carboxylic acid mcqs pdf
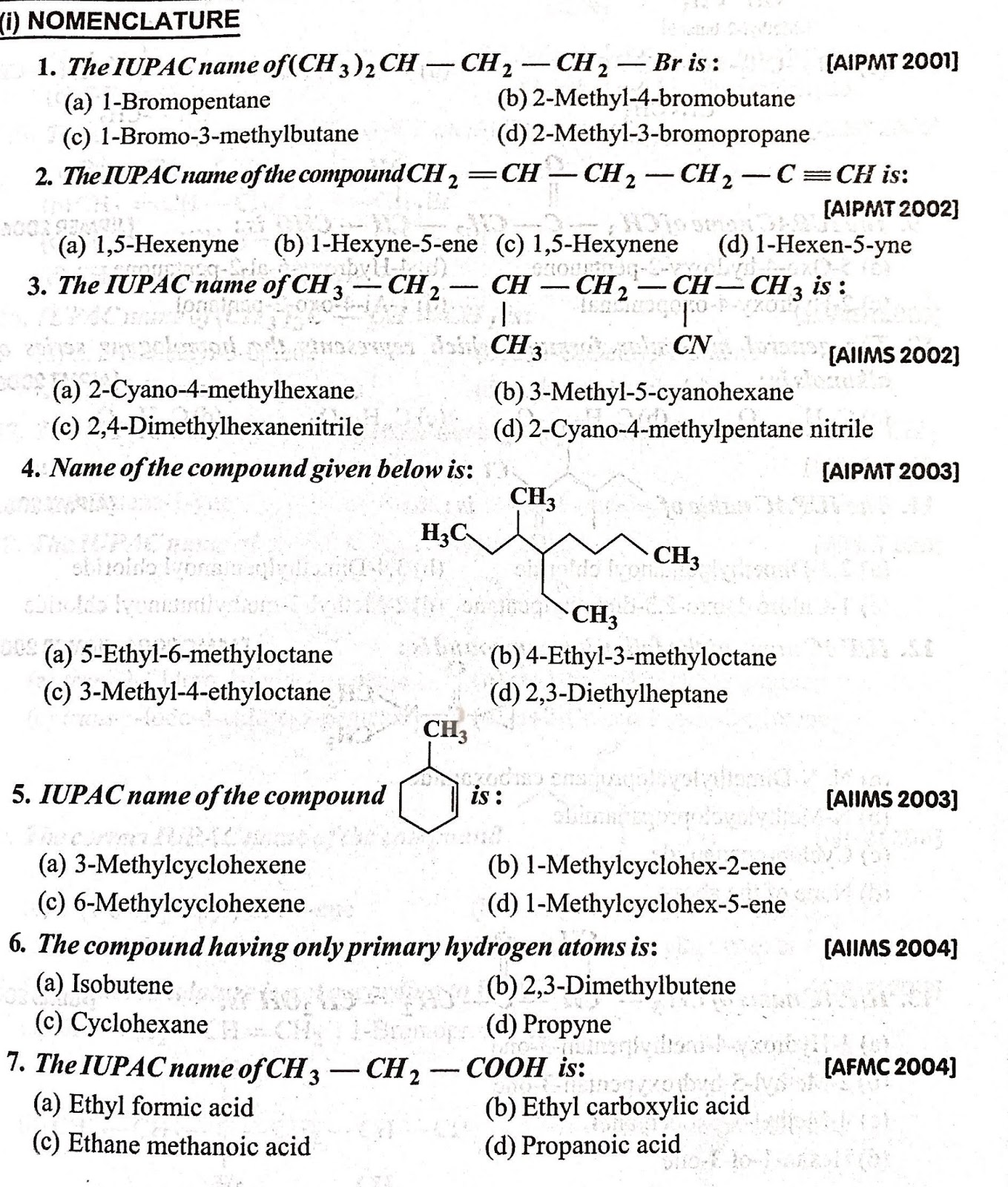
|
Carboxylic Acids and Esters (MCQ)
Carboxylic Acids and Esters (MCQ) 1 Which of the following reactions produce propan-1-ol? 1 The alkaline hydrolysis of 1-chloropropane 2 The acid hydrolysis of propyl methanoate 3 The acid hydrolysis of propanenitrile A 1 2 and 3 B Only 1 and 2 C Only 2 and 3 |
|
CHEMISTRY MULTIPLE CHOICE QUESTIONS
May 18 2021 · carboxylic acid ester Mevalonic acid 35-dihydroxy-3-methylpentanoic acid is involved in cholesterol formation in the body It is an oil that occurs as a mixture of the two interchanging molecules shown in the diagram HO CH20H C02H HO c What names are used to describe the pair of interchanging reactions I and Il? A B c D condensation and |
How to write a carboxylic acid study guide?
