carboxylic acid + hcl
How are carboxylic acids converted into acid chlorides?
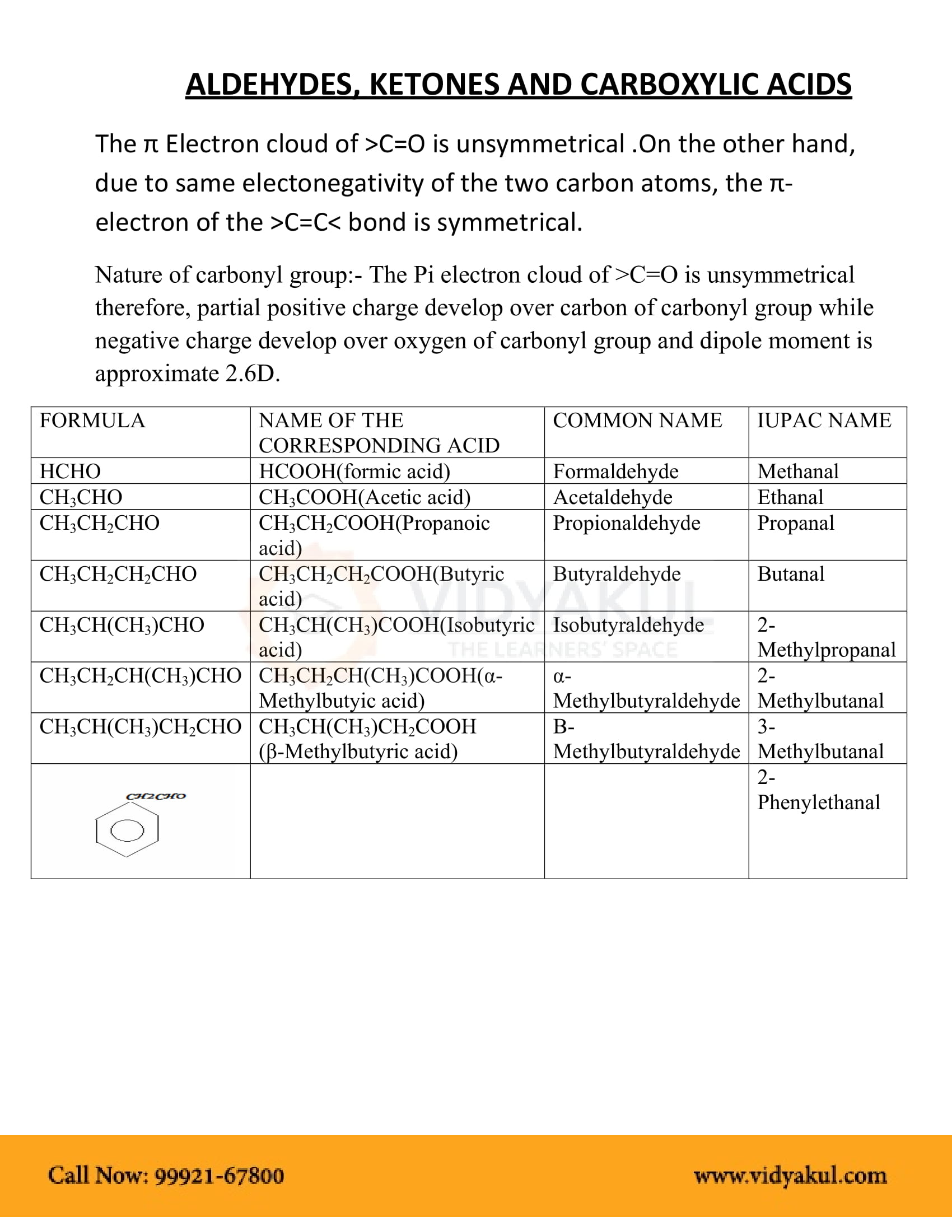
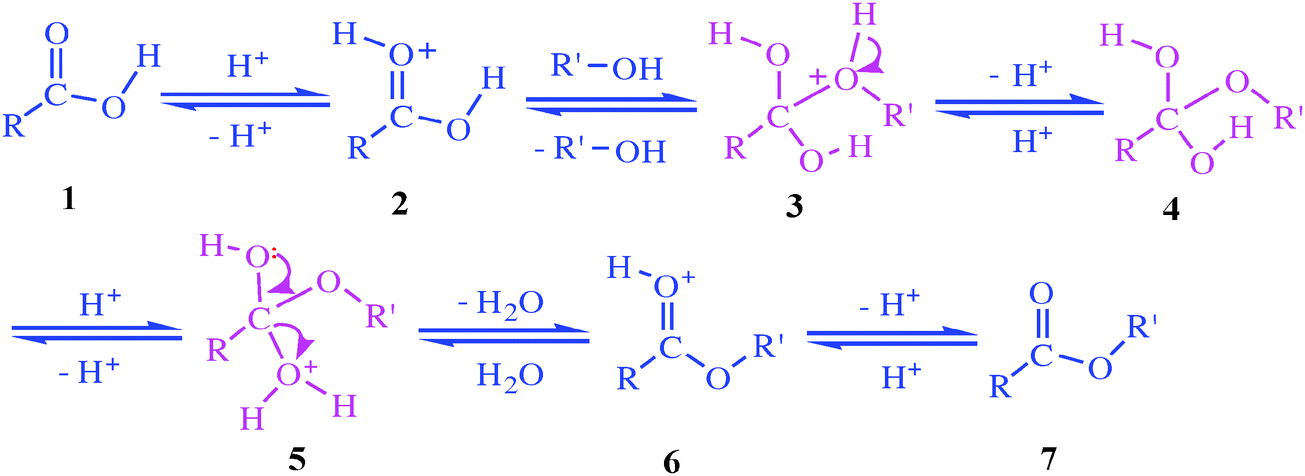
In the laboratory, carboxylic acids are converted into acid chlorides by treatment with thionyl chloride, SOCl 2. This reaction occurs by a nucleophilic acyl substitution pathway in which the carboxylic acid is first converted into an acyl chlorosulfite intermediate, thereby replacing the –OH of the acid with a much better leaving group.
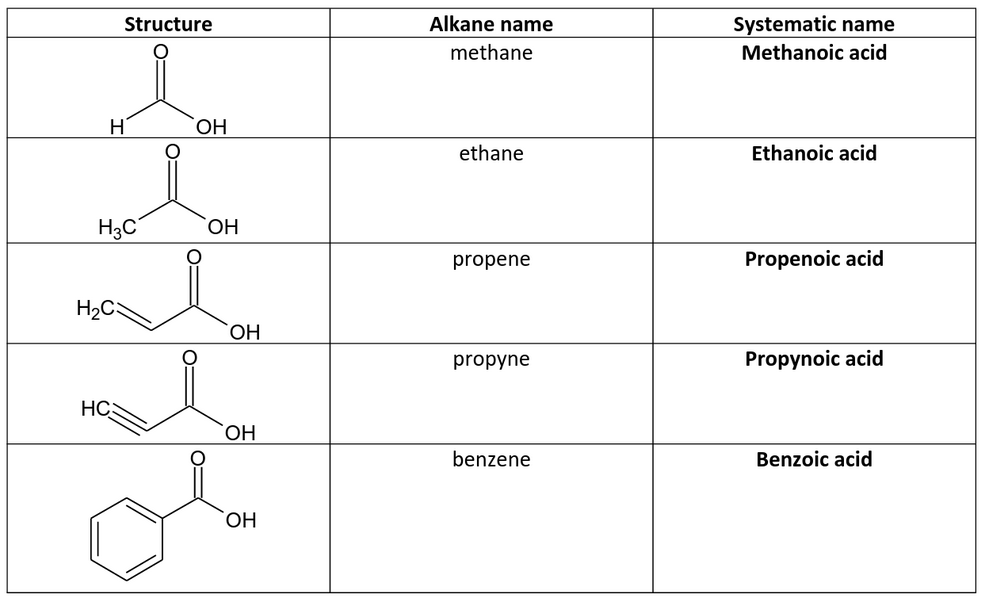
What is a carboxylic acid?
Carboxylic acid, any of a class of organic compounds in which a carbon atom is bonded to an oxygen atom by a double bond and to a hydroxyl group by a single bond. They are generally more acidic than other organic compounds containing hydroxyl groups but are generally weaker than mineral acids such as hydrochloric acid.
Are carboxylic acids polar?
Carboxylic acids are polar. Because they are both hydrogen-bond acceptors (the carbonyl −C (=O)−) and hydrogen-bond donors (the hydroxyl −OH ), they also participate in hydrogen bonding. Together, the hydroxyl and carbonyl group form the functional group carboxyl.
