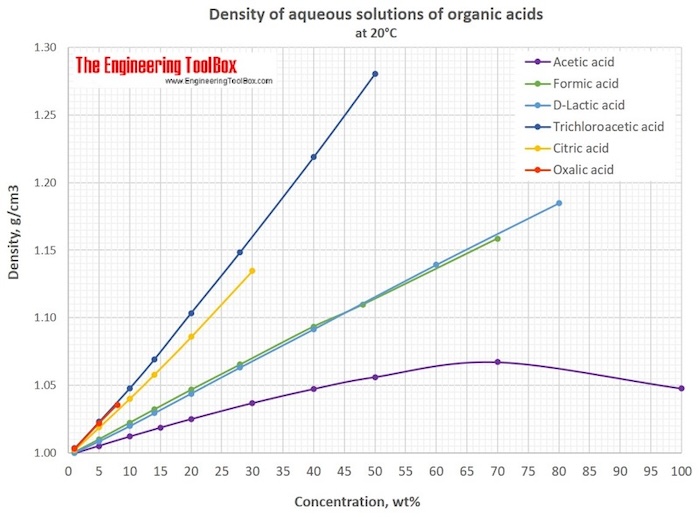
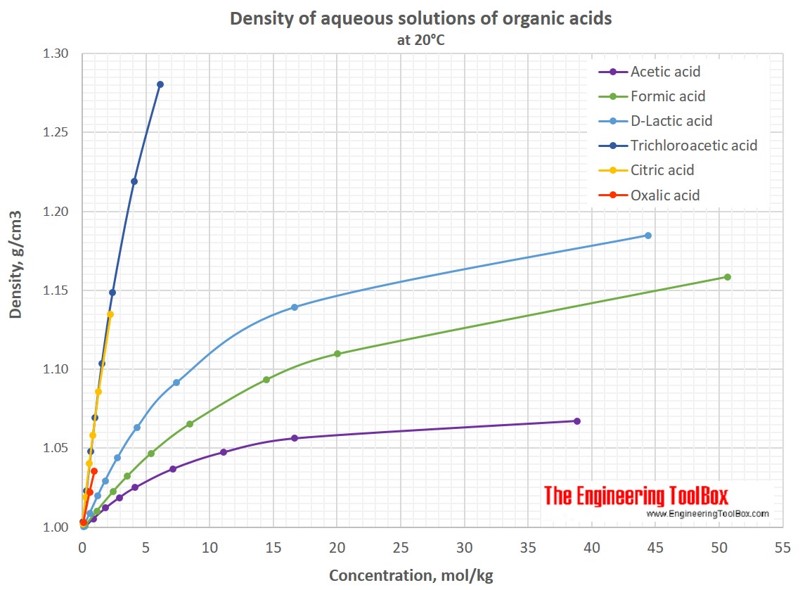
acetic anhydride density ml
How high is acetic anhydride IDLH?
Because 2,000 ppm is obviously too high to be selected as the IDLH, 1,000 ppm has been chosen. . . . Basis for revised IDLH: The revised IDLH for acetic anhydride is 200 ppm based on acute inhalation toxicity data in animals [Deichmann and Gerarde 1969]. This may be a conservative value due to the lack of relevant acute toxicity data for workers.
Why is acetic anhydride a nonplanar molecule?
Acetic anhydride, like most acid anhydrides, is a flexible molecule with a nonplanar structure. The pi system linkage through the central oxygen offers very weak resonance stabilization compared to the dipole-dipole repulsion between the two carbonyl oxygens.
What is the rate constant for acetic anhydride vapor-phase reaction?
Environment Canada; Tech Info for Problem Spills: Acetic Anhydride (Draft) p.31 (1982) The rate constant for the vapor-phase reaction of acetic anhydride with photochemically-produced hydroxyl radicals has been estimated as 8.4X10-14 cu cm/molecule-sec at 25 °C (SRC) using a structure estimation method (1).
