carboxylic acid notes pdf
|
Carboxylic Acids
R C O-H RCO2H RCOOH Carboxylic acids are classified according to the substituent that is bonded to the carboxyl carbon: Aliphatic acids have an alkyl group bound to the carboxyl group aromatic acid has an aryl group bound to the carboxyl group The simplest acid is formic acid |
|
Notes on Chapter 21
Notes on Chapter 21 - Carboxylic Acids I Introduction to carboxylic acids II Nomenclature of carboxylic acids III Spectroscopy of carboxylic acids IV Synthesis of carboxylic acids V Reactions of carboxylic acids Reduction of carboxylic acids Reactions with organolithium reagents Conversion to acid chlorides Conversion to esters Conversion |
|
Chapter 16: Carboxylic Acids Esters and Other Acid Derivatives
A carboxylic acid is an organic compound whose functional group is the carboxyl group What is a carboxyl group? A carboxyl group is a carbonyl group (C=O) with a hydroxyl group (—OH) bonded to the carboxyl carbon atom A general structural representation fit a carboxyl group is Abbreviated linear designations for the carboxyl group are |
|
39 Carboxylic Acids and Derivatives
Feb 3 2021 · Esterification Carboxylic acids react with alcohols in the presence of a strong acid catalyst to form esters and water H+ Carboxylic Acid + Alcohol Ester + water H O C C C O H The bit ending in –anoate comes from the carboxylic acid and includes the C in the C=O bond |
|
Ch 05 Carboxylic Acids and Esters
Carboxylic acids are weak organic acids which contain the carboxyl group (RCO2H): C OH carboxylic acid O C O the carboxyl H RCOOH RCO2H condensed ways of writing the carboxyl group H group • The tart flavor of sour-tasting foods is often caused by the presence of carboxylic acids Nomenclature of Carboxylic Acids 3 Nomenclature of Carboxylic Acids |
|
Carboxylic acids (RCOOH)
Structure of carboxylic acids and their derivatives Carboxylic acids have the following general formula: Some simple carboxylic acids: formic acid IUPAC: methanoic acid acetic acid IUPAC: ethanoic acid IUPAC: benzoic acid Since carbon can have only four bonds there are no cyclic carboxylic acids (i e the carboxyl group cannot form part of |
What is a carboxylic acid whose functional group is the carboxyl group?
A carboxylic acid is an organic compound whose functional group is the carboxyl group. What is a carboxyl group? A carboxyl group is a carbonyl group (C=O) with a hydroxyl group (—OH) bonded to the carboxyl carbon atom. A general structural representation fit a carboxyl group is
Why do carboxylic acids have Greek letters?
When Greek letters are used, Greek letters are used to designate the position of substituent relative to the carbon of the carboxyl group. The carbon of the carboxyl group is NOT given a Greek letter. 6) A special group of carboxylic acids are those that also have a keto group. They are called alpha-keto carboxylic acids
Why do carboxylic acids have higher boiling points than alcohols?
A given carboxylic acid molecule form two hydrogen bonds to another carboxylic acid molecule, producing a “dimer”, a complex with a mass twice that of a single molecule. Compounds with carboxyl groups have higher boiling points than alcohols. This is because the carboxyl groups hydrogen bond more strongly than alcohols.
Do carboxyl groups have a strong hydrogen bonding?
Carboxyl groups exhibit very strong hydrogen bonding. A given carboxylic acid molecule form two hydrogen bonds to another carboxylic acid molecule, producing a “dimer”, a complex with a mass twice that of a single molecule. Compounds with carboxyl groups have higher boiling points than alcohols.
