benzene pdf
|
Benzene Properties CAS No 71-43-2
Benzene is known to be a human carcinogen based on suficient evi-dence of carcinogenicity from studies in humans |
|
Benzene
71-43-2 Hazard Summary Benzene is found in the air from emissions from burning coal and oil gasoline service stations and motor vehicle exhaust Acute (short-term) inhalation exposure of humans to benzene may cause drowsiness dizziness headaches as well as eye skin and respiratory tract irritation and at high levels unconsciousness |
|
113: A Resonance Picture of Bonding in Benzene
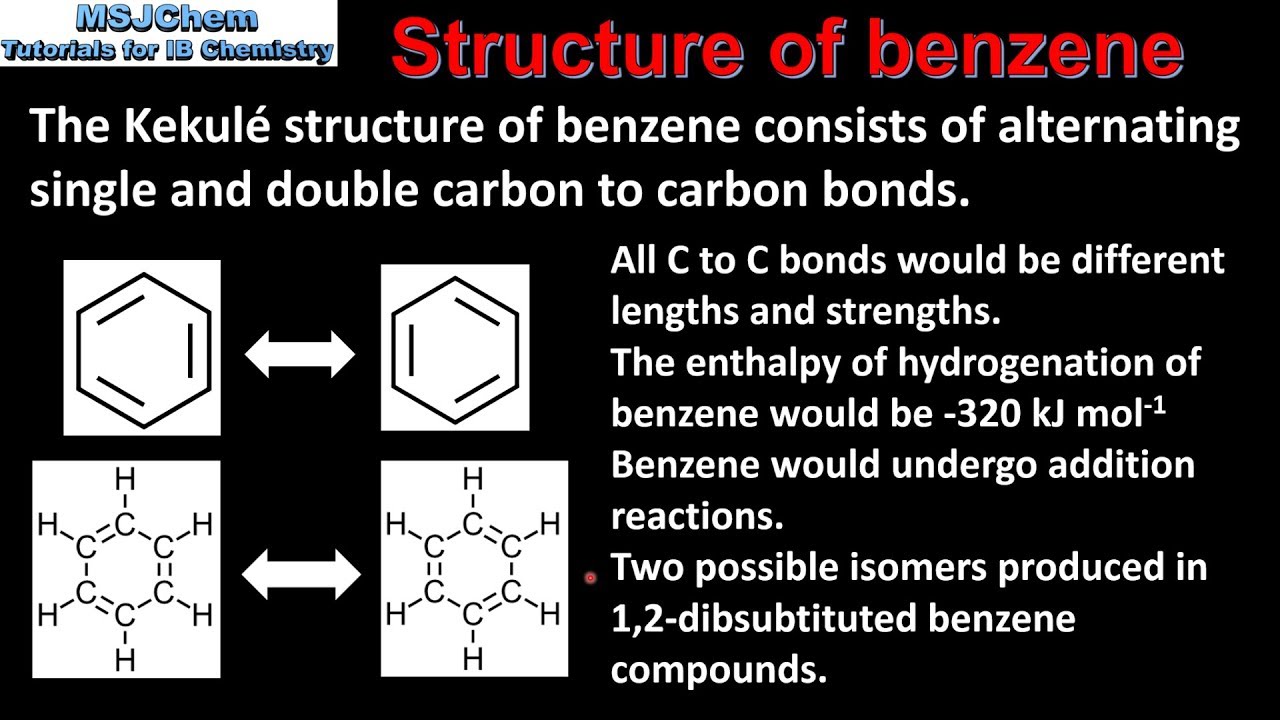
Benzene is a planar hexagonal cyclic hydrocarbon The C–C–C bond angles are 120° = sp2 hybridized Each carbon possesses an unhybridized p-orbital which makes up the conjugated π-system The six π-electrons are delocalized through the π-system |
|
Organic Chemistry II / CHEM 252 Chapter 14 – Aromatic Compounds
• The Stability of Benzene • Benzene is much more stable than the cyclohexatriene – A reasonable prediction for the heat of hydrogenation of hypothetical cyclohexatriene is -360 kJ mol-1 (3 x cyclohexene -120 kJ mol-1) – The heat of hydrogenation for benzene is -280 mol-1 152 kJ mol-1 more stable than hypothetical cyclohexatriene |
|
Reactions of Benzene & Its Derivatives
Chapter 22 1 Reactions of Benzene The most characteristic reaction of aromatic compounds is substitution at a ring carbon: Halogenation: H + Cl2 FeCl3 Cl + HCl Chlorobenzene Nitration: H2 SO4 H + HNO3 NO2 + H2 O Nitrobenzene Reactions of Benzene Sulfonation: SO3 H2 SO4 SO3 H Benzenesulfonic acid Alkylation: H + RX A lX3 R + HX An alkylbenzene |
|
CHAPTER 26: Benzene and Its Compounds
Benzene is a hexagonal and planar molecule with the formula C6H6 In benzene each carbon atom undergoes sp2 hybridisation to give three hybrid orbitals and an unchanged p orbital which is perpendicular to the plane containing the three hybrid orbitals Two of the hybrid orbitals overlap with two other hybrid orbitals of carbon |
Why is benzene a stable molecule?
