ich q9
|
ICH guideline Q9 on quality risk management - Step 5
EMA/CHMP/ICH/24235/2006. Committee for Human Medicinal Products. ICH guideline Q9 on quality risk management. Step 5. Transmission to CHMP. June 2005. |
|
Gggmmmpppeye
ICH Q9 Quality Risk Management. GI002c gggmmmpppeye www.gmpeye.co.kr. 3. QUALITY RISK MANAGEMENT. ICH Harmonised Tripartite Guideline. |
|
ICH E10
2005. 11. 9. ICH HARMONISED TRIPARTITE GUIDELINE. QUALITY RISK MANAGEMENT. Q9 ... Q9. Approval by the Steering Committee under Step 2 and. |
|
ICH guideline Q9 (R1) on quality risk management
2021. 11. 18. 16 December 2021. EMA/CHMP/ICH/24235/2006. Committee for Medicinal Products for Human Use. ICH guideline Q9 (R1) on quality risk management. |
|
Q9(R1)
ICH HARMONISED GUIDELINE. QUALITY RISK MANAGEMENT. Q9(R1). Draft version. Endorsed on 18 November 2021. Currently under public consultation. |
|
QUALITY RISK MANAGEMENT
2005. 11. 9. ICH HARMONISED TRIPARTITE GUIDELINE. QUALITY RISK MANAGEMENT. Q9 ... recommendation for adoption to the three ICH regulatory bodies. |
|
Quality Risk Management ICH Q9(R1)
2021. 11. 26. The ICH Q9 Guideline has been revised to address the following: •. The QRM principles and framework of ICH Q9 have been instrumental in ... |
|
ICH-Endorsed Guide for ICH Q8/Q9/Q10 Implementation Document
2011. 12. 6. The introduction of ICH Q9 states that: “…the protection of the patient by managing the risk to quality should be considered of prime importance ... |
|
Quality Risk Management ICH Q9
Disclaimer: The ICH Q9 briefing pack is offered as a supplementary explanation of the material in ICH Q9. It was prepared by some members of. |
|
Final Concept Paper ICH Q9(R1) - Quality Risk Management
2020. 11. 13. Limited and specific adjustments would be made to specific chapters and annexes of the current ICH. Q9 Guideline on Quality Risk Management (QRM) ... |
|
QUALITY RISK MANAGEMENT Q9(R1) - ICH
