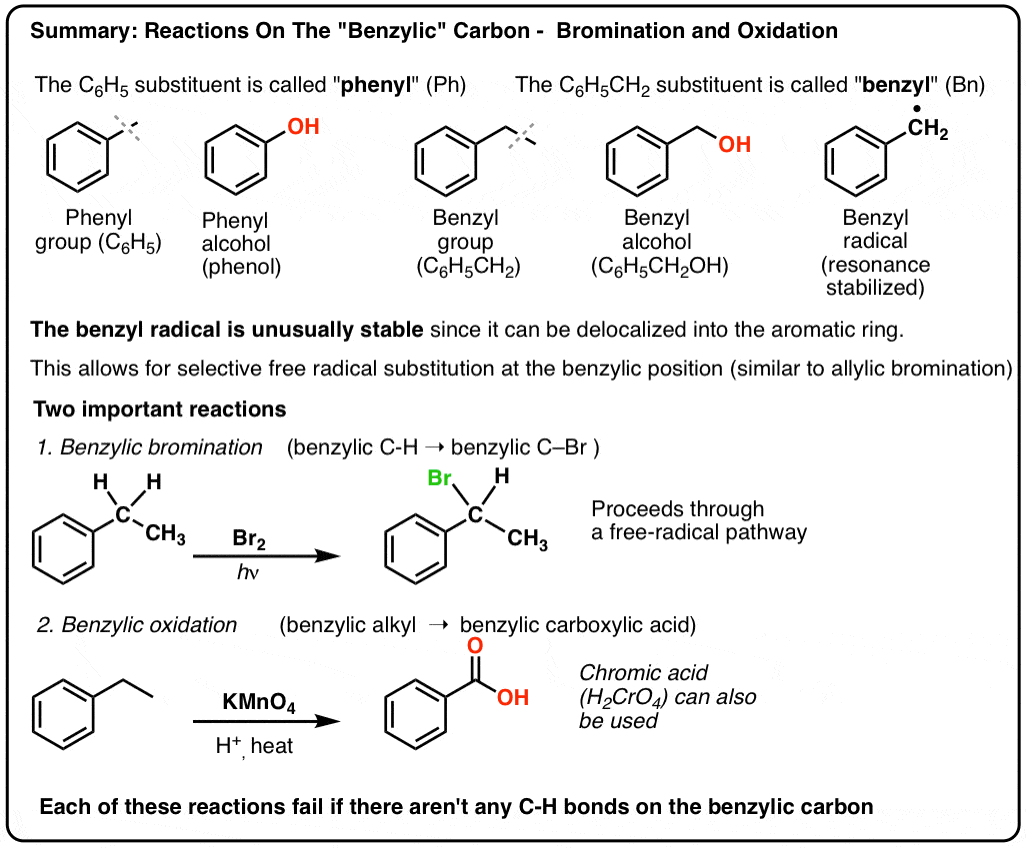
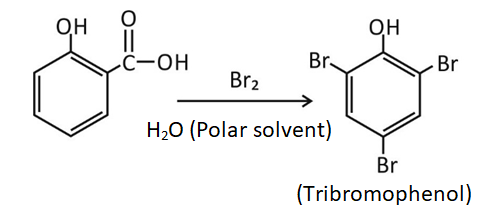
carboxylic acid reaction with bromine water
Why do bromides react with -bromo carboxylic acids?
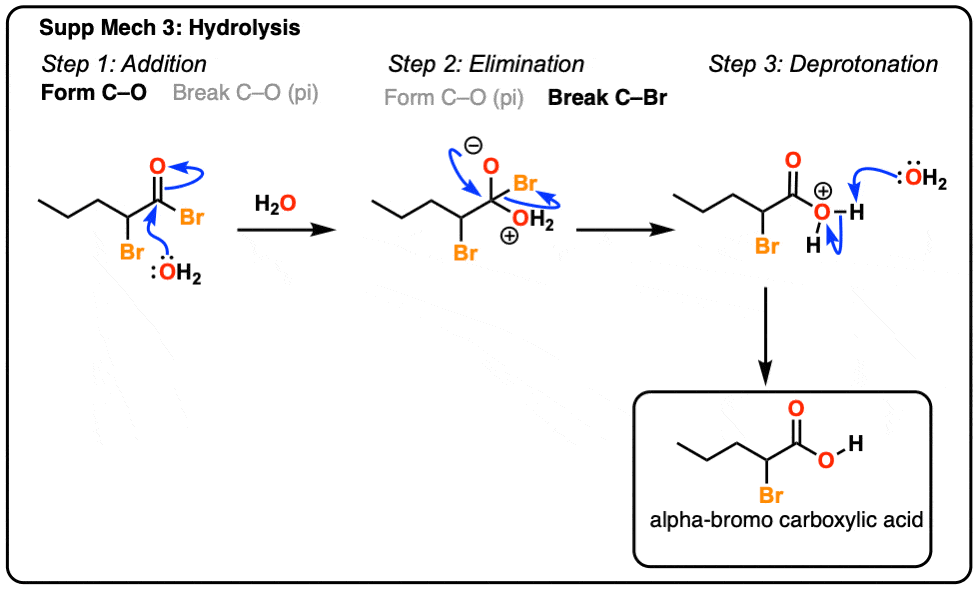
Because bromides are capable of reacting with a wide variety of nucleophiles, α -bromo carboxylic acids serve as important intermediates. Reaction of α -bromo carboxylic acids with an aqueous basic solution followed by an acidic work-up produces α -hydroxy carboxylic acids.