|
Labeling of high density lipoproteins with [3H] acetic anhydride
[3H]acetic anhydride at pH 7.2 for 30 min at room tempera- mg/ml (Table 1) was reacted with r3HIacetic anhydride as follows. |
|
ACETIC ANHYDRIDE CAS N°: 108-24-7
Acetic anhydride is used exclusively as a chemical intermediate and there is Other data e.g. relative density |
| 8-Synthesis-of-Aspirin.pdf |
|
Friday Worksheet Name: ………………. Analytical chemistry and
When acetic anhydride is used in place of acetic acid |
|
GENERAL TESTS PROCESSES AND APPARATUS
mination of bulk and tapped densities digestion test |
|
Synthesis of Acetaminophen
Using an automatic pipet (or a dispensing pump or a graduated pipet) add. 0.450 mL of water and 0.165 mL of acetic anhydride (MW= 102.1 |
|
Experiment 5 - Synthesis of Aspirin
Acetic anhydride stand with iron ring distilled water. 85% phosphoric acid wire gauze ice bath. 50 mL flask beaker of tap water filter paper. Büchner funnel. |
|
Radiolabeling of proteins and viruses in vitro by acetylation with
[r4C]acetic anhydride dissolved in small amounts of anhydrous dioxane. per ml of reaction mix- ture typically yields an acetylation density. |
|
Chem 11 Lab Manual
to a 250-mL graduated cylinder and determine the exact volume. 2.0 grams of salicylic acid and 5.0 mL of acetic anhydride (density = 1.08 g/mL) are ... |
|
1 University of Wisconsin Chemistry 116 Preparation and
If acetic anhydride is used instead of acetic acid the reaction Acetic anhydride (MW = 102 g/mol |
|
Acetic anhydride Product Number A6404 Store at - Sigma-Aldrich
1 Density: 1 082 g/ml 1 Molarity: 10 6 M (calculated from density and molecular weight) Synonyms: acetic oxide, acetyl oxide 1 Acetic anhydride is a reagent |
|
Acetic Anhydride
Acetic anhydride is a clear, colorless liquid with a very pungent Vapor density ( air = 1) 3 5 Density at 20°C g/ml 1 08 Solubility in water at 20°C g/L 20 |
|
Acetic Anhydride - OSHA
Acetic anhydride vapors may be irritating to the eyes, nose and throat Inhalation of vapors may applying 0 5 mL of a solution of 5 0 mg/mL 1-2PP in methylene chloride to each glass fiber 102 09 = molecular weight of acetic anhydride 3 8 |
|
Acetic Anhydride SAFETY DATA SHEET Section 2 Hazards
13 nov 2018 · When sold in quantities of less than or equal to 1 mL, or 1 g, with an Excepted Quantity Code of E1, E2, E4, or E5, this item meets the De Minimis |
|
Safety Data Sheet: Acetic anhydride - Carl Roth
Acetic anhydride ≥99 for synthesis Labelling of packages where the contents do not exceed 125 ml Signal word: Danger Molar mass 102 1 g/mol |
|
8-Synthesis of Aspirin
If acetic anhydride is used instead of acetic acid, the reaction is much faster and has a In the hood, measure out 3 0 mL of acetic anhydride in a small graduated cylinder The melting point of pure aspirin is 135°C, and the melting point of |
|
Chem 11 Lab Manual - Moorpark College
2 0 grams of salicylic acid and 5 0 mL of acetic anhydride (density = 1 08 g/mL) are mixed to produce aspirin Determine the percentage yield of the reaction if 1 9 g |
|
Chem 1B Lab Manual - Moorpark College
characterized by its melting point and by infrared spectroscopy next operation in the fume hood: pipet 1 0 mL of acetic anhydride into the flask in such a |
|
Experiment 5 - Synthesis of Aspirin
To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride The melting point range of pure aspirin is 138-140 °C and the melting point While the flask is still in the water bath, slowly add 2 mL of distilled water to the |


![Download PDF - Acetic Anhydride [19n0zyv1p54v] Download PDF - Acetic Anhydride [19n0zyv1p54v]](https://demo.vdocuments.mx/img/378x509/reader018/reader/2020012114/586e14d21a28abe2218b75cf/r-1.jpg)
![Acetic anhydride (SIDS) - [PDF Document] Acetic anhydride (SIDS) - [PDF Document]](https://i1.rgstatic.net/publication/280246774_A_Green_Process_for_Acetic_Acid_Production/links/55af775108ae6aa568b3b89a/largepreview.png)













![Acetic anhydride (SIDS) - [PDF Document] Acetic anhydride (SIDS) - [PDF Document]](https://ars.els-cdn.com/content/image/3-s2.0-B9780444639929000112-f11-03-9780444639929.jpg)