ICH Q9(R1) Guideline 1 1 1 INTRODUCTION 2 Risk management principles are effectively utilized in many areas of business and government 3 including finance insurance occupational safety public health pharmacovigilance and by 4 agencies regulating these industries In the pharmaceutical sector the principles and framework |
|
QUALITY RISK MANAGEMENT Q9(R1)
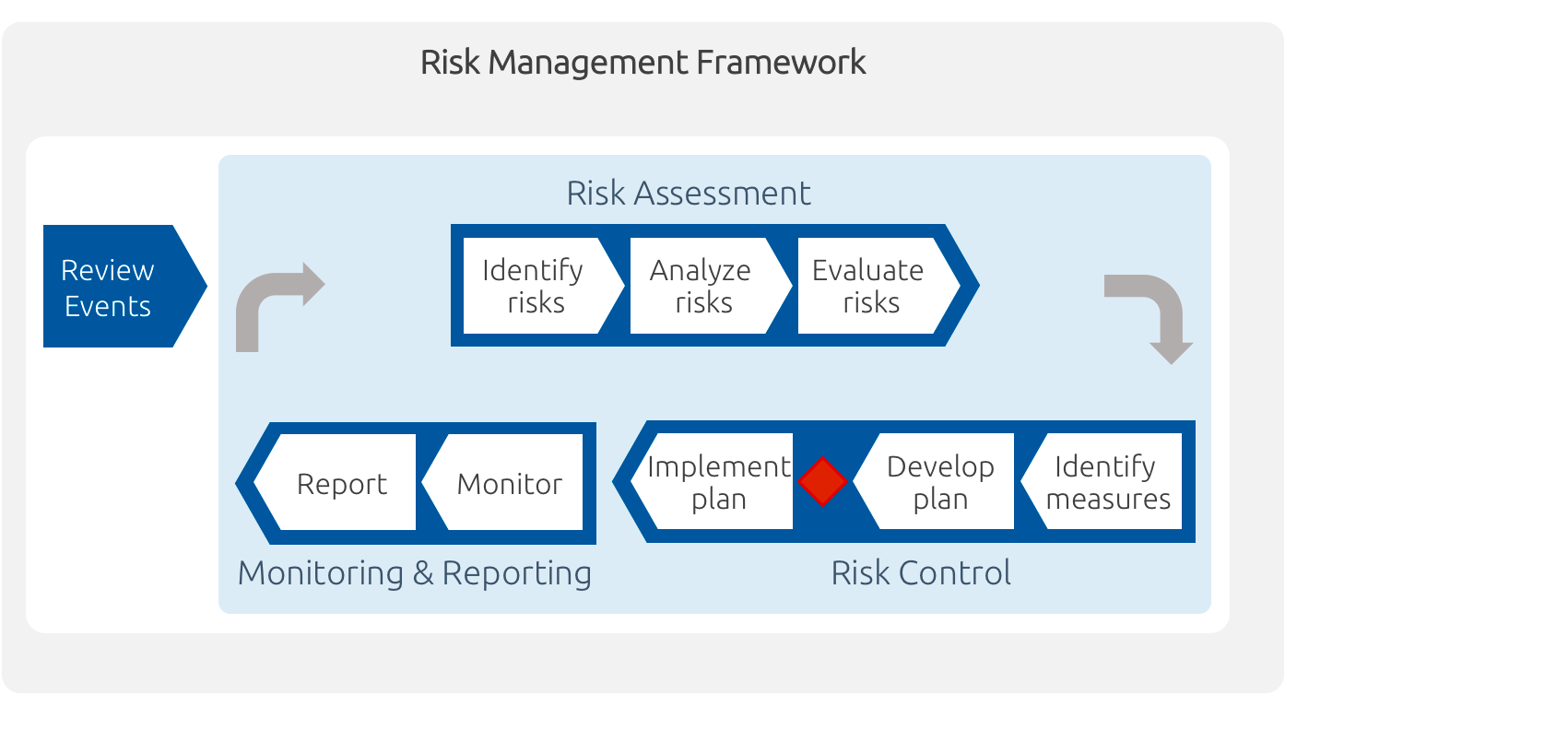
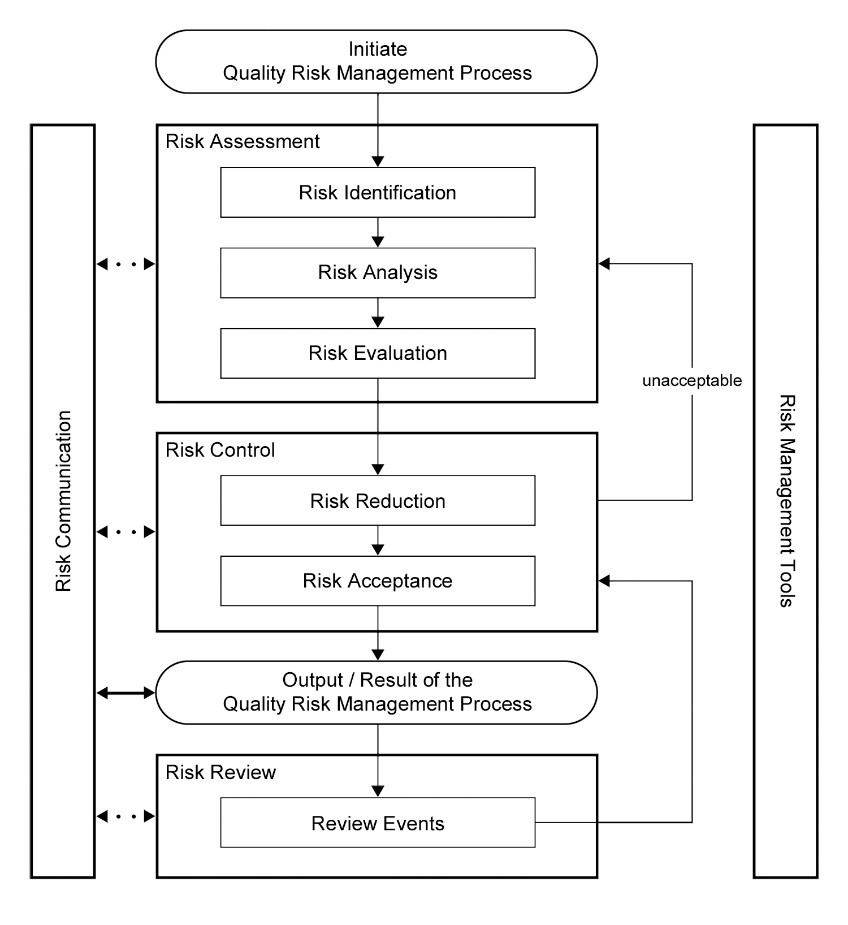
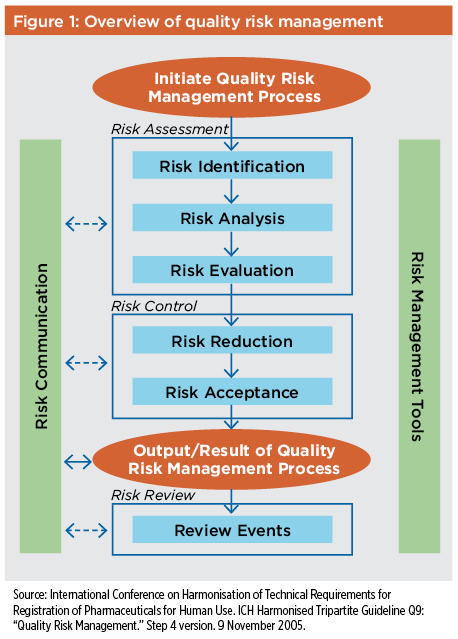
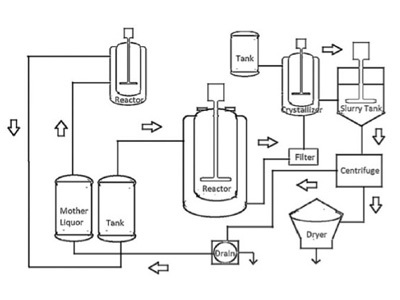
ICH Q9(R1) Guideline 3 4 GENERAL QUALITY RISK MANAGEMENT PROCESS Quality risk management is a systematic process for the assessment control communication and review of risks to the quality of the drug (medicinal) product across the product lifecycle A model for quality risk management is outlined in the diagram (Figure 1) |
|
Federal Register /Vol 88 No 86/Thursday May 4 2023
May 4 2023 · guidance for industry entitled ‘‘Q9(R1) Quality Risk Management ’’ The guidance was prepared under the auspices of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) The guidance is a targeted revision of the 2006 guidance for industry ‘‘Q9 Quality Risk |
|
Searches related to ich q9 filetype:pdf
ICH Q9 together with “Pharmaceutical development” [ICH Q8 Q8(R1)] and “Quality systems” [ICH Q10] provides opportunity for a revised optimised and less restrictive regulatory paradigm Based on scientific knowledge Enables continual improvement Greater transparency and efficiency Focusing on things that add value for patients |
What is Ich Q9?
- In the pharmaceutical sector, the principles and framework of ICH Q9, coupled with the official ICH training material that supports this guideline, are instrumental in enhancing the application of effective quality risk management by industry and regulators.
What is Q9 quality risk management?
- 1 Guidance for Industry1 Q9 Quality Risk Management This guidance represents the Food and Drug Administration's (FDA's) current thinking on this topic. It does not create or confer any rights for or on any person and does not operate to bind FDA or the public.
