absorica isotretinoin
|
ACCUTANE (isotretinoin capsules) Rx only CAUSES BIRTH
(isotretinoin capsules). Rx only. CAUSES BIRTH. DEFECTS. DO NOT GET. PREGNANT. CONTRAINDICATIONS AND WARNINGS. Accutane must not be used by female patients |
|
O ABSORICA LD is 0.4 to 0.8 mg/kg/day given in two divided doses
ABSORICA LD™ (isotretinoin) capsules for oral use. • Hypersensitivity to this product or any of its components (4.2 |
|
Regard to meals for 15 to 20 weeks (2.1) - o ABSORICA LD is 0.4 to
ABSORICA/ABSORICA LD safely and effectively. See full prescribing information for ABSORICA/ABSORICA LD. ABSORICA® (isotretinoin) capsules for oral use. |
|
Absorica (isotretinoin) Capsules
ABSORICA™ (isotretinoin) capsules for oral use. Initial U.S. Approval: 1982. WARNING: CAUSES BIRTH DEFECTS. See full prescribing information for complete |
|
ACCUTANE (isotretinoin capsules) Rx only CAUSES BIRTH
Accutane must not be used by female patients who are or may become pregnant. There is an extremely high risk that severe birth defects will result if pregnancy |
|
Absorica* (isotretinoin) Absorica LD* (isotretinoin micronized
Absorica* (isotretinoin) Absorica LD* (isotretinoin micronized) Isotretinoin is indicated for the treatment of severe recalcitrant nodular acne. |
|
Accutane (isotretinoin) Capsules Label
20 juin 2002 (isotretinoin). CAPSULES. CAUSES BIRTH DEFECTS. DO NOT GET PREGNANT. CONTRAINDICATIONS AND WARNINGS: Accutane must not be used by. |
|
MEDICATION GUIDE ABSORICA (ab-sore-i-kah) (isotretinoin
IMPORTANT: ABSORICA LD is not the same as ABSORICA or other isotretinoin generic products. Do not change between ABSORICA LD and ABSORICA or other |
|
Dermatologics: Acne Products - Isotretinoin
1 déc. 2021 Isotretinoin is a synthetic analogue of vitamin A and is FDA indicated for the ... Absorica LD (isotretinoin). Accutane (isotretinoin). |
|
NDA 18-662/S-056 Page 11 ACCUTANE (isotretinoin capsules) Rx
(isotretinoin capsules). Rx only. CAUSES BIRTH. DEFECTS. DO NOT GET. PREGNANT. CONTRAINDICATIONS AND WARNINGS. Accutane must not be used by female patients |
|
Absorica (isotretinoin) Capsules - FDA
Absorica™ is a retinoid indicated for the treatment of severe recalcitrant nodular acne in patients 12 years of age and older Nodules are inflammatory lesions with |
|
Prescribing Information - Absorica
ABSORICA LD™ (isotretinoin) capsules, for oral use Initial U S ABSORICA LD are reserved for patients with severe nodular acne who are unresponsive to |
|
Absorica - UHCprovidercom
1 août 2020 · Prior Authorization/Medical Necessity Medications Absorica*, Absorica LD* ( isotretinoin) P&T Approval Date 12/2018, 12/2019, 5/2020 |
|
Sun Pharma Introduces ABSORICA LD™(isotretinoin) Capsules for
4 fév 2020 · owned subsidiaries has launched ABSORICA LD™ (isotretinoin) capsules in U S for the management of severe recalcitrant nodular acne in |
|
Absorica LD (isotretinoin) - Professionals OptumRX
(isotretinoin) – New formulation approval • On November 5, 2019, the FDA approved Sun Pharmaceuticals' Absorica LD (isotretinoin) capsules, for the treatment |
|
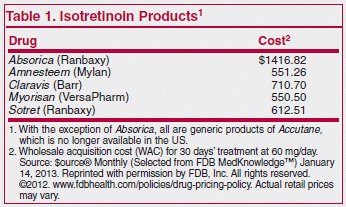
Medications Comments Absorica (isotretinoin) N/A Amnesteem
Comments Absorica (isotretinoin) N/A Amnesteem (isotretinoin) N/A I Isotretinoin may be approved for the treatment of acne if the following criteria are met: |
|
In Brief: Absorica for Acne - JAMA Network
The oral retinoid isotretinoin (Accutane, and others) is the most effective drug available for treatment of acne 1 The FDA has recently approved a new oral |
|
Lisotrétinoϊne (Accutane®) - CHEO
P5605F janvier 2012 L'isotrétinoϊne (Accutane®) L'isotrétinoϊne est utilisée pour le traitement de l'acné sévère, lorsque les autres traitements ont échoué |
|
MONOGRAPHIE COMPRENANT LES - Roche Canada
ACCUTANE™ ROCHE® (isotrétinoïne) est indiqué pour le traitement de : • l'acné nodulaire et/ou inflammatoire sévère • l'acne conglobata • l'acné réfractaire |