The six electrons from each p orbital are then delocalised around the ring. A delocalised electron does not belong to any carbon atom. This delocalisation of electrons gives benzene a relatively stable structure. In a benzene molecule, all the C-C bonds are identical, with a bond length of 140 pm.
What is an example of aromatic benzene?
Examples of aromatic Benzene is a hexagonal and planar molecule with the formula C6H6. to give three hybrid orbitals and an unchanged p orbital which is perpendicular to the plane containing the three hybrid orbitals. atoms and the other overlaps with the orbital of hydrogen.
What is benzene used for?
Benzene is used as a constituent in motor fuels; as a solvent for fats, waxes, resins, oils, inks, paints, plastics, and rubber; in the extraction of oils from seeds and nuts; and in photogravure printing. It is also used as a chemical intermediate.
Is benzene soluble in water?
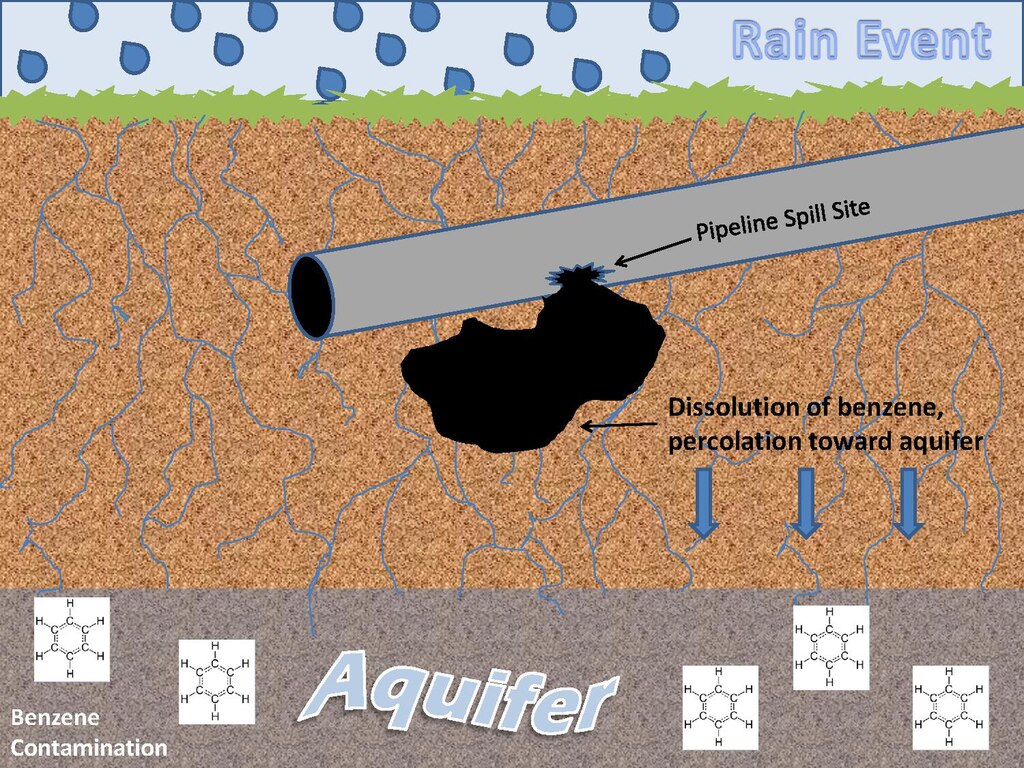
It is only slightly soluble in water, but it is miscible with alcohol, ether, chloroform, carbon disulfide, acetone, oils, carbon tetrachloride, gla-cial acetic acid, and most other organic solvents. Benzene is highly flammable (Akron 2009). Physical and chemical properties of ben-zene are listed in the following table. Source: HSDB 2009.
