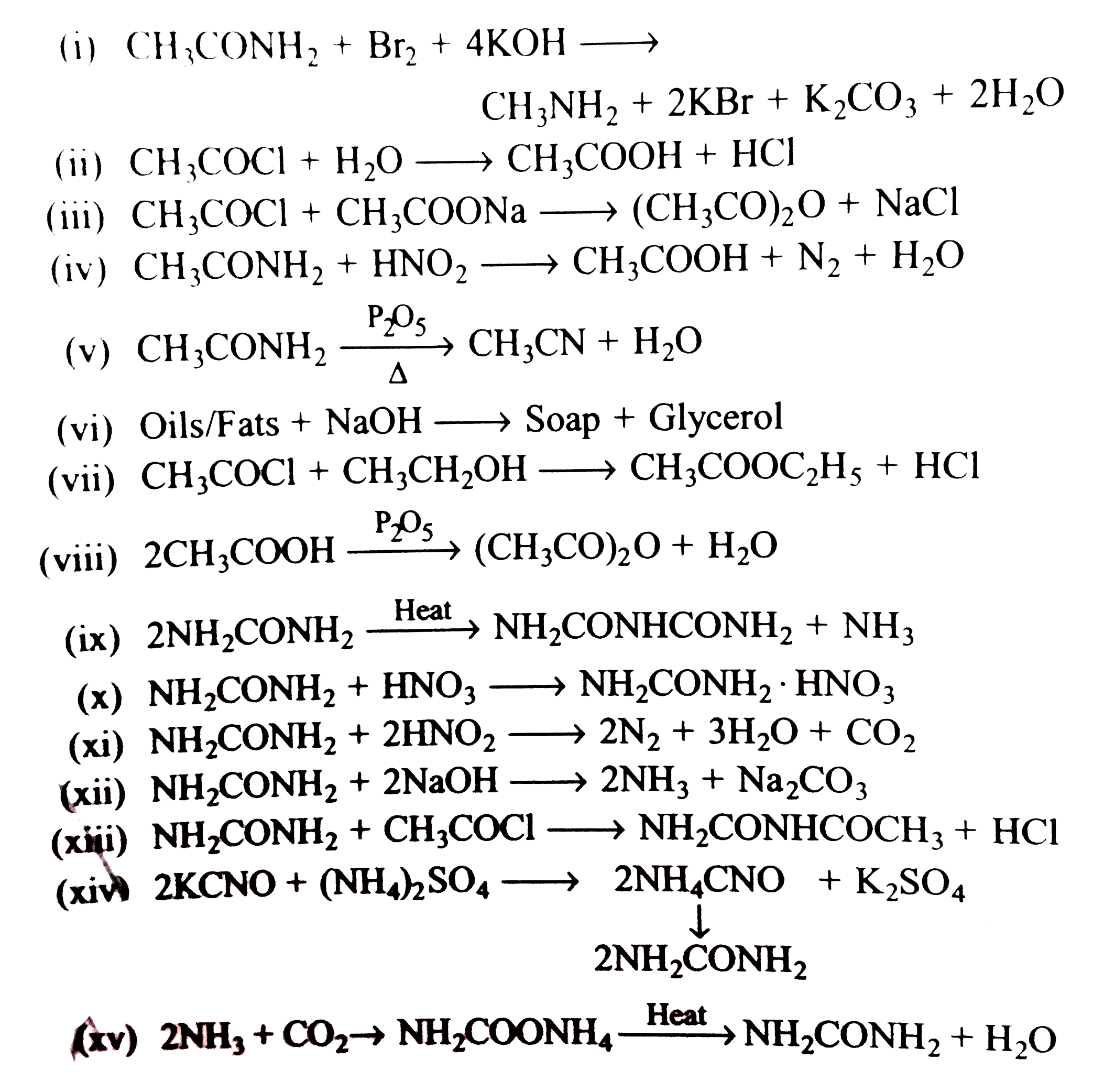acetamide with naoh
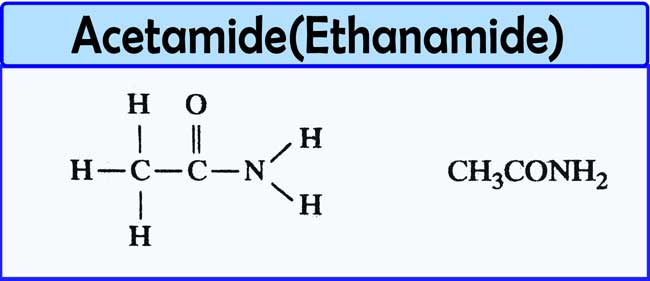
How do you make acetamide?
Alternatively acetamide can be obtained in excellent yield via ammonolysis of acetylacetone under conditions commonly used in reductive amination. It can also be made from anhydrous acetic acid, acetonitrile and very well dried hydrogen chloride gas, using an ice bath, alongside more valuable reagent acetyl chloride.
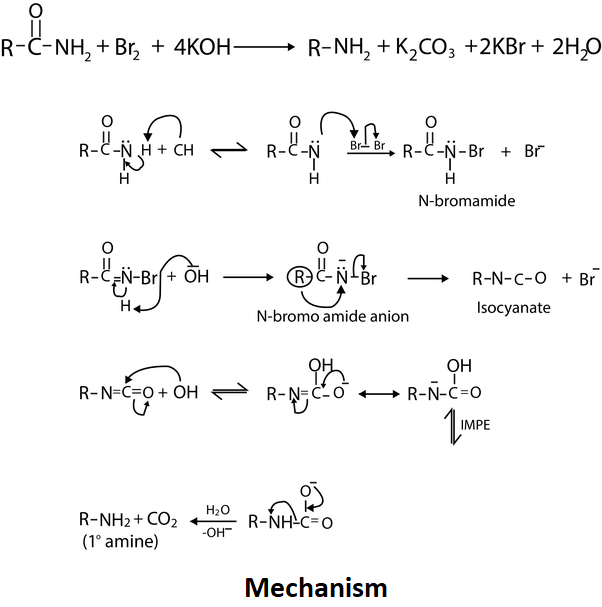
What happens if acetamide is heated with sodium hydroxide solution?
