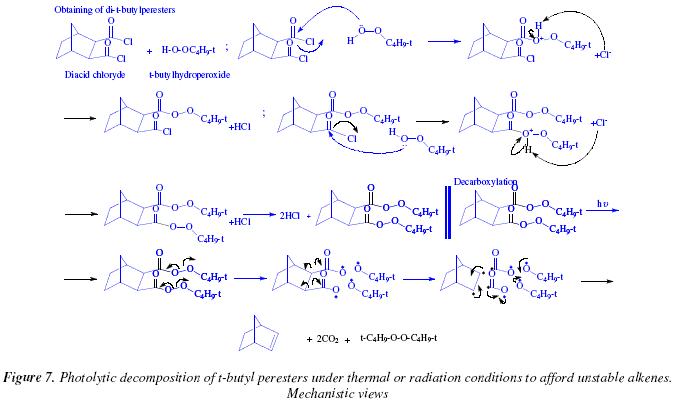
carboxylic acid product of hydrolysis
Does hydrolysis occur under acidic or basic conditions?
Hydrolysis can occur under acidic or basic conditions that determine the ionization of the carboxylic acid. Reactions under basic conditions will require a final neutralization step with dilute H + to recover the carboxylic acid. To reinforce our awareness of the pH sensitivity of carboxylic acids, both reaction maps are shown below.
What is a carboxylic acid hydrolysis reaction?
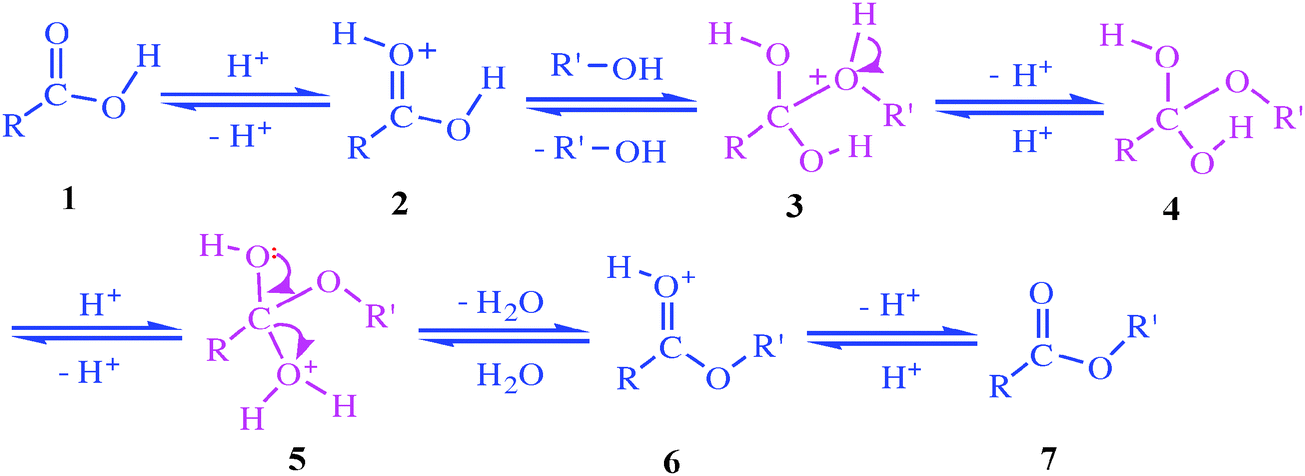
The carboxylic acid derivatives along can be hydrolyzed to produce carboxylic acids. These hydrolysis reactions have limited use in multiple-step synthesis because the acidic proton can be problematic for many organic reactions. Biochemically, hydrolysis reactions are very important in the metabolism of food, drugs, and other nutrients.
How to make carboxylic acid from an alkyl halide?
Since nitriles themselves are usually made by S N 2 reaction of a primary or secondary alkyl halide with CN –, the two-step sequence of cyanide displacement followed by nitrile hydrolysis is a good way to make a carboxylic acid from an alkyl halide ( RBr → RC≡N → RCO 2 H ).
What are the different types of carboxylic acid synthesis?
Carboxylic acid - Synthesis, Reactions, Properties: Most of the methods for the synthesis of carboxylic acids can be put into one of two categories: (1) hydrolysis of acid derivatives and (2) oxidation of various compounds.