Is Ich liable if a document is provided as is?
- The document is provided "as is" without warranty of any kind. In no event shall the ICH or the authors of the original document be liable for any claim, damages or other liability arising from the use of the document. The above-mentioned permissions do not apply to content supplied by third parties.
|
ICH guideline Q9 on quality risk management - European Medicines
These aspects include development, manufacturing, distribution, and the inspection and submission/review processes throughout the Page 4 ICH guideline Q9 |
|
ICH Q9 - AFMPS
L'objet du présent document est de proposer une approche systématique de la gestion du risque qualité Il sert de base ou de document ressource |
|
Final Concept Paper ICH Q9(R1) - Quality Risk Management
ICH Q9(R1) - Quality Risk Management Endorsed by the Management Committee on 13 November 2020 Type of Harmonisation Action Proposed |
|
Quality Risk Management ICH Q9
prepared by some members of the ICH Q9 EWG for example only; not an official policy/guidance July 2006, slide 1 ICH Q9 QUALITY RISK MANAGEMENT |
|
ICH Q9 - Grand Lyon économie
Gestion du risque qualité (ICH Q9) des procédés aseptiques OBJECTIFS • Comprendre les bénéfices à appliquer le QRM dans toutes les étapes de la vie du |
|
ICH Q8 - Académie Nationale de Pharmacie
2 mar 2011 · De ce fait, les notes explicatives IQH Q8, Q9 et Q10 virent le jour ICH Quality Vision “Develop a harmonized pharmaceutical quality system |
|
Quality Risk Management ICH Q9 - PMDA
The presentation does not represent official guidance or policy of authorities or industry Annex I: Methods Tools prepared by some members of the ICH Q9 EWG |
|
Formation en Gestion de risques–ICHQ9, FMEA - PBE Expert Inc
2 avr 2018 · Après un rappel des exigences réglementaires (ICH Q9, BP, ISPE): 3 L' approche Risk Management sera étayée et pratiquée à travers plusieurs |