Also, if acetamide is heated with sodium hydroxide solution, ammonia gas is given off and you are left with a solution containing sodium acetate. Peptide hydrolysis of proteins is amide hydrolysis. What biologists and biochemists call a peptide link (in proteins, for example) is what chemists call an amide link.
|
Dissolution of Cellulose in NaOH Based Solvents at Low Temperature
Abstract: Three low-cost types of complex solvents systems were carried out to dissolve cellulose which were NaOH/urea/ acetamide |
|
Studies on Chitin. V. Kinetics of Deacetylation Reaction
5. An alkali chitin solution was prepared with a polymer con- centration of 196 and an NaOH concentration the acetamide group and sodium hydroxide. However ... |
|
Standard Input
energies for the reaction: hydroxymethylacetamide → acetamide + formaldehyde. NaOH Methylol Methylol and Acet-. 104 k₁ sec.*¹ 10ªk. K litres E l/mole mole. |
|
NN-Dimethyl acetamide detector tube No.184 GASTEC
CH3CON(CH3)2 + NaOH → R・NH2. 2R・NH2 + H2SO4 →(R・NH3)2SO4. Reaction principle. Diffusion tube method. Calibration gas generation. Possible coexisting |
|
Multiple Choice Questions Unit (9) Chem-100
When ______ reacts with NaOH the product is sodium benzoate. A. benzoic acid. B The missing chemical in the reaction (acetamide + sodium hydroxide ? + ... |
|
Influence of pressure on the equilibrium of hydration of aliphatic
reported for acid- and base-catalyzed hydrolysis of acetic acid esters and for acid-catalyzed hydrolysis of acetamide. NaOH. 2.4. -4.5 e. NaOH. 25. -8.8 a. |
|
Chemical Compatibility Guide
Acetamide. A. D. D. D. A. A. B. B. A. B. B. A. A. D. -. A. A. B. B. A. A. D. A. A. D. Acetate Solvents ... (Sodium Hydroxide). D. D. D. B. B. D. B. A. B. -. B. -. |
|
Chapter 6 Amines and Amides
+ NaOH ---→ R—NH2 + H2O + NaCl. R. NH2. + H2O. +. -. OH. R. NH3. + base. (high pH) (acetamide). O. 42. Examples: Nomenclature of Amides. • Name the following ... |
|
Page 1 of 4 Enzymatic Assay of AMIDASE (EC 3.5.1.4) PRINCIPLE
Jul 12 1994 Acetamide + Hydroxylamine Amidase > Acethydroxamate + NH3. CONDITIONS: T = 37 ... Adjust to pH 7.2 at 37°C with 1 M NaOH.) B. 400 mM Acetamide ... |
|
CATALYSIS OF THE FORMATION AND HYDROLYSIS OF
The solution containing the acetamide was then transferred to a flask a solution con- taining about 3.3 g. of sodium hydroxide added and the flask connected |
|
Page 1 of 4 Enzymatic Assay of AMIDASE (EC 3.5.1.4) PRINCIPLE
Jul 12 1994 Acetamide + Hydroxylamine Amidase > Acethydroxamate + NH3. CONDITIONS: T = 37°C |
|
Studies on Chitin. V. Kinetics of Deacetylation Reaction
Deacetylation / Acetamide Group / Amino Group / centration of 196 and an NaOH concentration of 1096. ... the acetamide group and sodium hydroxide. |
|
Multiple Choice Questions Unit (9) Chem-100
When ______ reacts with NaOH the product is sodium benzoate. A. benzoic acid The missing chemical in the reaction (acetamide + sodium hydroxide ? |
|
Standard Input
The rate of formation of hydroxymethylacetamide from acetamide and formaldehyde has been measured in aqueous solution at different sodium hydroxide |
|
Acid- and Base-Mediated Hydrolysis of Dichloroacetamide
Dec 17 2021 in basic environments compared to the other dichloroacetamide safeners shown in (C) with 0.5 N NaOH. Rate constants for safener hydrolysis. |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VIII
The volume of activation for acetamide in concentrated acid provides no added and the solution was titrated with 2-N sodium hydroxide solution until the ... |
|
A Study of the Mechanism and Kinetics of the Thioacetamide
The relative importance of thioacetic acid and acetamide as intermediates is not known. 0.10 N NaOH:A after 2.2 minutes; B |
|
Influence of pressure on the equilibrium of hydration of aliphatic
Acetamide. NaOH. 25. -14.2 a. HC104. 55. -9.4 h. Propionamide. NaOH mediate.24 The base-catalyzed hydrolysis of acetamide. |
|
Chapter 6 Amines and Amides
Examples: Basicity of Amines. • Complete the following reactions: CH3NH2 + HCl. NHCH3. + HCl. N. CH3. CH3. H. CH3. + NaOH. |
|
Appendix – I
16 fév 2017 · -Hydroxy phenyl acetamide is react with Epichlorohydrin and Isopropylamine in presence of Sodium hydroxide base and Isopropyl alcohol as |
|
Reactions of Amines
NaOH amine base ammonium salt (acidic) • Mechanism: Required (protonation) • Reverse Mechanism: Required (deprotonation) • Amines are completely |