Carcinogenicity
Benzene is known to be a human carcinogen based on suficient evi-dence of carcinogenicity from studies in humans. ntp.niehs.nih.gov
Coast Guard, Dept. of Homeland Security
Comprehensive regulations have been established for safe transport of benzene on ships and barges. ntp.niehs.nih.gov
Department of Transportation (DOT)
Benzene is considered a hazardous material, and special requirements have been set for marking, labeling, and transporting this material. ntp.niehs.nih.gov
Emergency Planning and Community Right-To-Know Act
Toxics Release Inventory: Listed substance subject to reporting requirements. ntp.niehs.nih.gov

Naming benzene derivatives introduction Aromatic Compounds Organic chemistry Khan Academy

Benzene and its derivatives L-1 Unit-1 Organic Chemistry 2 B.pharma 3rd sem Carewell Pharma

Naming benzene derivatives Aromatic Compounds Organic chemistry Khan Academy
|
Aromatique
12 jan 2017 · - Dans la pratique on chauffe le benzène dans un mélange d'acide nitrique et d'acide sulfurique concentrés Problème 15 2 (S) ? Problème |
|
1 La molécule de benzène 111 Formule brute - AC Nancy Metz
: la force de la liaison est celle d'une liaison éthylénique un peu affaiblie 1 1 5 Données thermodynamiques Page 1 sur 30 LE BENZENE LES AROMATIQUES |
|
Benzène - Lab Cerba
Le benzène est un liquide incolore volatil inflammable d'odeur caractéristique C'est un hydrocarbure aroma- tique non substitué obtenu aujourd'hui à |
|
Cours de chimie de Polytechnique
les halogenes; avec le benzène on n'aura naturellement que les chlorure d'aluminium) our le benzene C6H6 donnera successivement |
|
Benzène - INRS
Il forme des mélanges azéotropiques avec l'eau (9117 p de benzène point d'ébullition 6925 °C) des alcools et des hydrocarbures C'est un excellent solvant |
|
Hydrocarbures Aromatiques - UNF3S
3 Réactivité des dérivés du benzène 3 1 Réaction de Substitution Electrophile Aromatique Hydrocarbures aromatiques : benzène et dérivés |
|
SE sur benzenepdf - Orgapolym
SUR LE BENZÈNE ET SES DÉRIVÉS www orgapolym com Les Cahiers de Chimie Organique pour les Étudiants de Prépa des licences fondamentales et appliquées |
|
Cours-de-chimie-organique-1967-356pdf
Le plus simple hydrocarbure de cette série est le benzène C'est pourquoi on appelle encore une série aromatique "une série des dérivés du benzène" Avant les |
|
Benzene and Its Derivatives
9 7 How Do Existing Substituents on Benzene Affect Electrophilic Aromatic Substitution? 9 8 What Are Phenols? HOW TO 9 1 How to Determine Whether |
|
Benzene et composes aromatiques - Fastef
Le benzène est un carbure d'hydrogène liquide vendu parfois dans le commerce sous le C 4 LE BENZENE ET LES COMPOSES AROMATIQUES 3H 1 ère |
|
1 La molécule de benzène 111 Formule brute - AC Nancy Metz
Si le premier substituant est plus encombrant, on a beaucoup plus de para, même à température ordinaire : Page 8 sur 30 LE BENZENE LES AROMATIQUES |
|
AROMATIQUES 1 Introduction Nomenclature C C C C C C benzène
12 jan 2017 · benzène (C6H6) H H H H H H C sp2, molécule plane CH3 CH3 parfois représenté CH3 toluène F Cl I Br fluorobenzène chlorobenzène |
|
CH 5 Composés aromatiques
CH 5 Composés aromatiques 1 Structure du composé de base : le benzène 2 Nomenclature 3 Substitution électrophile aromatique |
|
Hydrocarbures Aromatiques - UNF3S
2 Réactivité du benzène 2 1 Réaction de Substitution Electrophile Aromatique 3 Réactivité des dérivés du benzène 3 1 Réaction de Substitution Electrophile |
|
Benzène - INRS
Une valeur limite d'exposition professionnelle réglementaire contraignante dans l'air des lieux de travail a été établie en France pour le benzène (article R 4412 |
|
Benzène, toluène, xylènes (BTX - Lodel
ARTICLES , Etude de la distribution atmosphérique de composés organiques volatils aromatiques: benzène, toluène, xylènes (BTX) et du dioxyde d'azote |
|
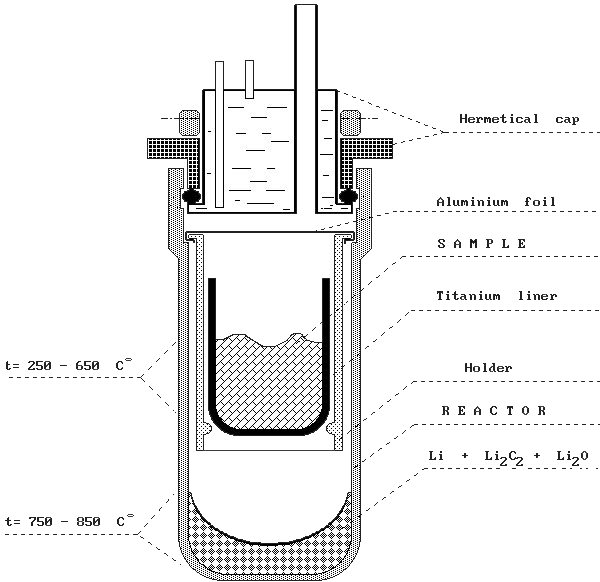
METHODE DE PREPARATION DE BENZENE TRITIE POUR LA
Le rendement est de 64 à 72 pour cent basé sur le mellitate de calcium mis en oeuvre Le benzène obtenu api "s une simple distillation dFJis une colonne Vigreux |



















![21 Benzene - [PDF Document] 21 Benzene - [PDF Document]](https://data01.123dok.com/thumb/qo/57/19my/Pki4ZiPT5VIqTD1so/cover.webp)


