carbonic acid
What type of acid is carbonic acid?
CO 2 + H 2 O ⇌ H 2 CO 3 The predominant species are simply loosely hydrated CO 2 molecules. Carbonic acid can be considered to be a diprotic acid from which two series of salts can be formed—namely, hydrogen carbonates, containing HCO 3−, and carbonates, containing CO 32− .
Is carbonic acid a diprotic acid?
Carbonic acid is a diprotic acid, and can hence form two types of salts, namely bicarbonates and carbonates. Addition of a small quantity of a base to H 2 CO 3 yields bicarbonate salts whereas addition of a base in excess yields carbonate salts.
Is carbonic acid a natural leaching agent?
Carbonic acid, which is formed by the dissolution and hydrolysis of CO 2 in water, is the major natural leaching agent in many temperate ecosystems. Carbonic acid is both weak and unstable and quickly dissociates into hydrogen ions (H +) and bicarbonate ions (HCO 3–)
What is the acid-base behaviour of carbonic acid?
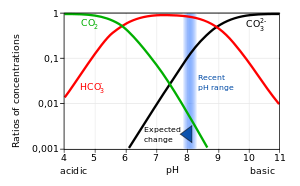
HCO 3− + H 2 O ⇌ H 3 O + + CO 32− However, the acid-base behaviour of carbonic acid depends on the different rates of some of the reactions involved, as well as their dependence on the pH of the system. For example, at a pH of less than 8, the principal reactions and their relative speed are as follows: CO 2 + H 2 O ⇌ H 2 CO 3 (slow)
|
On the Influence of Carbonic Acid in the Air upon the Temperature of
Throughout this paper Arrhenius refers to carbon dioxide as “carbonic acid” in accordance with the convention at the time he was writing. Contrary to some |
|
ON THE INFLUENCE OF CARBONIC ACID IN THE AIR UPON THE
air but in a high degree by aqueous vapor and carbonic acid |
|
9 chemistry of carbonic acid in water
The next sections will be devoted to analysing the chemical composition of carbonate waters. 9.2. CARBONIC ACID EQUILIBRIA. In the presence of gaseous CO2 |
|
CARBONIC ACID AND BICARBONATE IN URINE
Although carbonic acid may be regarded as only inci- dentally a urinary acid the bulk of its production leaving the body by way of the lungs |
|
The Crystal Structure of Carbonic Acid 2279
3 sept. 2022 Carbonic acid H2CO3 |
|
The Crystal Structure of Carbonic Acid 2279
3 sept. 2022 Carbonic acid H2CO3 |
|
Carbonic Acid and Its Mono- and Diprotonation: NMR ab - Initio
Abstract: The structures and l3C NMR chemical shifts of carbonic acid and its mono- and diprotonated forms were calculated using ab initio and IGLO methods |
|
CARBON DIOXIDE SEQUESTRATION BY DIRECT MINERAL
The CO2 is dissolved in water to form carbonic acid (H2CO3) which dissociates to H+ and. HCO3. -. The H+ reacts with the mineral |
|
Dissociation constants for carbonic acid determined from field
A number of workers have recently shown that the thermodynamic constants for the dissociation of carbonic acid in seawater of Mehrbach et al. are more |
|
THE EQUILIBRIUM BETWEEN OXYGEN AND CARBONIC ACID IN
the acid radical of hemoglobin which increases in strength when oxyhemoglobin is formed we may write mass law equations analogous to Equation 5 for carbonic |
|
Chemistry of carbonic acid in water - Department of Nuclear
dissolved carbonic acid, H2CO3 with a = [H2CO3] + [CO2aq] dissolved bicarbonate, HCO3 - with b = [HCO3 -] dissolved carbonate, CO3 2- with c = [ CO3 2-] |
|
Dissociation constants for carbonic acid determined from - NOAA
A number of workers have recently shown that the thermodynamic constants for the dissociation of carbonic acid in seawater of Mehrbach et al are more reliable |
|
Carbonic Acid Derivatives and Heterocumulenes and Their
An overreaction may occur, though, if the heteroatom nucleophile is acylated by the initially formed carbonic acid derivative The acylation of heteroatom |
|
Carbonic Acid Production and the Role of Carbonic - CORE
The result suggests that oxidative decarboxylation yields carbonic acid (HCO3- and H+), which dissociates to form molecular carbon dioxide Brain carbonic |
|
Carbonic Acid - Safety Data Sheet
19 mar 2015 · Carbonic Acid Soln, Created by Global Safety Management, Inc -Tel: 1-813-435 -5161 - www gsmsds com SECTION 1 : Identification of the |











![Solved: 2] PKa Of Carbonic Acid H2CO3 Is 636(3 Marks Solved: 2] PKa Of Carbonic Acid H2CO3 Is 636(3 Marks](https://www.bmj.com/content/s4-1/76/474.extract.jpg)













![120] Similar To Carbonic Acid (H2COs) Hydrogenpdf - 012345678 9 120] Similar To Carbonic Acid (H2COs) Hydrogenpdf - 012345678 9](https://www.bmj.com/content/1/903/562.extract.jpg)