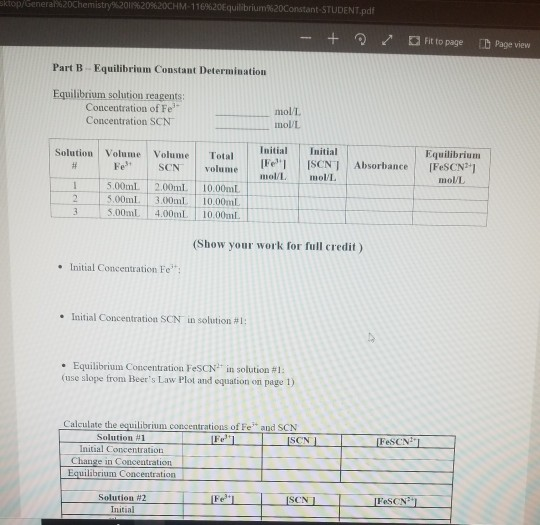
calculate the initial concentration of fe3+ and scn
|
Equilibrium Constant Determination INTRODUCTION
initial moles Fe3+ = (MFe3+)(VFe 3+) = 2 00 x 10-3 mol/L (0 0100 L) = 2 00 x 10-5 mol Fe3+ |
|
Experiment 3 Measurement of an Equilibrium Constant
Thus the concentration of Fe3+ that remains at equilibrium is the initial concentration of Fe3+ minus the concentration of Fe3+ that formed product which is the same as [FeSCN2+] eq The equilibrium concentrations of Fe3+ and for the same reasons SCN– can thus be expressed as [Fe3+] eq = [Fe 3+] initial – [FeSCN 2+] eq (3) [SCN–] eq |
Why does Fe3+ remain at equilibrium?
This is because as equation (1) shows, for every one FeSCN2+ produced, one Fe3+ ion is used up. Thus the concentration of Fe3+ that remains at equilibrium is the initial concentration of Fe3+ minus the concentration of Fe3+ that formed product, which is the same as [FeSCN2+]eq.
Can titration give a low concentration of Fe2+?
I am agreeing Prof. Zietsman, titration would give you fe concentration. otherwise, you may use XRF or ICP-EOS for its concentration, too Dear Hannah Brown. The determination of such low concentrations of Fe2+ better to determine photometrically using simple complex with o-phenanthroline with standard addition approach or calibration curve.
How do you find the initial concentration of Fe3+?
To find the initial concentration of Fe3+, use the dilution equation: (M1V 1)/V 2 = M2, where V2 = 10 mL. [FeSCN2+] at equilibrium is determined using Beer’s Law; x is the amount of FeSCN2+ created (determined experimentally). When creating the calibration plot how will you figure out the concentration of Fe SCN 2+?
What is the equilibrium concentration of fescn2+?
The absorbance of this solution is measured, and the equilibrium FeSCN2+ (aq) concentration is found to be 1.0 × 10 –3 M. How do you find the concentration of Fe3+? – To find the initial concentration of Fe3+, use the dilution equation: (M1V 1)/V 2 = M2, where V2 = 10 mL.
|
Spectroscopic Determination of an Equilibrium Con- stant
To find the initial concentration of SCN– use the dilution equation: (M1V 1)/V 2 = M2 |
|
Determination of an Equilibrium Constant Fe3+ + SCN- ? FeSCN2+
Initial concentrations: pre-equilibrium concentrations of all reactants and products a. Usually require dilution calculations and are not “bottle label” |
|
EXPERIMENT 3 - Equilibrium Constants
But before calculations can be performed with equilibrium reactions 1M Fe3+ (aq) and 1M SCN-(aq) |
|
A = abC
allows the equilibrium concentration of FeSCN2+ to be calculated. With this information and the initial concentrations of Fe3+ and SCN. |
|
General Chemistry II Lab #7 – Determination of the Equilibrium
solution and the equilibrium concentration of FeSCN2+ we can calculate the equilibrium moles Fe3+ = initial moles Fe3+ – equilibrium moles FeSCN2+. |
|
Untitled
15 avr. 2014 Accurate Solution Concentrations ... Calculations [Fe3+ initial and [SCN]initial: ... Table for equilibrium concentrations of FeSCN2+. |
|
Experiment #7 – Determination of the Equilibrium Constant for the
From the concentration of Fe3+ and SCN. – initially present in each mixture and the equilibrium concentration of FeSCN2+ calculate the concentration of |
|
Lab 13.3 Determining Kc via Colorimetry
To prepare the standard solution a very large concentration of Fe3+ will be added to a small initial concentration of SCN. |
|
Experiment 3 Measurement of an Equilibrium Constant
and SCN. – solutions of known initial concentrations. The equilibrium concentration of the FeSCN because as equation (1) shows for every one FeSCN. |
|
Lab Investigation 5 - What Is the Formation Constant for FeSCN2+?
If you know the initial. (before equilibrium) concentrations of Fe3+ and SCN– you can use a reaction table to calculate the equilibrium concentrations of |
|
Equilibrium Constant Determination INTRODUCTION Every
solution and the equilibrium concentration of FeSCN2+, we can calculate the equilibrium equilibrium moles Fe3+ = initial moles Fe3+ – equilibrium moles FeSCN2+ above, calculate Keq for solutions 1, 3 and 5 on the basis that Fe( SCN)2 |
|
Determination of an Equilibrium Constant Fe3+ + SCN - MCTCteach
Application of the dilution equation M1V1 = M2V2 is required to calculate the initial concentrations of Fe3+ and SCN- for each of the six trials Initially we |
|
Link to sample equilibrium experiment report
15 avr 2014 · Accurate Solution Concentrations Calculations [Fe3+'initial and [SCN-Jinitial : Fe3+= Table for equilibrium concentrations of FeSCN2+ |
|
The equilibrium constant for the formation of iron(III) - Chemistry
standard curve to help us determine the concentration of Fe(SCN)2+ at Example 2: Assume an initial concentration of [Fe3+] = 0 00100 M and an initial |
|
Procedure & Report - Santa Monica College
written as Fe3+ and Fe(H2O)5SCN2+ is written as FeSCN2+ (Equation 2b) Also The initial concentrations of the reactants—that is, [Fe3+] and [SCN – ] prior |
|
Equilibrium PDF
Objective: In this experiment, you will determine the equilibrium constant, Kc, for the formation of the complex Fe(SCN)2+ You will also HSCN and Fe3+ knowing their initial concentrations and the stoichiometry of the reaction Kc = [C] c[D]d |
|
Determining An Equilibrium Constant Using Spectrophotometry and
19 jui 2009 · [Fe3+ ] [SCN- ] To evaluate the equilibrium constant for this reaction, one must first determine the concentrations of the three ions There are |
|
Experiment 6: Seeing red: determination of an equilibrium constant
How do we determine the equilibrium concentrations of the free Fe3+ ion and the added to the cuvette, calculate the molar amount of SCN- initially present |
|
Calibration Curve
Fe3+ (aq) + SCN- (aq) Fe(SCN)2+ (aq) ○ Equilibrium concentrations of starting points Determine Solution Concentrations by Spectrophotometry |