS). ? Design and Content considerations are: ? PQS should be well structured and clear / consider. |
|
PHARMACEUTICAL QUALITY SYSTEM
PHARMACEUTICAL QUALITY SYSTEM. Q10. Current Step 4 version dated 4 June 2008. This Guideline has been developed by the appropriate ICH Expert Working Group. |
|
Implementation of Q8 Q9 & Q10
ICH Q10 Pharmaceutical Quality System. The PQS covers the entire lifecycle of a product including : Pharmaceutical. Development. Commercial. Manufacturing. |
|
THE EXTENT OF POPULATION EXPOSURE TO ASSESS CLINICAL
9 mai 2007 DRAFT CONSENSUS GUIDELINE. PHARMACEUTICAL QUALITY SYSTEM. Q10 ... Relationship of ICH Q10 to Regional GMP Requirements. |
|
GMP Guide Chapter 1 Q10 implementation final
31 jan. 2013 to align with the concepts and terminology described in the ICH Q10 tripartite guideline on Pharmaceutical Quality System. The title of the ... |
|
Introduction to Q10 Pharmaceutical Quality System
9 mai 2007 h i i. i t. t d. h t lit emphasizing an integrated approach to quality risk management and science.” Brussels July 2003. Page 5. ICH : a 3 ... |
|
ICH HARMONISED TRIPARTITE GUIDELINE
ICH Q10 describes one comprehensive model for an effective pharmaceutical quality system that is based on International Standards Organisation (ISO) quality concepts includes applicable Good Manufacturing Practice (GMP) regulations and complements ICH Q8 “Pharmaceutical Development” and ICH Q9 “Quality Risk Management” |
|
ICH guideline Q10 on pharmaceutical quality system - Step 5
ICH Q10 describes one comprehensive model for an effective pharmaceutical quality system that is based on International Standards Organisation (ISO) quality concepts includes applicable Good Manufacturing Practice (GMP) regulations and complements ICH Q8 “Pharmaceutical Development” and ICH Q9 “Quality Risk Management” |
|
Guidance for Industry Q10 Pharmaceutical Quality System - Food and D
ICH Q10 to Requirements Regulatory Enablers Knowledge Quality Approaches Management Risk Design and Management Content ISO Standards and ICH Q7 Considerations Quality Manual 9 ontent:§2– Management Responsibility ManagementCommitment QualityPolicyQualityPlanning ResourceManagement InternalCommunication ManagementReview Managementof |
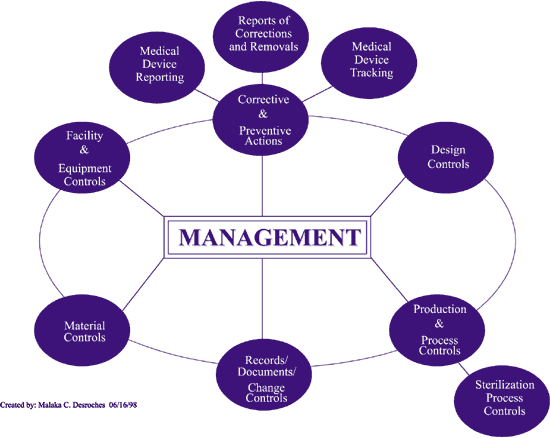
What is ICH Q10?
- Regional GMP requirements, the ICH guidance “Q7 Good Manufacturing Practice Guidance for Active Pharmaceutical Ingredients,” and ISO quality management system guidelines form the foundation for ICH Q10. To meet the objectives described below, ICH Q10 augments GMPs by describing specific quality system elements and management responsibilities.
What is the Q10 model?
- However, the Q10 model’s intent is to enhance these elements to promote the lifecycle approach to product quality. These four elements are: Process performance and product quality monitoring system Corrective action and preventive action (CAPA) system Change management system Management review of process performance and product quality
What is a Pharmaceutical Quality System (PQS)?
- (ICH Q10) Pharmaceutical Quality System (PQS): Management system to direct and control a pharmaceutical company with regard to quality. (ICH Q10 based upon ISO 9000:2005) Preventive Action: Action to eliminate the cause of a potential non-conformity or other undesirable potential situation.
Is the Q10 model required under regional GMP regulations?
- The elements described below might be, required in part under regional GMP regulations. However, the Q10 model’s intent is to enhance these elements in order to promote the lifecycle approach to product quality.
|
ICH guideline Q10 on pharmaceutical quality system - Step 5
ICH Q10 describes one comprehensive model for an effective pharmaceutical quality system that is based on International Standards Organisation (ISO) quality |
|
ICH Q10 Pharmaceutical Quality System
flexible regulatory approaches ➢ ICH Q8 Pharmaceutical Development ➢ ICH Q9 Quality Risk Management ➢ ICH Q10 Pharmaceutical Quality System |
|
ICH Q10 - Pharmaceutical Quality System
ICHQ10 8 ICH Q10 - Pharmaceutical Quality System ▫ The objective is to: ▫ Achieve product realisation ▫ Establish and maintain a state of control |
|
Quality Management Systems ICH Q10 - HPRA
27 sept 2012 · Basis for Pharmaceutical System • ICH Q10 Summary • Pharmaceutical Quality System Elements • Management Responsibilities • Enablers |
|
ICH Q10 Pharmaceutical Quality System (PQS) - DCVMN
Wednesday Morning ▫ Introduction to ICH Q10 – Pharmaceutical Quality System ▫ Management of Deviations/Investigations and CAPA Wednesday Morning |
|
ICH Q 10 Pharmaceutical Quality System PQS
PQS (Q10) Quality Risk Management (Q9) Pharmaceutical Development (Q8 Q8R) An ICH Vision for Pharmaceutical Quality Quality System Existing |
|
ICH Q10/WHO TRS A Regulatory Perspective - Pharmexcil
is manufactured under a quality management system and meets the ICH Q10 ▫ Pharmaceutical Quality System or PQS ▫ Current is Step 4, Version dated |
|
ICH Q10 - cloudfrontnet
WHITE PAPER PHARMA BIOTECH The ICH Q10 Guideline 'Pharmaceutical Quality System' was signed off as a Step 4 document at the ICH meeting in |
|
Implementing a Modern Pharmaceutical Quality System (ICH Q10 WG)
9 oct 2014 · ➢ In 2008, the International Conference on Harmonization published the ICH tripartite guideline titled, Pharmaceutical Quality System Q10 The |