![ICH Q9 Quality Risk Management - [PDF Document] ICH Q9 Quality Risk Management - [PDF Document]](https://i1.wp.com/manoxblog.com/wp-content/uploads/2018/11/screenshot-www.pda_.org-2018-11-07-08-22-17-059.jpeg?resize\u003d758%2C452\u0026ssl\u003d1)









![ICH Q9 Quality Risk Management - [PDF Document] ICH Q9 Quality Risk Management - [PDF Document]](https://ich.org/assets/images/header-img.png)









![ICH Q9 (eng) - [PDF Document] ICH Q9 (eng) - [PDF Document]](https://i1.rgstatic.net/publication/287673416_A_practical_risk-based_approach_to_assess_vial's_dimensions_deviations_effect_on_the_aseptic_filling_processing_according_to_ICH_Q9_guideline/links/5bb35db7299bf13e605a46fb/largepreview.png)








![ICH Q9 (eng) - [PDF Document] ICH Q9 (eng) - [PDF Document]](https://i1.rgstatic.net/publication/330744090_Quality_Risk_Management_Framework_Guidance_for_Successful_Implementation_of_Risk_Management_in_Clinical_Development/links/5c622b9592851c48a9cd4ada/largepreview.png)






![ICH Q9 (eng) - [PDF Document] ICH Q9 (eng) - [PDF Document]](https://ars.els-cdn.com/content/image/1-s2.0-S1110093117300704-gr3.jpg)













![ICH Q9 Quality Risk Management - [PDF Document] ICH Q9 Quality Risk Management - [PDF Document]](https://demo.fdocuments.in/img/742x1000/reader018/reader/2020012121/5a71852c7f8b9ab6538cd1fc/r-1.jpg?t\u003d1610150185)

![PDF] Support de formation sur le management de la qualite et PDF] Support de formation sur le management de la qualite et](https://d3i71xaburhd42.cloudfront.net/fe9ffff3216599f696c79602b0bd54e9eacda1cf/5-Figure1-1.png)
![PDF] Analysis and critical review of ICH Q8 Q9 and Q10 from a PDF] Analysis and critical review of ICH Q8 Q9 and Q10 from a](https://demo.dokumen.tips/img/380x512/reader018/reader/2020011511/5870213f1a28ab7f428b7fd3/r-1.jpg)





![PDF] PQLI Application of Science- and Risk-based Approaches (ICH PDF] PQLI Application of Science- and Risk-based Approaches (ICH](https://img.yumpu.com/49776524/1/500x640/aseptic-processing-risk-management-a-reviewpdf-fmea-.jpg)







![PDF] Analysis and critical review of ICH Q8 Q9 and Q10 from a PDF] Analysis and critical review of ICH Q8 Q9 and Q10 from a](https://0.academia-photos.com/attachment_thumbnails/34319836/mini_magick20190324-13250-ud51o.png?1553424487)