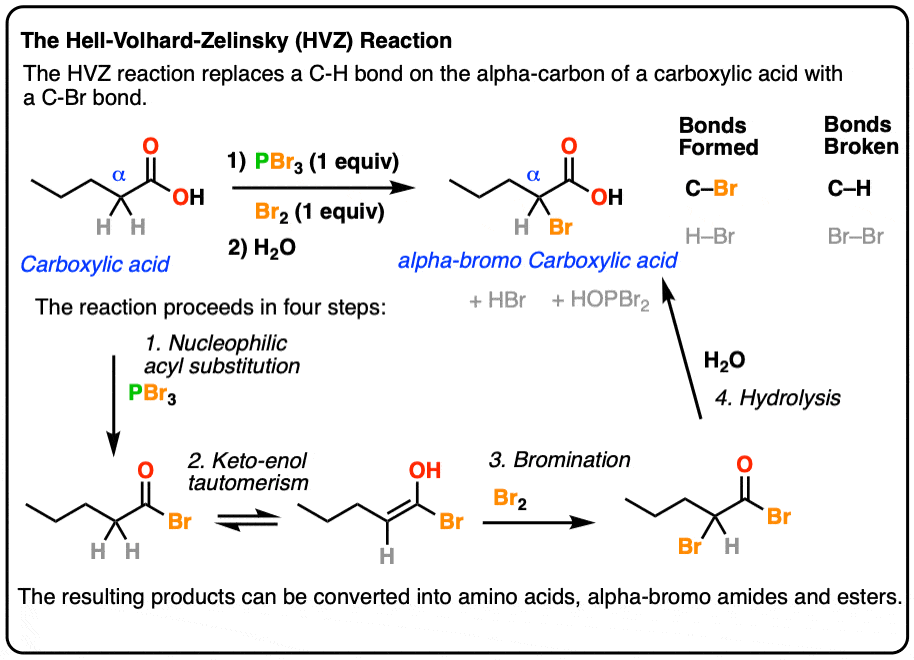
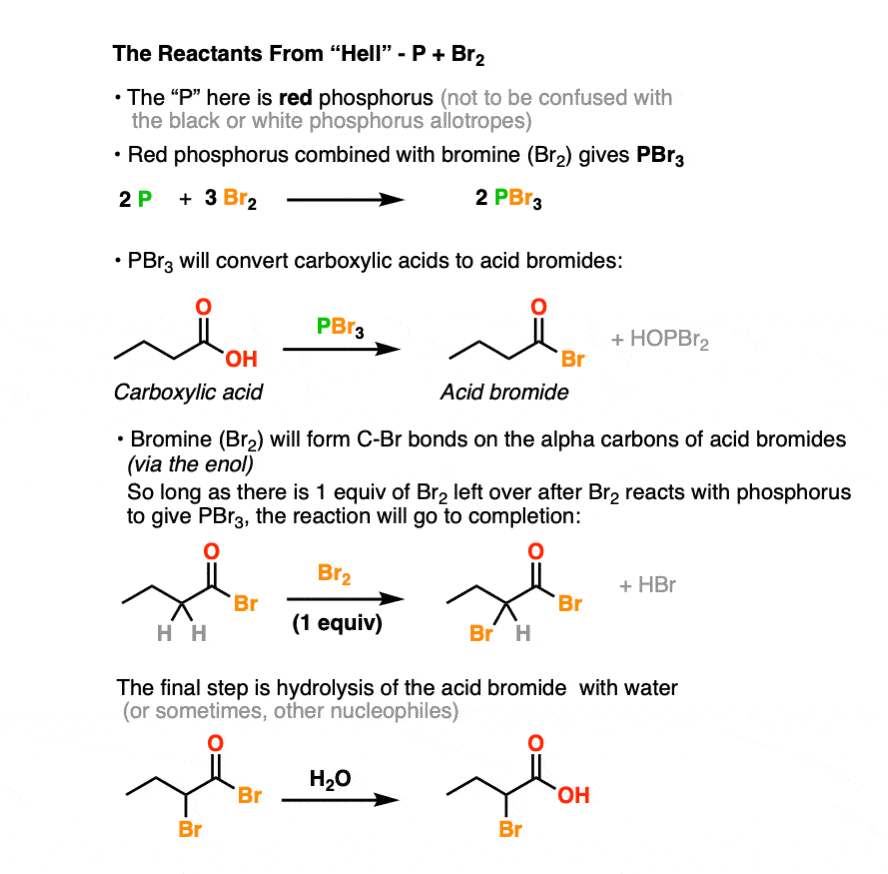
What happens when carboxylic acid reacts with PBR 3?