|
CARBOXYLIC ACIDS
Carboxylic acids are organic compounds containing the carboxyl The common name of a carboxylic acid (R-COOH) is derived by adding the suffix –ic acid to ... |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. |
|
Carboxylic-acids.pdf
STRUCTURE OF CARBOXYLIC ACID. • Carbon atom of carboxyl group is sp2 – hybridized and forms one ?- bond with hydrogen or. |
|
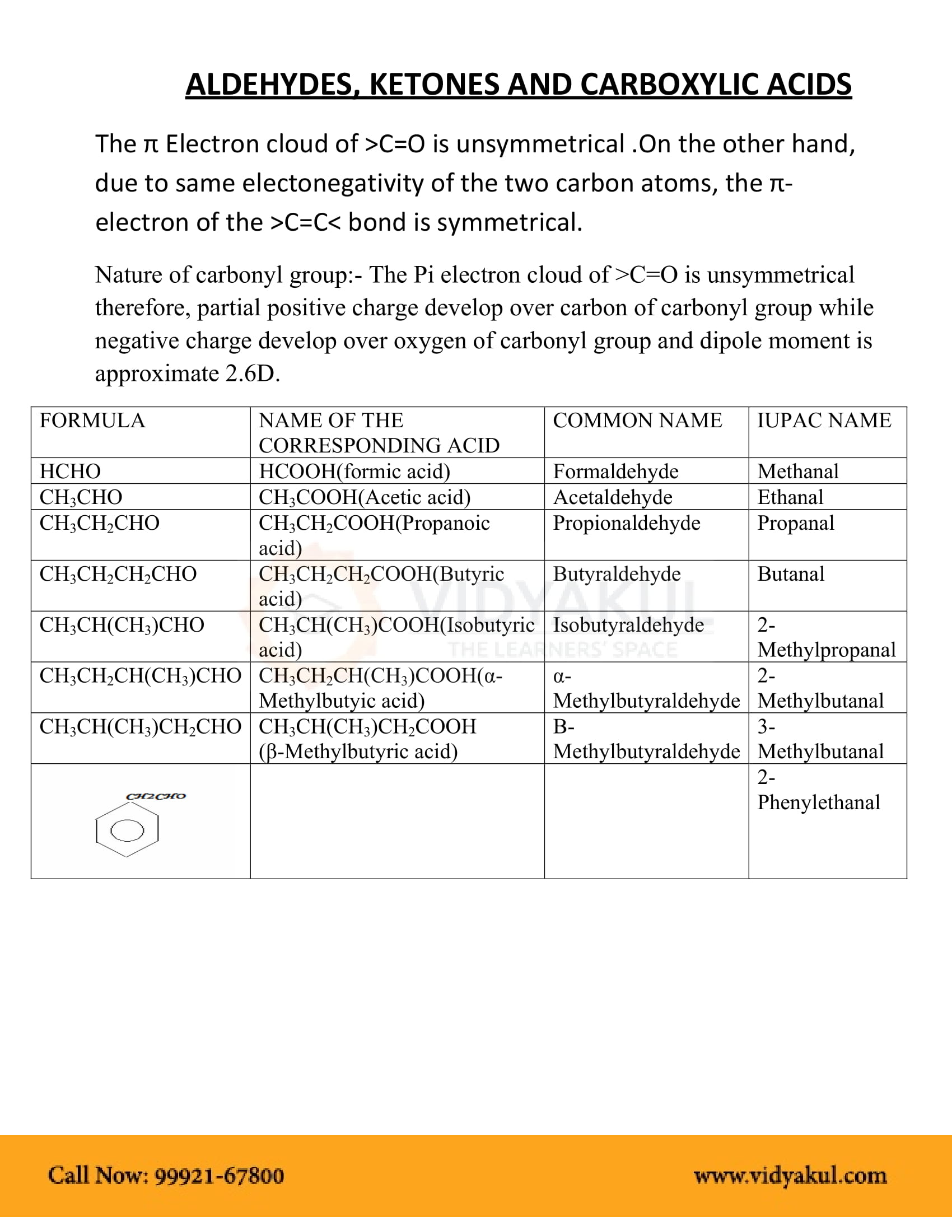
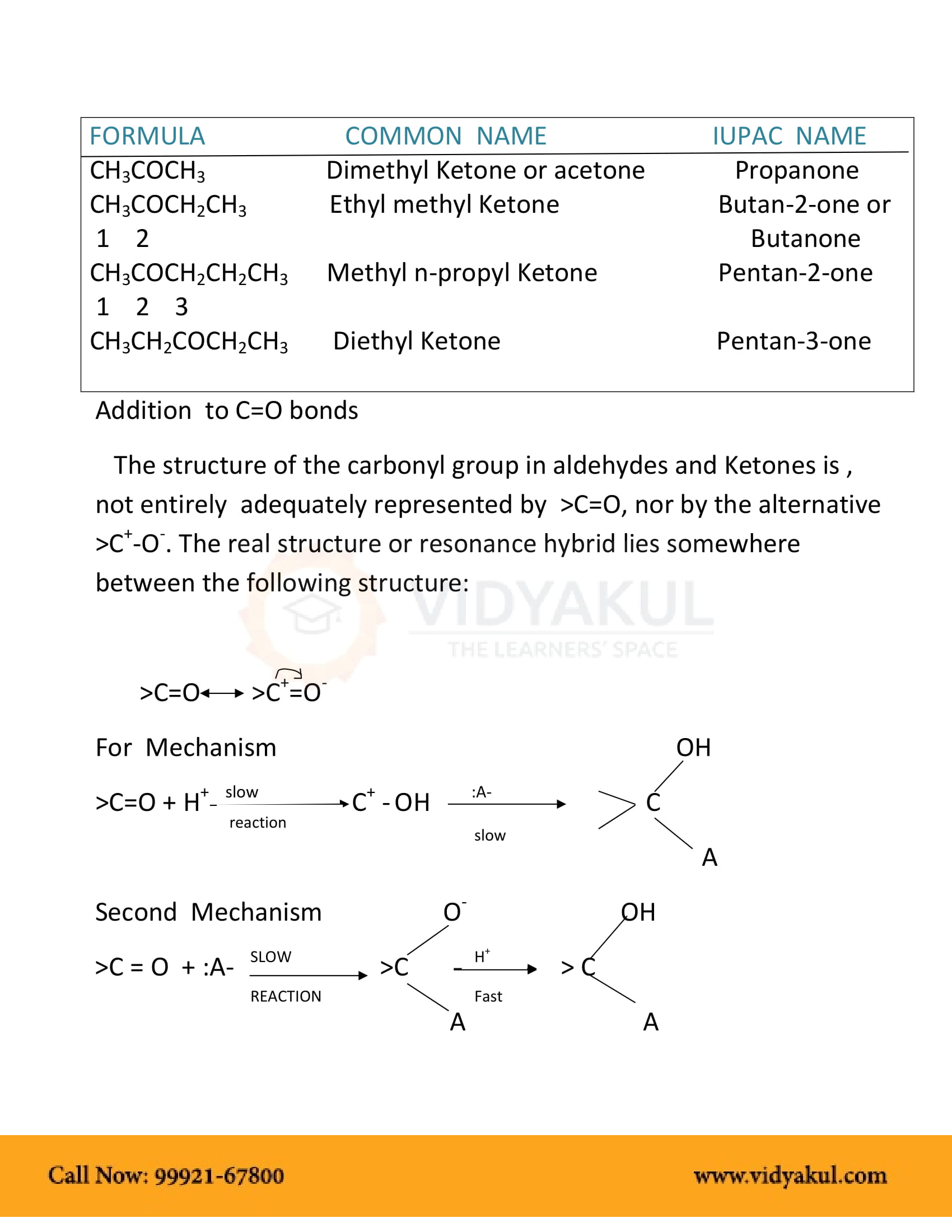
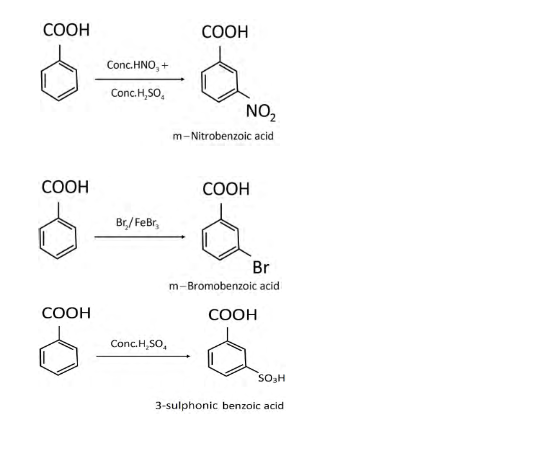
ALDEHYDES KETONES AND CARBOXYLICACIDS
Aldehydes Ketones and Carboxylic Acids. Notes. Chemistry of Organic. Compounds. 29.1 Aldehydes and Ketones. You have some familiarity with these classes of |
|
Carboxylic acid
Carboxylic Acids. Compounds containing. Carboxylic are Called. QN p-. 0 ?????? ??????? ??. H-C. CH3C .OH formic. In Carbonylic acids the. |
|
Carboxylic Acids
Structure of the Carboxyl Group. The most stable conformation of formic acid is an almost planar arrangement of the molecule. The carbon is sp2 hybridized and |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
The carbonyl compounds in which carbon of carbonyl group is bonded to carbon or hydrogen and oxygen of hydroxyl moiety (-OH) are known as carboxylic acids |
|