CARBOXYLIC ACIDS STUDY GUIDE 1. Name the following carboxylic acids and carboxylate ions: 56 2. Write the general chemical equation (using an R group) showing the formation of a hydrogen ion from a carboxylic acid. Is this reaction reversible or irreversible? Are carboxylic acids weak or strong acids in general?
Which method is used for determining molecular mass of carboxylic acids?
Silver salt method is used for determining molecular mass of carboxylic acids. Carboxylic acids from insoluble salts which upon heating decompose to leave a residue of metallic silver, then this residue is filtered, dried and estimated. Was this answer helpful? A 0.607 g of a silver salt of a tribasic acid on combustion gave 0.37 g of Ag.
What is the formula for carboxylic acid?
The general formula of all Carboxylic Acids is C n H 2n+1 COOH. Where n = number of carbon atoms present in the molecule -1. The first five Carboxylic Acids are Methanoic Acid (HCOOH), Ethanoic Acid (CH 3 COOH), Propanoic Acid (CH 3 CH 2 COOH), Butanoic Acid (CH 3 CH 2 CH 2 COOH), and Pentanoic Acid (CH 3 CH 2 CH 2 CH 2 COOH). Q6.
Why are carboxylic acids acidic?
Answer: Carboxylic Acids are acidic because the hydrogen atom in the carboxylic acid group is directly attached to the highly electronegative oxygen atom. Due to this reason, the hydrogen atom can easily be lost in the solution leading to the formation of a hydrogen ion (H +) and a carboxylate anion (RCOO – ).
|
Acces PDF Physical Chemical Self Test Solution Copy - covid19.gov
Structure and Theory MCQs Chapter 3: Benzene: Chemical Compound MCQs Chapter 4: Carbonyl Compounds MCQs Chapter 5: Carboxylic Acids and Acyl Compounds MCQs |
|
File Type PDF Section 23 Chemical Properties Answers Copy
Sep 3 2022 Practice Carboxylic Acids and Acyl. Compounds MCQ book PDF with answers |
|
Download Free Answers To Chemistry (PDF) - covid19.gov.gd
Practice Carboxylic Acids and Acyl Compounds MCQ book PDF with answers test 5 to solve MCQ questions bank: Acidity of carboxylic acids |
|
Online Library Physical Chemical Self Test Solution - covid19
7th Grade Science MCQ PDF book helps to practice test questions from exam prep Practice Carboxylic Acids and Acyl Compounds MCQ book PDF with answers ... |
|
Physical Chemical Self Test Solution
(MCQs) PDF: Quiz & Practice Tests with Answer Key (Grade 7 Science Question Bank Practice Carboxylic Acids and Acyl Compounds MCQ book PDF with answers ... |
|
Online Library Chemistry Chapter 6 Study Guide (PDF) - covid19
3 days ago Compounds MCQ book PDF with answers test 5 to solve MCQ questions bank: Acidity of carboxylic acids |
|
Download Free Section 23 Chemical Properties Answers (PDF
Sep 3 2022 Carboxylic Acids and Acyl Compounds Worksheet Chapter 6: Chemical Bonding ... Level Chemistry MCQ PDF book helps to practice test questions ... |
|
Download File PDF Physical Chemical Self Test Solution [PDF
Choice Questions and Answers (MCQs) PDF: Quiz & Practice Tests with Answer Key (Grade 7 Practice Carboxylic Acids and Acyl Compounds MCQ book PDF with. |
|
Section 23 Chemical Properties Answers
Solve Carboxylic Acids and Acyl Compounds study guide PDF with answer key Level Chemistry MCQ PDF book helps to practice test questions from exam prep ... |
|
Download File PDF Chapter 16 Acid Base Equilibria Solubility
Acidity of carboxylic acids acyl chlorides |
Key Information about Carboxylic Acids
Carboxylic acids are organic compounds characterized by the presence of a carboxyl group (-COOH). They are commonly found in various natural and synthetic substances and play essential roles in biological processes and industrial applications.