The reaction stats with the reaction of the carboxylic acid with PBr 3 to form the acid bromide and HBr. The HBr then catalyzes the formation of the acid bromide enol which subsequently reacts with Br 2 to give alpha bromination. Lastly, the acid bromide reacts with water to reform the carboxylic acid. Explain why the following reaction occurs.
Can a carboxylic acid be substituted with a bromine phosphorous tribromide?
We could make an amino acid, starting from this carboxylic acid. This is a three carbon carboxylic acid. This is propanoic acid. If we react propanoic acid with, once again, a bromine phosphorous tribromide and then water, this is the HVZ reaction, which we know allows us to substitute the alpha position.
|
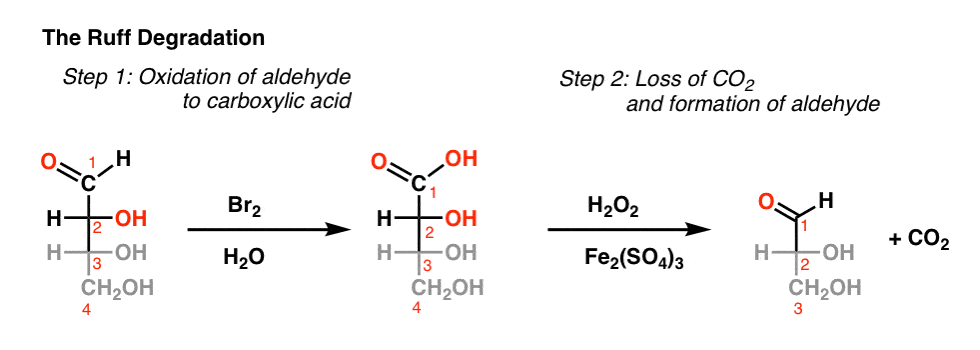
Mechanism of Oxidative Decarboxylation of ?-Hydroxy Acids by
keto acid 2 is not an intermediate and the reaction can often be stopped at the aldehyde 3 stage oxidation of mandelic acid by bromine water p H = 8.5 |
|
THE BROMINE DEGRADATION OF THE SILVER SALTS OF BILE
The reaction of bromine with the silver salt of a carboxylic acid to yield (20 per cent excess) of silver nitrate in 150 cc. of water was. |
|
Testsforfunctionalgroups - inorganiccompounds
These compounds undergo addition reaction with bromine water or the solution of bromine in carbon tetrachloride chloroform or glacial acetic acid. |
|
NZQA - NCEA Level 2 Chemistry (91165) 2019
Compound B reacting with bromine water will be a slow reaction requiring with C which is a carboxylic acid in an acid-base reaction / neutralisation. |