INFRARED SPECTROSCOPY (IR)
The remainder of this presentation will be focused on the IR identification of various functional groups such as alkenes alcohols |
|
Carboxylic Acids
13.1 What Are Carboxylic Acids? The functional group of a carboxylic acid is a carboxyl group so named because it is made up of a carbonyl group and |
|
New Strategy for Easy Sensitive and Selective Detection of THC
THC Carboxylic Acid Direct from Hair. Use of LC-MRM. 3 on SCIEX QTRAP. ®. 5500 LC-MS/MS System. Dr. Axel Besa. AB SCIEX Darmstadt |
|
Carboxylic Acids
Chapter 10 Lecture Notes: Carboxylic Acids, Amines, and Amides Educational Goals 1 Given the structure of a carboxylic acid, carboxylate ion, ester, amide, |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
1 Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides Educational Goals 1 Given the structure of a carboxylic acid, carboxylate ion, ester, amide, |
|
Chapter 16: Carboxylic Acids, Esters, and Other Acid Derivatives
Although we see within a carboxyl group both a carbonyl group (C=O) and hydroxyl group (—OH) 16 2 IUPAC Nomenclature for Carboxylic Acids The naming of |
|
CARBOXYLIC ACIDS
Carboxylic acids may to dissociate to a carboxylate anion and a hydrogen ion ( proton) Page 17 Substituent Effects on the Acidity of Aliphatic Carboxylic Acids 19 |