Examples of Carboxylic Acids
1. Acetic acid (CH3COOH) - Found in vinegar.
2. Citric acid (C6H8O7) - Found in citrus fruits like lemons and oranges.
3. Formic acid (HCOOH) - Found in ant stings and certain plants.
4. Butyric acid (CH3CH2CH2COOH) - Found in butter and dairy products.
5. Salicylic acid (C7H6O3) - Used in skincare products as an exfoliant.
Practice Exercises for Carboxylic Acids
- Write the structural formula for acetic acid.
- Identify the functional group present in citric acid.
- Name the carboxylic acid with the formula CH3CH2COOH.
- Draw the condensed structural formula for formic acid.
- List two uses of salicylic acid in everyday products.
Solutions:
- CH3COOH
- Carboxyl group (-COOH)
- Butyric acid
- HCOOH
- Skincare products (e.g., acne treatment, exfoliating creams)
Case Studies on Carboxylic Acids
1. The role of acetic acid in vinegar production and food preservation.
2. Citric acid's function as a flavor enhancer and preservative in food and beverage industry.
3. Formic acid's use in leather tanning and textile industry.
4. Butyric acid's importance in the fermentation process of dairy products.
5. Salicylic acid's application in pharmaceuticals for treating acne and skin conditions.
Subcategories in Carboxylic Acids
1. Aliphatic carboxylic acids: Straight-chain or branched carboxylic acids.
2. Aromatic carboxylic acids: Carboxylic acids containing an aromatic ring.
3. Hydroxy carboxylic acids: Carboxylic acids containing hydroxyl (-OH) group.
4. Dicarboxylic acids: Carboxylic acids with two carboxyl groups.
5. Unsaturated carboxylic acids: Carboxylic acids containing carbon-carbon double bonds.
These subcategories help classify and understand the structural diversity of carboxylic acids.
Important Notes on Carboxylic Acids
- Carboxylic acids are characterized by the carboxyl functional group (-COOH).
- They are weak acids that can donate a proton (H+) to form carboxylate ions.
- Many carboxylic acids have distinctive odors and flavors.
- They are widely used in food industry, pharmaceuticals, and chemical synthesis.
- Carboxylic acids participate in various chemical reactions including esterification, hydrolysis, and decarboxylation.
Step-by-Step Guide to Understanding Carboxylic Acids
- Learn the structure and functional group of carboxylic acids.
- Study the nomenclature and naming conventions for different carboxylic acids.
- Explore the physical and chemical properties of carboxylic acids.
- Understand the significance and applications of carboxylic acids in various industries.
- Practice reactions and synthesis involving carboxylic acids to reinforce learning.
Common Questions and Answers about Carboxylic Acids
Q: What distinguishes carboxylic acids from other organic compounds?
A: Carboxylic acids contain a carboxyl functional group (-COOH), which is unique to this class of compounds.
Q: How do carboxylic acids contribute to food flavor and preservation?
A: Carboxylic acids act as flavor enhancers and natural preservatives in various food products.
Q: What are some common reactions of carboxylic acids?
A: Common reactions include esterification, hydrolysis, decarboxylation, and reduction.
Q: How are carboxylic acids named according to IUPAC nomenclature?
A: Carboxylic acids are named by replacing the -e ending of the corresponding alkane with -oic acid.
Q: What are the industrial applications of carboxylic acids?
A: Carboxylic acids are used in the production of plastics, pharmaceuticals, flavors, fragrances, and agricultural chemicals.
Multiple Choice Questions about Carboxylic Acids
- What is the functional group present in carboxylic acids?
- Hydroxyl group (-OH)
- Carbonyl group (C=O)
- Carboxyl group (-COOH)
- Amino group (-NH2)
- Which of the following is an example of a carboxylic acid?
- Propanol
- Butanal
- Acetic acid
- Hexane
- What is the chemical formula of acetic acid?