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. |
|
The Catalytic Hydrogenation of the Reissert Compound and
acid hydrolysis of the latter gives benzaldehyde product from ethanol and from acetic acid ... quinoline-2-carboxylic acid by refluxing with aque-. |
|
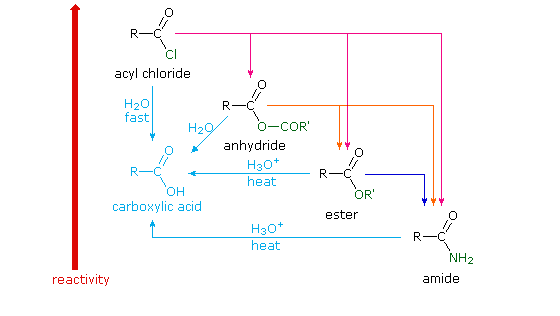
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
(Compare this reaction with addition to the carbonyl group of an aldehyde or ketone.) Although the resulting addition products are stable in some cases in most |
|
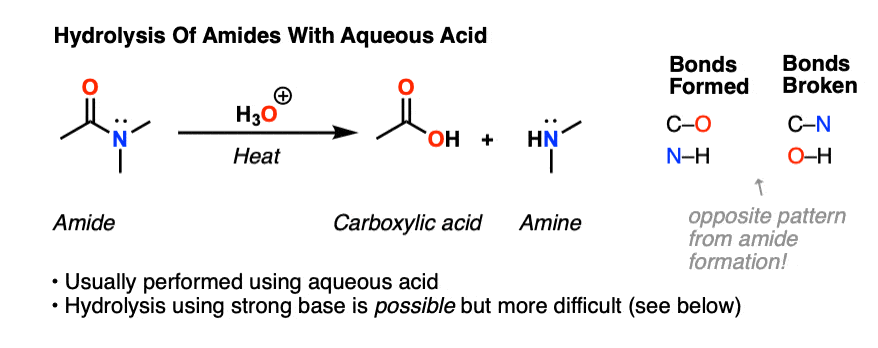
Hydrolysis of Amides to Carboxylic Acids Catalyzed by Nb2O5
Dec 25 2563 BE In the hydrolysis of 1.0 mmol acetamide (1a) |
|
MicrowavE Assisted hydrolysis of MeldrumS Acid Derivatives and
the corresponding malonic acids which on decarboxylation with pyridine yield mono-carboxylic acids. Monoalkyl. Meldrum's acids resist alkaline hydrolysis |
|
A Study of Optimal Conditions for Reducing Sugars Production from
Hydrolysis by 0.25 M acetic acid could produce 30.36% yield of reducing sugars under the same conditions. Enzymatic hydrolysis of 1.5% cassava peels by |
|
Acylium Ion Formation in the Reactions of Carboxylic Acid
ring has been inferred from reaction products with other electrophilic reagents e.g. |
|
HYDROLYSIS 2016.pdf
carbon centre such as with carboxylic acid derivatives including esters |
|
Carboxylic acid participation in amide hydrolysis. Competition
of the hydrolysis products prevents the intermediate formationof an imide. participation by a neighboring carboxylic acid is a reaction that. |
|
Intermediates in the Reactions of Carboxylic Acid Derivatives. IV
Intermediates in theReactions of Carboxylic Acid Derivatives. IV. intermediate from which the products of hydrolysis are formed without any appreciable. |
|
HYDROLYSIS
In the C-O cleavage reaction, the mechanism is also a direct displacement SN2 reaction, where the phosphate ester anion is acting as the leaving group |
|
Synthesis of Carboxylic Acids
R 1 NaOR 2 R-X H3O+, heat • Mechanism: Deprotation/Alkylation covered previously The hydrolysis of the esters to acids will be required (see reaction 8b) |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions; • account for the irreversibility of the hydrolysis reaction under basic |
|
Estimated carboxylic acid ester hydrolysis rate constants for - CORE
28 sept 2011 · carboxylic acid ester groups that may be amenable to hydrolysis in various food and beverage products Acid- (kA) and base- (kB) catalyzed |
|
Carboxylic Acid Derivatives Addition/Elimination
Definition: A carboxylic acid derivative undergoes hydrolysis (bond breaking with The products for anhydride hydrolysis are two carboxylic acids, the extent to |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
The reaction of carboxylic acid derivatives of the type R–C(O)–Het with electrophiles oxy late, which would amount to a “total hydrolysis of the nitrile”: |











![Chapter 5 Carboxylic Acids and Esters - [PDF Document] Chapter 5 Carboxylic Acids and Esters - [PDF Document]](https://ars.els-cdn.com/content/image/3-s2.0-B9780080523491001682-gr71.gif)