|
Lab 14: Qualitative Organic Analysis
deprotonated by NaOH to form water-??soluble alkoxides. Only strong acids like carboxylic acids will react with NaHCO3. Compounds that are not soluble in |
|
Tracking the formation of new brominated disinfection by- products
to the bromination of surfactant degradation products like sulfophenyl carboxylic acids (SPC) or. 36 even hydroxylated SPCs. Despite the identification of |
|
Surface Modification of Polyethylene through Bromination
hydroxyl carbonyl |
|
Bromination of Cinnamic acid
stilbene (in acetic acid solution) with bromine (added dropwise Temperature reaction is easily controlled and the cold water bath is rarely necessary. |
|
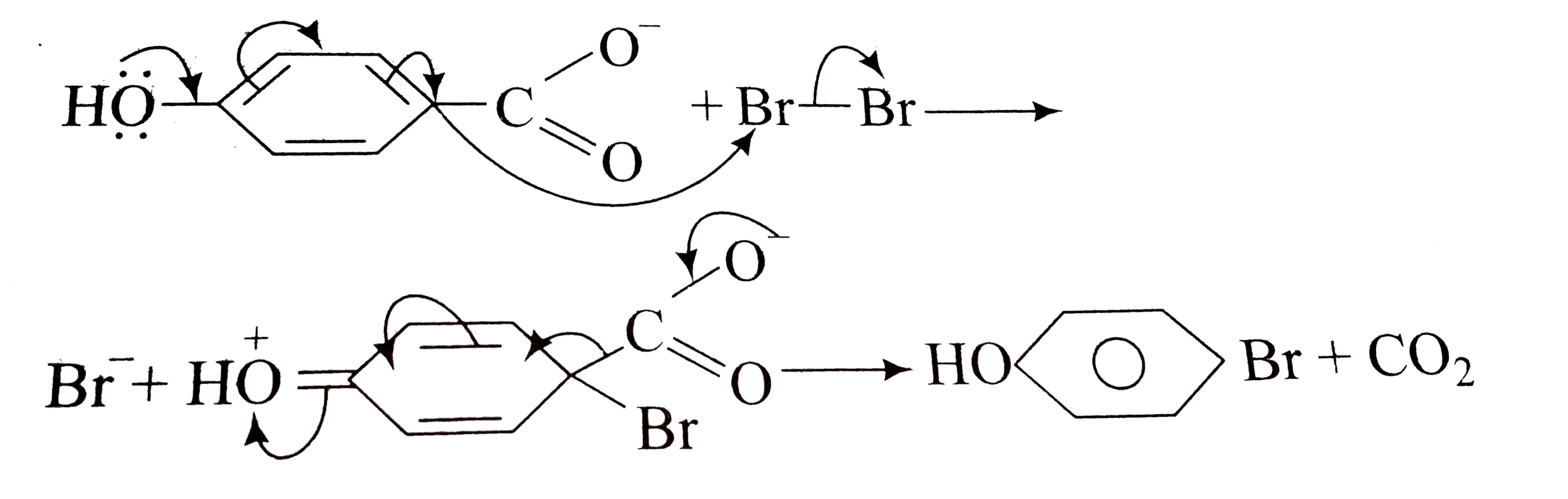
Mechanism of Oxidative Decarboxylation of a-Hydroxy Acids by
The oxidative pattern of a-hydroxy acids by acidic bromine water is shown to be similar to the oxalic acid - bromine water reaction. |
|
NZQA - NCEA Level 2 Chemistry (91165) 2013
bromine water for one liquid correct. In (b):. Water used to water by reacting fresh samples with bromine water. ... carboxylic acid. |
|
Reaction with bromine water, Br2(aq)? - No Brain Too Small
Reaction with bromine water, Br2(aq)? Amine: turns red litmus paper blue; blue litmus stays blue Amines turn green UI solution blue Carboxylic acid: turns blue litmus paper red; red litmus stays red |
|
Unit 5 Chemistry - Pearson Schools and FE Colleges
