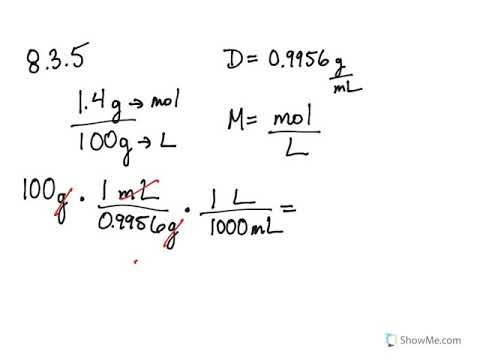
calculate the molarity of a solution that contains 15.7 g of caco3
Is molarity the same as concentration?
Molarity is not the same as concentration, although they are very similar. Concentration is a measure of how many moles of a substance are dissolved in an amount of liquid, and can have any volume units. Molarity is a type of concentration, specifically moles per liter of solution. What is the molarity of water?
What are the problems on a molarity?
The problems on a molarity usually involve the calculation of either the molar concentration of a solute, given the mass of a solute and the volume of a solution, or the mass of a solute for the desired molar concentration given the volume of a solution.
What is molarity calculator?
This molarity calculator is a tool for converting the mass concentration of any solution to molar concentration (or recalculating grams per ml to moles). You can also calculate the mass of a substance needed to achieve a desired molarity. This article will provide you with the molarity definition and the molarity formula.
How do you calculate molar concentration?
First, it can calculate the molar concentration of a solute given a solute chemical formula, the mass of the solute and the volume of the solution. Second, it can calculate the mass of a solute given a solute chemical formula, the volume of the solution and the desired molar concentration of a solute.

Molarity Molality Volume & Mass Percent Mole Fraction & Density

Molarity

Solution Stoichiometry
|
CHEMISTRY 311 - ASSIGNMENT 1
combined standard solution containing 100.0 ?g/L of NO3 H2O(s) ? CaCO3(s) a) Determine the concentration of calcium in the mineral sample in mg/g. |
|
1. CONCENTRATION UNITS A solution is a homogeneous mixture
(c) Calculate the molarity of a solution of 3.6 g of NaOH in 300 mL of solution. the volume (in mL) of a 0.355 M NaOH solution which would contain 0.200. |
|
There are a number of different ways of expressing solute
To convert from molarity to mg/L (or ppm in dilute aqueous solution) 100. g CaCO3 ... the density is 1.05 g/mL |
|
? gCaCl 11.11 CaCl mol1 soln L812.0 CaCl g 2.08 ?
What is the molarity of a solution that was prepared by dissolving 14.2 g of NaNO3 of a solution that contains 5.5 g of HCl (molar mass = 36.5 g/mol). |
|
1. (4 pts) Calculate the mass percent of calcium chloride in each of
(4 pts) What is the molarity of a solution that contains 14.0 g of ammonium bromide dissolved in enough water to make 150.0 mL of solution? Solute? Solvent? |
|
Limiting Reactant & Theoretical Yield
A strip of zinc metal having a mass of 2.00 g is placed in an aqueous solution containing 2.50 g of silver nitrate causing the following reaction to occur;. |
|
Gas Stoichiometry Chemistry 110
1] Given the equation: 2 NH3 (g) + 3 Cl2(g) ---> N2(g) + 6 HCl(g) 6] What is the molarity of a 5.00 L sodium hydroxide solution that would completely ... |
|
Untitled
Review calculations of Molarity (M) which is defined as moles of solute per liter of a 5.00 L vessel initially contains 15.7 g of H? and 294 g of Iz. |
|
Chapter 4 - Stoichiometry of Chemical Reactions
Igniting the mixture initiates a vigorous chemical reaction that rapidly is calculated by multiplying the coefficient of any formula containing that ... |
|
Untitled
of the resulting solution is 0.974 g/mL (Use total mass and density to determine the volume). The molarity of NH in the solution is. A) 0.00353 B) 0.882. |
|
1 EDTA Titration Calculations The hardness of water is due in part to
0 405 g of CaCO3 is dissolved in HCl and diluted to a volume of 250 00 mL What is the molarity of calcium ions in the solution? CaCO3(s) + 2HCl(aq) → Ca2+ |
|
1412exp47pdf
CH2-COOH It contains 6 donor atoms (underlined) that form a complex ion with Ca²+ (2) Use the standardized EDTA to determine the [CaCO3] in an unknown water sample molar ratio of Ca²+ to EDTA is 1 to 1 Ideally Use a dry 250- beaker to obtain about 120 mL of stock EDTA solution, and take this to your desk |
|
Examples w answers
To convert from molarity to mg/L (or ppm in dilute aqueous solution), multiply by the e g , total hardness is usually reported as the mass of CaCO3 that would be required to Example: A groundwater sample has been determined to contain 100 ppm Ca and 80 the density is 1 05 g/mL, calculate the molar concentration |
|
CHEMISTRY 311 - ASSIGNMENT 1
combined standard solution containing 100 0 μg/L of NO3 included in the molar mass calculation Solution: mass NaNO3 = 100 0 ug N L Converting the analyte concentration from ppm CaCO3 to Normality using an equivalent weight of |
|
1 A solution - CEE 370
To find the molarity, the molecular weight of NaHCO3 must be found Solution: a CaCl2 + Na2CO3 = CaCO3 + NaCl Elements Reactants Products Ca 1 1 fluoride concentration of 1 0 mg/L be obtained in water that contains 200 mg/L |
|
Experiment &# 11 Titration of a Commercial Antacid
The active ingredient in Tums is calcium carbonate, CaCO3, a base There are On average, a 1 3 gram tablet contains 0 5 g of titration with NaOH to figure out the amount of excess acid Then Therefore it is important to boil the solution when the carbonate reacts The equation for molarity is Molarity = moles/ volume |
|
Worksheet: Molarity Name______________ CHEMISTRY
To make a 4 00 M solution, how many moles of solute will be needed if 12 0 liters of solution are What is the molarity of a solution of HNO3 that contains 12 6 grams HNO3 in 1 0 L of Solution “A” contains 50 0 g of CaCO3 in 500 0 mL of |
|
1 Determination of calcium by Standardized EDTA Solution
Dry about 1 gram of calcium carbonate (CaCO3) in the oven for 2 hours Transfer to the standardization, since you must have enough EDTA to titrate the unknown solution Calculate the average molarity and label your EDTA solution |
|
Stoichiometry keypdf
Honors Chemistry: Unit 11 - Molarity and Stoichiometry How many liters of 0 5 M calcium hydroxide do you need in order to have 5 5 moles of Find the number of moles of sulfuric acid needed to react with 5 5 moles of What mass of catcium carbonate is needed to make 1 2 liters of a 1 7 M calcium carbonate solution? |
|
1 CONCENTRATION UNITS A solution is a homogeneous mixture
(b) Calculate the molarity of a solution of 4 8 mole of HCl in 600 mL of solution (k) Calculate the volume (in mL) of a 0 500 M Na2CO3 solution that contains |