|
Aldehydes, Ketones and Carboxylic Acids - Ncert Help
Chemistry Notes for class 12 Chapter 12 Aldehydes, Ketones and Carboxylic Acids In aldehydes, the carbonyl group ( )C=O) is bonded to carbon and |
|
Chapter 18: Carboxylic Acids 181: Carboxylic Acid Nomenclature
They are significantly more acidic than water or alcohols Bronsted Acidity (Ch 1 13): Carboxylic acids transfer a proton to water to give H3O+ and carboxylate |
|
CARBOXYLIC ACIDS - Spiro Academy
STRUCTURE OF CARBOXYLIC ACID • Carbon atom of carboxyl group is sp2 – hybridized and forms one σ- bond with hydrogen or carbon atom depending |
|
Organic Chemistry II / CHEM 252 Chapter 18 – Carboxylic Acids and
Introduction 2 • The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl compounds or carboxylic acid derivatives |
|
Aldehydes, Ketones and Carboxylic Acids - NIOS
Aldehydes, Ketones and Carboxylic Acids Notes Chemistry of Organic Compounds 29 1 Aldehydes and Ketones You have some familiarity with these |
|
211 Introduction Carboxylic Acids 211 Introduction Carboxylic
25 avr 2012 · The carbon of the carboxylic acid moiety is assigned the locant position 1 Copyright 2012 John Wiley Sons, Inc Klein, Organic Chemistry 1e |