ethanoic acid (a carboxylic acid) 1 Which molecules are soluble in water? 2 Which molecule will react with bromine, turning it colourless? 3 Which molecule is |
|
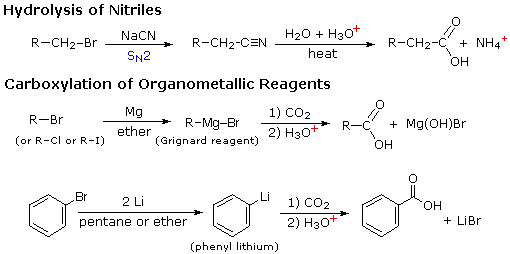
224 ALPHA BROMINATION OF CARBOXYLIC ACIDS
20 sept 2019 · 1 write an equation to illustrate the Hell-Volhard-Zelinskii reaction formed from the reaction of a given carboxylic acid with bromine and phosphorus Lastly, the acid bromide reacts with water to reform the carboxylic acid |
|
116 Chapter 22 Carbonyl Alpha-Substitution Reactions Tautomers
22 4: Alpha Bromination of Carboxylic Acids: The Hell–Volhard–Zelinskii (HVZ) Reaction α-bromination of a carboxylic acid OH C C H O Br2, PBr3, AcOH |
|
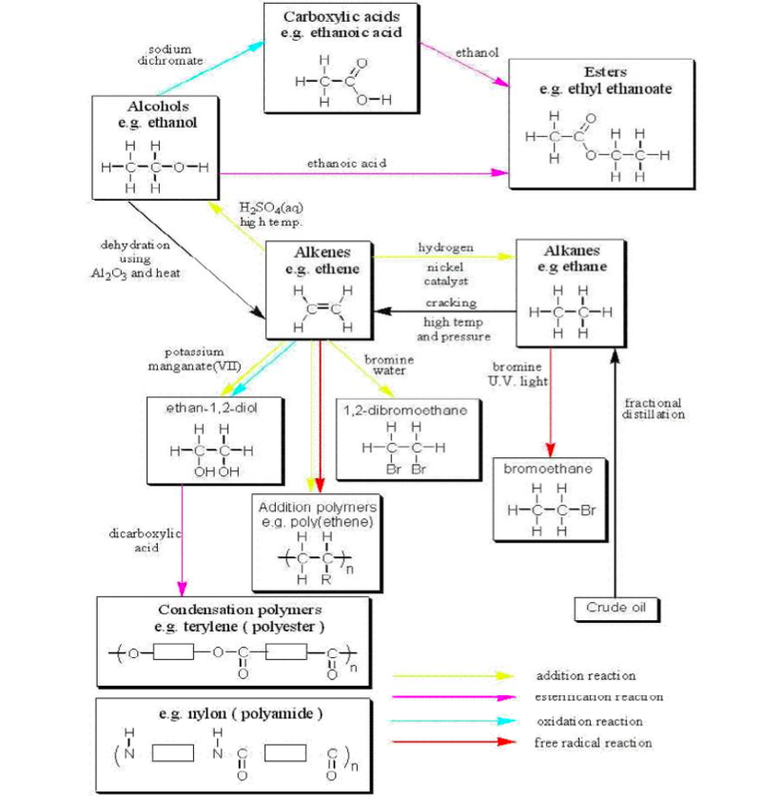
Alcohols Alkenes Carboxylic acids
sodium ethanoate + water + carbon dioxide Carboxylic acids dissolve in water Carboxylic acids react acid catalyst Chlorine, bromine or iodine can be added |
|
THE BROMINE DEGRADATION OF THE SILVER - ScienceDirect
The reaction of bromine with the silver salt of a carboxylic acid to yield an alkyl bromide of 12 9 gm (20 per cent excess) of silver nitrate in 150 cc of water was |





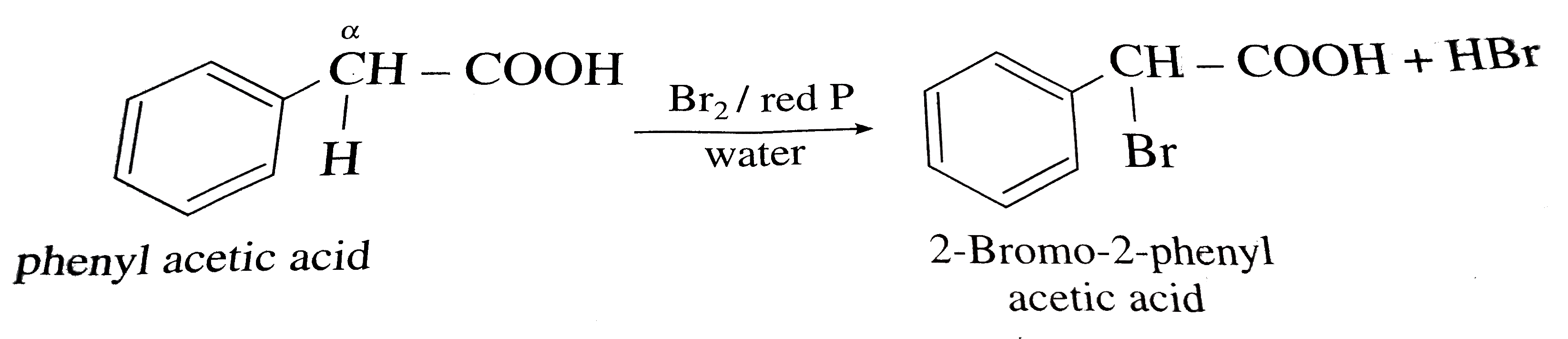




![12 Aldehydes Ketones and Carboxylic acids - [PDF Document] 12 Aldehydes Ketones and Carboxylic acids - [PDF Document]](https://d10lpgp6xz60nq.cloudfront.net/q-thumbnail/23559201.png)













![Organic Reagents - [PDF Document] Organic Reagents - [PDF Document]](https://www.vedantu.com/question-sets/3c2ebad6-49c6-41a6-b400-ab3a4d229ca98660327638115868396.png)