How do carboxylic acids react with thionyl chloride?
This action is not available. Carboxylic acids react with Thionyl Chloride ( SOCl2 S O C l 2) to form acid chlorides. During the reaction the hydroxyl group of the carboxylic acid is converted to a chlorosulfite intermediate making it a better leaving group. The chloride anion produced during the reaction acts a nucleophile.
|
Dissolving Carboxylic Acids and Primary Amines on the Overhead
9 mars 2010 Carboxylic acids (or primary amines) that are only slightly soluble in water are dissolved by the addition of aqueous NaOH. (or HCl). |
|
Chapter 5 Carboxylic Acids and Esters
Learn to recognize the carboxylic acid ester |
|
PRODUCT SPECIFICATION 2-Methylpiperidine-4-carboxylic acid HCl
2-Methylpiperidine-4-carboxylic acid HCl. CAS RN. 1427380-46-8. M. F.. C. 7. H. 14. ClNO. 2. M. W.. 179.64 g/mol. Test. Specification. Color reported. Form. |
|
CH3CH2COOH(aq) + H2O(l) CH3CH2COO-(aq) + H3O+(aq
Propanoic acid CH3CH2COOH |
|
N Goalby chemrevise.org 1 Carboxylic Acids
With HCl an amide will be hydrolysed and split up in to a carboxylic acid and an ammonium salt. Secondary amides can be hydrolysed by aqueous acids. |
|
6.1.3 revision guide carboxylic acids and esters
and esters. CH3COOH + SOCl2. ? CH3COCl + SO2 + HCl. Formation of acyl chloride from a carboxylic acid. Reaction: carboxylic acid ? acyl chloride. |
|
Prime Scholars
oxoimidazolidine-4-carboxylic acid moiety and de alkylation has done with 15% HCl in. 14 –dioxane. Shiba etal. 3 reported the synthesis of. |
|
Carboxylic acid derivatives
Preparation of acyl chlorides. Acid chlorides can not be prepared by reaction between carboxylic acid and HCl. This is because the carbonyl. |
|
Chem 360 Jasperse Ch. 20 21 Notes + Answers. Carboxylic Acids
Mechanism: Not Required. • Easy (but smelly) reaction. Side products HCl and SO2 are gases so can just evaporate away leaving clean |
|
Carboxylic Acids
the acid can be liberated from its salt by treatment with a stronger acid. e.g. RCOO¯ Na+. (aq) + HCl(aq) —> RCOOH + NaCl(aq). |
|
Synthesis of Carboxylic Acids
Mechanism: Not Required • Easy (but smelly) reaction Side products HCl and SO2 are gases, so can just evaporate away leaving clean, useful product So no |
|
Acid-Base Extraction
Neutralization of the carboxylic acid salt and excess sodium bicarbonate with concentrated hydrochloric acid It is critical to add an amount of HCl necessary to |
|
Chem 263 Nov 24, 2009 Properties of Carboxylic Acids Since
24 nov 2009 · Other carboxylic acid derivatives, such as acid chlorides, anhydrides, dissociation found with strong mineral acids such as HCl and H2SO4 |
|
CARBOXYLIC ACIDS AND THEIR DERIVATIVES 1 Carboxylic
sulfuric acid formic acid reacts with phenols with the formation of corresponding triphenylmethane dyes (see p 196) Reagents: powdered magnesium, dilute HCl , |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
HCl 50 Na OH conc H Cl , Δ + NaCN workup with H3O via Fig 7 8 Partial hydrolysis of a nitrile to a primary carboxylic acid amide under acidic condi- |
|
85 Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl
+ SO2 + HCl Nucleophilic acyl substitution reactions of acid halides 1 Anhydride formation; Acid chlorides react with carboxylic acids to give acid anhydrides |
|
Carboxylic acid deri
Fisher esterification reaction: acid-catalyzed reaction of carboxylic acids with 1° or 2° alcohols to give esters Reagents: ROH (usually solvent), HCl (strong acid) |










![Carboxylic Acids and Derivatives - [PDF Document] Carboxylic Acids and Derivatives - [PDF Document]](https://data01.123dok.com/thumb/y8/gn/jd2z/Pki4ZiPT5VIqTD1so/cover.webp)