- C2H4O2
- CH3COOH
- C6H12O6
- H2SO4
- Which type of carboxylic acid contains a carbon-carbon double bond?
- Aliphatic carboxylic acid
- Aromatic carboxylic acid
- Unsaturated carboxylic acid
- Hydroxy carboxylic acid
- What is the common name for ethanoic acid?
- Acetic acid
- Formic acid
- Propionic acid
- Butyric acid
About Carboxylic Acids
Carboxylic acids are organic compounds containing a carboxyl functional group (-COOH). They are versatile compounds with diverse applications in various industries including food, pharmaceuticals, and chemicals.
Important Elements to Remember in Carboxylic Acids
1. Carboxylic acids contain the carboxyl functional group (-COOH).
2. They exhibit acidic properties due to the presence of the carboxyl group.
3. Carboxylic acids are versatile compounds with applications in various industries.
4. Understanding the nomenclature, properties, and reactions of carboxylic acids is essential in organic chemistry.
5. Practice and experimentation are key to mastering the concepts related to carboxylic acids.
|
ORGANIC CHEMISTRY
The formic acid has the following characteristic chemical property, unlike the other saturated carboxylic acids: A is the weakest acid B has reducing character |
|
Mht-cet paper pattern - Target Publications
MHT-CET 2019 Question Paper in PDF format MCQs in each chapter are divided into three sections: 12 11 Methods of preparation of carboxylic acids |
|
24 February 2003 This exam has seven (7) questions, two cover
24 fév 2003 · B In which selection are the carboxylic acid derivatives arranged from a) acid anhydride, acid chloride, carboxylic amide, carboxylic ester |
|
Mcq On General Organic Chemistry - OpenBeauchef
pdf includes multiple choice questions and answers (MCQs) for 10th-grade competitive exams "Carboxylic Acids and Acyl Compounds MCQs" pdf covers quiz |
|
MCQs
Of the following four reactions, formic acid and acetic acid differ in which respect? (a) Replacement of hydrogen by sodium (b) Formation of ester with alcohol (c) |
|
Department of Chemistry BSc-II Chemistry Question Bank Organic
Salicyladehyde condense with acetic anhydride in presence of base catalyst then the product is a Cinnamylacrylic acid b Chrysin c Furylacrylic acid d Coumarin |
|
Organic Chemistry
Furan-2-carboxylic acid C 2-acetylfuran D 2-formylfuran 6 The synthesis of 1,2 -dimethylimidazole using Debus-Radziszewski involves following reactants A |
|
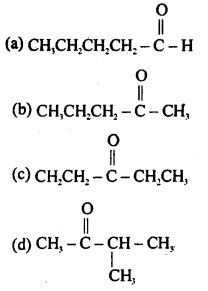
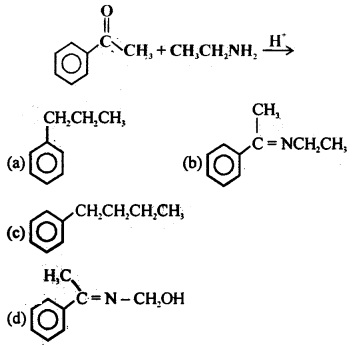
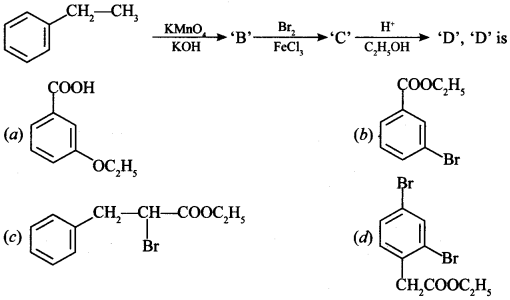
Aldehydes, Ketones and Carboxylic Acids - NCERT
The carbonyl compounds in which carbonyl group is bonded to oxygen are known as carboxylic acids, and their derivatives (e g esters, anhydrides) while in |










![Carboxylic Acids and Derivatives - [PDF Document] Carboxylic Acids and Derivatives - [PDF Document]](https://studygramhome.files.wordpress.com/2019/12/chemistry-mcqs-for-class-12-with-answers-chapter-12-aldehydes-ketones-and-carboxylic-acids-13.png?resize\u003d580%2C172)